Articles
- Page Path
- HOME > J Mov Disord > Volume 17(2); 2024 > Article
-
Original Article
Comparing Montreal Cognitive Assessment Performance in Parkinson’s Disease Patients: Age- and Education-Adjusted Cutoffs vs. Machine Learning -
Kyeongmin Baek1*
 , Young Min Kim2*
, Young Min Kim2* , Han Kyu Na2,3
, Han Kyu Na2,3 , Junki Lee1
, Junki Lee1 , Dong Ho Shin4
, Dong Ho Shin4 , Seok-Jae Heo5
, Seok-Jae Heo5 , Seok Jong Chung2,6,7
, Seok Jong Chung2,6,7 , Kiyong Kim8
, Kiyong Kim8 , Phil Hyu Lee2,3
, Phil Hyu Lee2,3 , Young H. Sohn2,3
, Young H. Sohn2,3 , Jeehee Yoon1
, Jeehee Yoon1
 , Yun Joong Kim2,6,7
, Yun Joong Kim2,6,7

-
Journal of Movement Disorders 2024;17(2):171-180.
DOI: https://doi.org/10.14802/jmd.23271
Published online: February 13, 2024
1Department of Computer Engineering, Hallym University, Chuncheon, Korea
2Department of Neurology, Yonsei University College of Medicine, Seoul, Korea
3Department of Neurology, Severance Hospital, Yonsei University Health System, Seoul, Korea
4Massachusetts College of Pharmacy & Health Sciences, Boston, USA
5Department of Biomedical Systems Informatics, Yonsei University College of Medicine, Seoul, Korea
6Department of Neurology, Yongin Severance Hospital, Yonsei University Health System, Yongin, Korea
7YONSEI BEYOND LAB, Yongin, Korea
8Department of Electronic Engineering, Kyonggi University, Suwon, Korea
- Corresponding author: Yun Joong Kim, MD, PhD Department of Neurology, Yongin Severance Hospital, 363 Dongbaekjukjeon-daero, Giheung-gu, Yongin 16995, Korea / Tel: +82-31-5189-8140 / Fax: +82-31-5189-8565 / E-mail: yunjkim@yuhs.ac
- Corresponding author: Jeehee Yoon, PhD Department of Computer Engineering, Hallym University, 1 Hallymdaehak-gil, Chuncheon 24252, Korea / Tel: +82-33-248-2316 / Fax: +82-33-242-2524 / E-mail: jhyoon@hallym.ac.kr
- *These authors contributed equally to this work.
Copyright © 2024 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 582 Views
- 60 Download
ABSTRACT
-
Objective
- The Montreal Cognitive Assessment (MoCA) is recommended for general cognitive evaluation in Parkinson’s disease (PD) patients. However, age- and education-adjusted cutoffs specifically for PD have not been developed or systematically validated across PD cohorts with diverse education levels.
-
Methods
- In this retrospective analysis, we utilized data from 1,293 Korean patients with PD whose cognitive diagnoses were determined through comprehensive neuropsychological assessments. Age- and education-adjusted cutoffs were formulated based on 1,202 patients with PD. To identify the optimal machine learning model, clinical parameters and MoCA domain scores from 416 patients with PD were used. Comparative analyses between machine learning methods and different cutoff criteria were conducted on an additional 91 consecutive patients with PD.
-
Results
- The cutoffs for cognitive impairment decrease with increasing age within the same education level. Similarly, lower education levels within the same age group correspond to lower cutoffs. For individuals aged 60–80 years, cutoffs were set as follows: 25 or 24 years for those with more than 12 years of education, 23 or 22 years for 10–12 years, and 21 or 20 years for 7–9 years. Comparisons between age- and education-adjusted cutoffs and the machine learning method showed comparable accuracies. The cutoff method resulted in a higher sensitivity (0.8627), whereas machine learning yielded higher specificity (0.8250).
-
Conclusion
- Both the age- and education-adjusted cutoff methods and machine learning methods demonstrated high effectiveness in detecting cognitive impairment in PD patients. This study highlights the necessity of tailored cutoffs and suggests the potential of machine learning to improve cognitive assessment in PD patients.
- Cognitive impairment is one of the most common and significant nonmotor symptoms of Parkinson’s disease (PD). Mild cognitive impairment (MCI) affects 20%–50% of patients with PD, 15%–20% of whom experience it at the time of diagnosis [1,2]. Cross-sectional studies have indicated that the prevalence of dementia in PD patients is approximately 30%, and the cumulative prevalence over an 8- to 12-year follow-up period has been reported as 60%–83% [1-4]. For the diagnosis of MCI or dementia in PD patients, the Movement Disorders Society (MDS) Task Force has proposed the following two sets of tests: a practical set (Level I) for screening for cognitive impairment and a second set (Level II) consisting of comprehensive neuropsychological tests that assess individual cognitive domains [5,6]. The Montreal Cognitive Assessment (MoCA), originally developed as a screening tool for MCI in the general population [7], is recommended as a Level I test due to its reliability and validity [6,8].
- The selection of an optimal cutoff for a diagnostic screening test hinges on balancing sensitivity and specificity to suit the requirements of a particular study or clinical context. Nasreddine et al. [7] initially recommended a cutoff score of 26 for identifying MCI; however, a lower cutoff of 23 was proposed following a recent meta-analysis [9]. In the context of memory clinic outpatients, two distinct cutoff points were suggested: a specific cutoff of 24 (92% specificity) and a sensitive cutoff of 26 (91% sensitivity) [10]. Most studies proposing a cutoff have relied on normative data from either a general population cohort or patients with memory impairments [10,11].
- Total MoCA scores are independently influenced by age, education level, race, and ethnicity [11-13]. In PD, previous studies in English-speaking populations have reported good efficacy of using a single MoCA cutoff score between 24 and 26 for differentiating MCI and/or dementia from normal cognition [14-18]. However, in many non-English speaking populations, which exhibit a wide range of educational levels, cutoff scores vary significantly depending on the study, suggesting that a single cutoff may not be universally applicable [19,20]. In a recent study focusing on less educated patients with PD, cutoff scores of 19 or lower were proposed for those with 9 years of education or less [20]. In non-English-speaking populations, cutoffs for cognitive impairment in PD patients have not been systematically validated. These studies are limited by the small number of PD patients, wide ranges of education levels or ages, and use of cohorts that may not be directly applicable to PD [19]. Using MoCA domain scores from patients with PD in the Parkinson’s Progression Markers Initiative (PPMI) data repository, we previously demonstrated that machine learning analysis could be used to classify cognitive impairment in PD patients with a cutoff of 26 [21]. However, to date, a comparison between age- and education-adjusted cutoff scores and machine learning analyses in non-English-speaking populations has not been made.
- In this study, we developed age- and education-adjusted cutoffs for patients with PD whose cognitive diagnoses were determined through comprehensive neuropsychological assessment. We conducted a validation study in independent PD cohorts and compared the efficacy of machine learning analysis with age- and education-adjusted cutoffs in identifying cognitive impairment in PD patients.
INTRODUCTION
- Participants
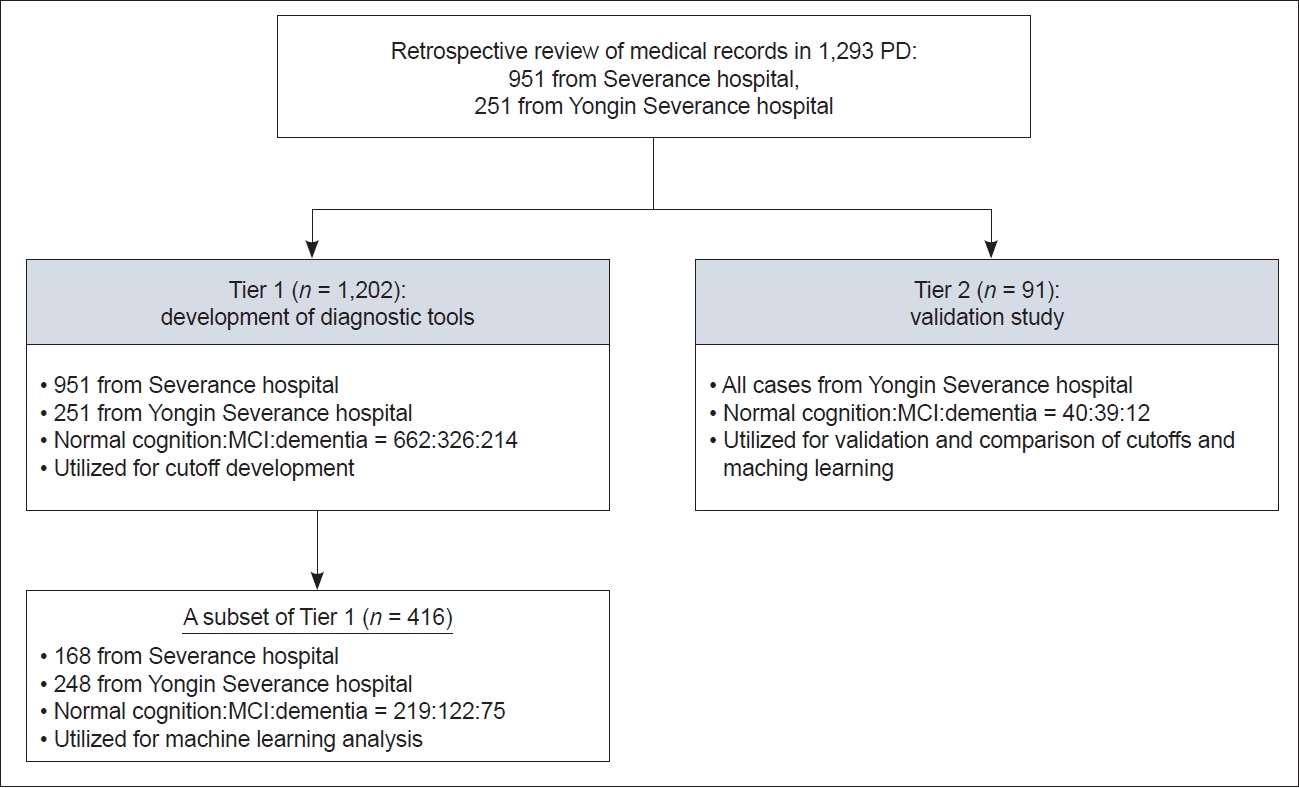
- We conducted a retrospective review of medical records of PD patients diagnosed according to the MDS’s clinical diagnostic criteria [22]. These patients had undergone comprehensive neuropsychological tests and the MoCA in two referral hospitals. The exclusion criteria were as follows: 1) had structural brain lesions related to cognitive changes, such as multiple lacunes in the basal ganglia, old cerebrovascular lesions, intracranial hemorrhage, or hydrocephalus; 2) had severe white matter changes; or 3) used anticholinergic medication. This study received approval from the Yonsei University Yongin Severance Hospital Institutional Review Board (No. 9-2021-0181). Informed consent was waived because of the retrospective nature of the study and the analysis used anonymous clinical data. A total of 1,293 PD patients were enrolled and divided into two data tiers based on the subsets and specific objectives (Figure 1). Tier 1 included 1,202 PD patients (951 patients from Severance Hospital and 251 patients from Yongin Severance Hospital). A subset of Tier 1 patients, consisting of 416 PD patients (168 patients from Severance Hospital and 248 patients from Yongin Severance Hospital; 219 with normal cognition, 122 with MCI, and 75 with dementia) of whom MoCA domain scores and all parameters for machine learning (including category-cued semantic recall scores and multiple-choice cued recall scores) were available, was used to construct the optimal machine learning model. The remaining 786 PD patients, who lacked recognition memory scores (category-cued semantic recall and multiple-choice cued recall), were not included in the machine learning test. Total MoCA scores from 1,202 PD patients were utilized to develop age- and education-adjusted cutoff values. There were 662 individuals with normal cognition and 540 with cognitive impairment (326 with MCI and 214 with dementia). Tier 2 comprised 91 consecutive PD patients (40, 39, and 12 with normal cognition, MCI, and dementia, respectively) who underwent comprehensive neuropsychological tests at Yongin Severance Hospital from January 1, 2023 to August 31, 2023. For a direct comparison of performance between machine learning and cutoff scores, the results of the MoCA, including domain scores or total scores from these 91 PD patients, were used.
- Diagnosis of cognitive status
- To evaluate cognitive performance, all patients completed the Korean version (Korean 7.1, K2-Chuncheon) of the Montreal Cognitive Assessment (K-MoCA) for initial cognitive function screening (Level I test) and the Seoul Neuropsychological Screening Battery (SNSB), a comprehensive neuropsychological test suite (Level II test). The SNSB, a standardized neuropsychological test battery extensively used in South Korea [23], is used to evaluate five cognitive domains: attention and working memory, visuospatial function, language, memory, and executive function. To assess activities of daily living (ADL) related to cognitive performance, two instrumental ADL scales were utilized: the Korean Instrumental Activities of Daily Living scale and the Seoul Activities of Daily Living scale. ADL impairment was identified when both ADL scales indicated abnormalities [24]. For diagnosing cognitive status, only the results from the SNSB and instrumental ADL scales were considered. Two neurologists and one neuropsychologist came to a consensus to diagnose PD dementia diagnosis, as previously described [25], following the clinical diagnostic criteria proposed by the MDS Task Force [5,26]. When diagnosing MCI, we adhered to the criteria set by the MDS Task Force [6], utilizing the SNSB. For MCI diagnosis, two tests represented each of the five cognitive domains: attention and working memory function were assessed using the digit span task (backward) and the Color-Word Stroop test; language, through the Korean version of the Boston Naming Test and Wechsler Adult Intelligence Scale-IV Similarities; visuospatial function, via the Rey Complex Figure Test copy and clock copying; memory, through the Seoul Verbal Learning Test; and executive function, through the Controlled Oral Word Association Test and 10-point Clock Drawing Test. Scores on each cognitive test were considered abnormal if they fell below the 1.5 standard deviation mark of the age-, sex-, and education-specific norms. A diagnosis of PD-MCI was determined if impairments in at least two tests across these five cognitive domains were demonstrated.
- Statistical analysis
- Using logistic regression, we derived age- and education-adjusted MoCA cutoffs for cognitive impairment in patients with PD [27]. A logistic regression model was constructed to predict cognitive impairment (normal cognition vs. cognitive impairment) with three independent variables: age, total MoCA score, and education level. To prevent overfitting and ensure the model’s generalizability, fivefold cross-validation was employed [28]. The logistic regression model was trained using this method, and the average regression coefficients were calculated. A receiver operating characteristic (ROC) curve, along with the area under the curve, was plotted to assess the model’s ability to differentiate between the two groups [29]. The ROC curves generated for each fold were analyzed, and an optimal average probability threshold was established. The MoCA cutoff value for each age and education level was then calculated using the logistic regression equation, incorporating the estimated optimal regression coefficients and probability threshold. The analysis was divided into different age groups: median ages ranging from 56 to 80 years, each encompassing patients within a 5-year range from the median. The age groups 51–55, 81–85, and 86–95 years were combined into one group as an exception. Additionally, years of education were divided into the following six groups: 0, 0.5–3, 4–6, 7–9, 10–12, and > 12 years. For the performance comparison of the age- and education-adjusted cutoffs, cutoffs developed from Korean patients with vascular cognitive impairment were referenced [30].
- Machine learning analysis was conducted using previously established methods, employing support vector machine (SVM), random forest (RF), and logistic regression models [21,31,32]. These models were applied to analyze detailed cognitive and biological data associated with PD. The MoCA scores were calculated in one of two ways: by calculating domain subtotal scores in the visuospatial/executive, abstraction, memory, orientation, language, and attention domains or by utilizing each of the 30 individual item scores. Additionally, the total number of words in the verbal fluency test from the MoCA and the score on the interlocking pentagon copying test from the Mini-Mental State Examination were included. The presence of depression was determined using either the Short version of the Geriatric Depression Scale or the Beck Depression Inventory. The demographic data, including age, sex, years of education, handedness, and duration of PD diagnosis, were also included as independent variables. To assess the impact of including the scores of the interlocking pentagon copying test on the results, analyses were repeated with this score added as an additional predictor variable.
- In the Tier 1 dataset designated for the machine learning test (n = 416), there was an imbalance between the number of patients with normal cognition and those with cognitive impairment. Predictions from an unbalanced dataset are often skewed toward the majority group [33]. To mitigate this bias, the dataset was balanced by randomly undersampling the majority group before training the machine learning algorithms. Subsequently, the dataset was randomly divided into training and testing subsets at an 80:20 ratio, a commonly used split in machine learning models. The proportion of patients with PD-cognitive impairments (PD-CIs) in the training and testing datasets was consistent with that in the original dataset through stratified sampling [34]. The training and testing of the model were repeated 100 times with randomly generated training and testing datasets, and the average model accuracy was computed across all iterations. The evaluation of the classification models was based on sensitivity, specificity, positive predictive value, and negative predictive value.
- Feature importance scores are utilized to assess the relative significance of each feature when constructing a prediction model. These scores are calculated based on the machine learning algorithm used [35]. In the case of a linear SVM, feature importance is determined by the coefficient statistics correlating each feature with the output (dependent) variable [36]. The feature scores provided by the SVM model were averaged across all trained models to arrange the features in descending order of importance. Negative scores are indicative of features that are crucial for classifying a patient as having normal cognition, while positive scores denote features associated with cognitive impairment.
- All analyses were conducted using Python version 3.9.13, along with the scikit-learn 1.3.0 package (Python Software Foundation, Beaverton, OR, USA).
- Data availability
- To facilitate replication of the procedures and results, qualified investigators may request anonymized data following ethics clearance and approval from the corresponding author.
MATERIALS & METHODS
- Clinical and demographic characteristics
- The demographic and clinical characteristics of the PD patients are summarized in Table 1 and Supplementary Table 1 (in the online-only Data Supplement). In Tier 1 (n = 1,202), patients in the PD-CI group were older and were predominantly male. There were no differences in education level. In the Tier 1 subset for machine learning tests (n = 416), PD-CI patients were older, had a longer duration of PD, more severe parkinsonian motor symptoms, and more likely to suffer from depression. In Tier 2, which was designed for the validation study (n = 91), the PD-CI patients were older and exhibited more severe parkinsonian motor symptoms. Across both tiers and the Tier 1 subset, total MoCA scores were significantly lower in the PD-CI group. In both the Tier 1 and Tier 2 subsets, cognitive complaints reported by caregivers were more common in the PD-CI subgroup. The frequency of failure in the pentagon copying test was greater in the order of PD-MCI to PD-related dementia.
- Age- and education-adjusted cutoffs of the MoCA
- The cutoffs for cognitive impairment across different education levels and ages, determined via logistic regression, are presented in Table 2. For patients with a median age between 53 and 68 years and more than 12 years of education, 25 points was established as the cutoff. In contrast, for patients with a median age between 69 and 83.5 years, the cutoff was set at 24 points. A lower education level generally corresponded to lower cutoff values: 22 points for 10–12 years of education (median age range: 62–83.5 years), 21 points for 7–9 years of education (median age range: 53–69 years), and 20 points for the same educational level but an older age range (70–83.5 years). For those with 4–6 years of education, the cutoff was 19 points (median age range: 53–77 years). The cutoff for a lower education level (0.5–3 years) ranged between 18 and 16 points. For patients with PD with no formal education, the cutoff was set at 16 points (median age range: 60–80 years).
- Comparison of machine learning method performances
- A comparison of machine learning analysis methods using MoCA domain scores revealed that the SVM method yielded the highest accuracy (0.7624), while the RF method exhibited the lowest (0.7404). The area under the ROC curve ranged from 0.8193 to 0.8577. However, all accuracy levels fell within one standard deviation, as detailed in Table 3. This ranking of performance across different machine learning methods was consistent when applied to various types of datasets. The inclusion of interlocking pentagon copying test scores with MoCA domain scores led to improved performance, which was still within the standard deviation range. Analyses using binary results of the 30 items in the MoCA, as opposed to domain scores, resulted in the lowest performance, yet it was also within the standard deviation range. Across all outcomes from various machine learning models and datasets, specificity consistently exceeded sensitivity. In the machine learning analysis using SVM, the factors contributing to accuracy were ranked in the following order of importance: years of education, subtotal scores of orientation, memory, visuospatial/executive function, attention, motor Unified Parkinson’s Disease Rating Scale scores, and subtotal language score, as shown in Supplementary Table 2 (in the online-only Data Supplement). Notably, cognitive complaints reported by caregivers contributed to accuracy, whereas those reported by patients did not. The contribution of the pentagon copying test to performance followed that of the subtotal test in terms of visuospatial/executive function. The website URL for the SVM-based machine learning test of the MoCA is “http://pdmoca.com.”
- Head-to-head comparison of diagnostic performance among various cutoff methods and machine learning methods
- We subsequently compared the performances of various cutoffs and the machine learning method, specifically the SVM, using a new dataset of 91 PD patients (Table 4). Among the cutoff methods, the highest accuracies were achieved when using the age- and education-adjusted cutoffs, which demonstrated high sensitivity (0.8627) and moderate specificity (0.7250). Applying a uniform cutoff of 25 or 24 for identifying cognitive impairment in PD patients resulted in reduced accuracy (0.7033 or 0.7582), with a significant decrease in specificity (0.4000 or 0.5250). Analysis using cutoffs developed for Korean vascular cognitive impairment indicated lower accuracy (0.7582) with high specificity (0.9250) but low sensitivity (0.6274), implying that these cutoffs were less stringent than the age- and education-adjusted cutoffs for PD. The machine learning analysis using SVM, based on MoCA domain scores, showed comparable accuracies but with higher specificity (0.8500) and slightly lower sensitivity (0.7843) than when the age- and education-adjusted cutoffs were used.
RESULTS
- Our findings indicate that both age- and education-adjusted cutoff values, as well as machine learning analysis using MoCA domain scores, effectively distinguish PD-CIs with notable accuracy. The age- and education-adjusted cutoffs demonstrated higher sensitivity, while machine learning analysis, specifically the SVM utilizing MoCA domain scores, showed greater specificity. To our knowledge, this study is the first where age- and education-adjusted MoCA cutoffs were developed from a PD patient cohort. Our results confirm that MoCA scores are sensitive to variations in both age and education levels. Prior research examining the validity of the MoCA for diagnosing PD-MCI or PD dementia in English-speaking populations suggested a universal cutoff score of 25 (sensitivity 0.70, specificity 0.75) or 24 (sensitivity 0.87, specificity 0.75), irrespective of age and education level [15,18]. Our findings indicate that for individuals with education levels above 12 years, the cutoff is 25 or 24 within a median age range of 53 to 83.5 years. With each 3-year decrease in education level, the cutoff score decreased by a median of 2 points (ranging from 1 to 3 points). When our age- and education-adjusted cutoffs were applied to our Tier 2 cohort, the performance of these cutoff values was comparable to that of previous studies. However, the specificity was lower when a single cutoff score was applied to the same cohort. In conclusion, within a population with varying education levels, using a single cutoff score may lead to inaccurate results.
- Several studies have reported MoCA cutoffs for screening MCI and/or dementia in non-English-speaking populations. However, systematic validation in PD groups has been limited [19,37]. Age- and education-adjusted cutoffs developed based on these populations vary significantly and are influenced by factors such as participant enrollment methods, underlying disorders related to cognitive impairment, cognitive diagnosis approaches, assessment methods, languages used, and the range of ages and education levels [11,38]. Consequently, directly comparing our cutoff scores or performance with those of these studies is impractical.
- Our study on developing cutoff scores differs from previous research in several ways. First, while most studies include participants with normal cognition, MCI, or dementia from various neurocognitive disorder etiologies, our study exclusively involved PD patients with three different cognitive diagnoses. This approach differs from that used in other studies on Korean vascular cognitive impairment patients, where the derived ageand education-adjusted cutoffs showed reduced diagnostic accuracy. Therefore, the need for disease-specific cutoff criteria, which may result in better diagnostic performance, warrants further investigation. Second, the cognitive diagnoses in our study were determined using the gold standard method, namely, comprehensive neuropsychological assessments, as recommended by the MDS Task Force [5,6]. This approach likely led to more accurate cognitive diagnoses than did those in other studies. Notably, cognitive diagnosis using machine learning tests demonstrated greater specificity than did that using age- and education-adjusted cutoffs. This could be attributed to the machine learning test’s ability to mimic our cognitive diagnoses, which are based on comprehensive assessments of multiple cognitive domains. Lastly, unlike most studies that group age into 60s or 70s, we utilized median ages ranging from ± 5 years. This was feasible due to the large number of participants in our study. Given that age is a continuous variable, grouping subjects into broad age categories may not be as accurate. Our results indicated that within the same education level, two or three different cutoff scores emerged, and the ages at which these cutoff scores changed varied across education levels.
- The accuracy of machine learning analyses using the database from the PPMI cohort in our previous study was comparable to that in the present study [21]. Although the inclusion of cognitive complaints enhanced the classification performance of machine learning in our previous study, feature importance analyses in the current study revealed that a caregiver’s report of cognitive complaints was not a highly significant factor. The accuracy of the machine learning analysis in this study is akin to that of the PPMI data, even when the variable “cognitive complaint” is excluded. This discrepancy might stem from differences in the data collection methods used, as all patients with MCI and dementia were considered to have cognitive complaints according to the PPMI operations manual.
- Visuospatial dysfunction is recognized as an early symptom of PD-MCI and is strongly associated with PD dementia, and failing the pentagon copying test is a known predictor of dementia risk in PD patients [39]. However, our results indicate that including the pentagon copying score in machine learning analysis did not enhance diagnostic performance. This outcome may be attributed to the redundancy of features between the pentagon copying test and subtotal scores of visuospatial/executive function. This inference is supported by the significantly greater frequency of failing pentagon copying test scores in the PD-CI group than in the PD-normal cognition group according to our data and by the fact that both tests are close in terms of ranking in the feature importance analysis.
- Our study has several limitations. First, the data were sourced from two hospitals that used identical neuropsychological tests to evaluate cognitive domains. The validation was performed at only one center and involved a relatively small participant group. Ideally, our cutoff values should be validated at other centers using different types of neuropsychological tests. Second, our cutoffs were based on the Korean version of the MoCA, and the impact of linguistic variations may be significant. It would be beneficial to validate whether our cutoff scores are applicable to MoCA tests in different languages among patients with PD. Third, our study merged MCI and dementia categories, although some research has suggested specific cutoffs for each [14-17]. We believe that screening for cognitive impairment by combining MCI and dementia is practical in clinical settings. This is because a cutoff solely for MCI, excluding dementia, often results in a high rate of false-positives and low specificity [9,16,17]. Additionally, some patients with PD experience early and rapid cognitive decline, progressing to dementia [40]. We did not investigate separate cutoffs for dementia due to the limited number of dementia patients in our validation cohort. Assessing ADL related to cognitive dysfunction may be more crucial in diagnosing dementia than relying solely on a MoCA cutoff score.
- In conclusion, a single MoCA cutoff score is inadequate for screening for cognitive impairment in PD patients across diverse education levels. Both age- and education-adjusted cutoff methods and machine learning, particularly the SVM approach, demonstrated high effectiveness in detecting cognitive impairment in PD patients. This underscores the potential of machine learning to enhance cognitive assessments in PD patients.
DISCUSSION
Supplementary Material
Supplementary Table 1.
Supplementary Table 2.
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Funding Statement
This research was supported by a grant of the Korea Health Technology R&D Project through the Korean Healthy Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2023-00265377). This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. NRF-2022R1F1A1063098).
-
Author contributions
Conceptualization: Yun Joong Kim. Data curation: Young Min Kim, Han Kyu Na, Dong Ho Shin, Seok Jong Chung. Formal analysis: Kyeongmin Baek, Junki Lee, Kiyong Kim, Jeehee Yoon, Seok-Jae Heo. Funding acquisition: Jeehee Yoon, Yun Joong Kim. Methodology: Jeehee Yoon, Yun Joong Kim. Project administration: Jeehee Yoon, Yun Joong Kim. Resources: Seok Jong Chung, Yun Joong Kim, Phil Hyu Lee, Young H. Sohn. Supervision: Jeehee Yoon, Yun Joong Kim. Writing—original draft: Young Min Kim, Yun Joong Kim. Writing—review & editing: Jeehee Yoon, Yun Joong Kim, Phil Hyu Lee, Young H. Sohn.
Notes
- None
Acknowledgments
MoCA data from 416 patients with PD, who underwent comprehensive neuropsychological testing, were divided in an 8:2 ratio (training:test) for machine learning analysis. Accuracy was assessed by averaging results from randomly sampled MoCA data (n = 100).
LR, linear regression; RF, random forest; SVM, support vector machine; SD, standard deviation; PPV, positive predictive value; NPV, negative predictive value; AUROC, area under the receiver operating characteristic curve.
- 1. Litvan I, Aarsland D, Adler CH, Goldman JG, Kulisevsky J, Mollenhauer B, et al. MDS task force on mild cognitive impairment in Parkinson’s disease: critical review of PD-MCI. Mov Disord 2011;26:1814–1824.ArticlePubMedPMC
- 2. Svenningsson P, Westman E, Ballard C, Aarsland D. Cognitive impairment in patients with Parkinson’s disease: diagnosis, biomarkers, and treatment. Lancet Neurol 2012;11:697–707.ArticlePubMed
- 3. Buter TC, van den Hout A, Matthews FE, Larsen JP, Brayne C, Aarsland D. Dementia and survival in Parkinson disease: a 12-year population study. Neurology 2008;70:1017–1022.ArticlePubMed
- 4. Riedel O, Klotsche J, Spottke A, Deuschl G, Förstl H, Henn F, et al. Frequency of dementia, depression, and other neuropsychiatric symptoms in 1,449 outpatients with Parkinson’s disease. J Neurol 2010;257:1073–1082.ArticlePubMedPDF
- 5. Dubois B, Burn D, Goetz C, Aarsland D, Brown RG, Broe GA, et al. Diagnostic procedures for Parkinson’s disease dementia: recommendations from the Movement Disorder Society task force. Mov Disord 2007;22:2314–2324.ArticlePubMed
- 6. Litvan I, Goldman JG, Tröster AI, Schmand BA, Weintraub D, Petersen RC, et al. Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: Movement Disorder Society task force guidelines. Mov Disord 2012;27:349–356.ArticlePubMedPMCPDF
- 7. Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 2005;53:695–699.ArticlePubMed
- 8. Skorvanek M, Goldman JG, Jahanshahi M, Marras C, Rektorova I, Schmand B, et al. Global scales for cognitive screening in Parkinson’s disease: critique and recommendations. Mov Disord 2018;33:208–218.ArticlePubMedPDF
- 9. Carson N, Leach L, Murphy KJ. A re-examination of Montreal Cognitive Assessment (MoCA) cutoff scores. Int J Geriatr Psychiatry 2018;33:379–388.ArticlePubMedPDF
- 10. Thomann AE, Berres M, Goettel N, Steiner LA, Monsch AU. Enhanced diagnostic accuracy for neurocognitive disorders: a revised cut-off approach for the Montreal Cognitive Assessment. Alzheimers Res Ther 2020;12:39.ArticlePubMedPMCPDF
- 11. Borland E, Nägga K, Nilsson PM, Minthon L, Nilsson ED, Palmqvist S. The Montreal Cognitive Assessment: normative data from a large Swedish population-based cohort. J Alzheimers Dis 2017;59:893–901.ArticlePubMedPMC
- 12. Classon E, van den Hurk W, Lyth J, Johansson MM. Montreal Cognitive Assessment: normative data for cognitively healthy Swedish 80- to 94-yearolds. J Alzheimers Dis 2022;87:1335–1344.ArticlePubMedPMC
- 13. Milani SA, Marsiske M, Cottler LB, Chen X, Striley CW. Optimal cutoffs for the Montreal Cognitive Assessment vary by race and ethnicity. Alzheimers Dement (Amst) 2018;10:773–781.ArticlePubMedPMCPDF
- 14. Dalrymple-Alford JC, MacAskill MR, Nakas CT, Livingston L, Graham C, Crucian GP, et al. The MoCA: well-suited screen for cognitive impairment in Parkinson disease. Neurology 2010;75:1717–1725.ArticlePubMed
- 15. Hoops S, Nazem S, Siderowf AD, Duda JE, Xie SX, Stern MB, et al. Validity of the MoCA and MMSE in the detection of MCI and dementia in Parkinson disease. Neurology 2009;73:1738–1745.ArticlePubMedPMC
- 16. Kandiah N, Zhang A, Cenina AR, Au WL, Nadkarni N, Tan LC. Montreal Cognitive Assessment for the screening and prediction of cognitive decline in early Parkinson’s disease. Parkinsonism Relat Disord 2014;20:1145–1148.ArticlePubMed
- 17. Marras C, Armstrong MJ, Meaney CA, Fox S, Rothberg B, Reginold W, et al. Measuring mild cognitive impairment in patients with Parkinson’s disease. Mov Disord 2013;28:626–633.ArticlePubMedPMCPDF
- 18. Damian AM, Jacobson SA, Hentz JG, Belden CM, Shill HA, Sabbagh MN, et al. The Montreal Cognitive Assessment and the mini-mental state examination as screening instruments for cognitive impairment: item analyses and threshold scores. Dement Geriatr Cogn Disord 2011;31:126–131.ArticlePubMedPDF
- 19. Ozdilek B, Kenangil G. Validation of the Turkish version of the Montreal Cognitive Assessment Scale (MoCA-TR) in patients with Parkinson’s disease. Clin Neuropsychol 2014;28:333–343.ArticlePubMed
- 20. Kim JI, Sunwoo MK, Sohn YH, Lee PH, Hong JY. The MMSE and MoCA for screening cognitive impairment in less educated patients with Parkinson’s disease. J Mov Disord 2016;9:152–159.ArticlePubMedPMCPDF
- 21. Jeon J, Kim K, Baek K, Chung SJ, Yoon J, Kim YJ. Accuracy of machine learning using the Montreal Cognitive Assessment for the diagnosis of cognitive impairment in Parkinson’s disease. J Mov Disord 2022;15:132–139.ArticlePubMedPMCPDF
- 22. Postuma RB, Berg D, Stern M, Poewe W, Olanow CW, Oertel W, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord 2015;30:1591–1601.ArticlePubMed
- 23. Ryu HJ, Yang DW. The Seoul Neuropsychological Screening Battery (SNSB) for comprehensive neuropsychological assessment. Dement Neurocogn Disord 2023;22:1–15.ArticlePubMedPMCPDF
- 24. Kang SJ, Choi SH, Lee BH, Kwon JC, Na DL, Han SH, et al. The reliability and validity of the Korean instrumental activities of daily living (K-IADL). J Korean Neurol Assoc 2002;20:8–14.
- 25. Chung SJ, Lee HS, Kim HR, Yoo HS, Lee YH, Jung JH, et al. Factor analysis-derived cognitive profile predicting early dementia conversion in PD. Neurology 2020;95:e1650–e1659.ArticlePubMed
- 26. Emre M, Aarsland D, Brown R, Burn DJ, Duyckaerts C, Mizuno Y, et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 2007;22:1689–1707.quiz 1837. ArticlePubMed
- 27. Reibnegger G, Schrabmair W. Optimum binary cut-off threshold of a diagnostic test: comparison of different methods using Monte Carlo technique. BMC Med Inform Decis Mak 2014;14:99.ArticlePubMedPMCPDF
- 28. Jung Y, Hu J. A K-fold averaging cross-validation procedure. J Nonparametr Stat 2015;27:167–179.ArticlePubMedPMC
- 29. Habibzadeh F, Habibzadeh P, Yadollahie M. On determining the most appropriate test cut-off value: the case of tests with continuous results. Biochem Med (Zagreb) 2016;26:297–307.ArticlePubMedPMC
- 30. Kang Y, Park J, Yu K, Lee B. A reliability, validity, and normative study of the Korean-Montreal Cognitive Assessment (K-MoCA) as an instrument for screening of vascular cognitive impairment (VCI). Korean J Clin Psychol 2009;28:549–562.Article
- 31. Breiman L. Random forests. Mach Learn 2001;45:5–32.
- 32. Couronné R, Probst P, Boulesteix AL. Random forest versus logistic regression: a large-scale benchmark experiment. BMC Bioinformatics 2018;19:270.PubMedPMC
- 33. He H, Garcia EA. Learning from imbalanced data. IEEE Trans Knowl Data Eng 2009;21:1263–1284.Article
- 34. Elfil M, Negida A. Sampling methods in clinical research; an educational review. Emerg (Tehran) 2017;5:e52.PubMedPMC
- 35. Menze BH, Kelm BM, Masuch R, Himmelreich U, Bachert P, Petrich W, et al. A comparison of random forest and its Gini importance with standard chemometric methods for the feature selection and classification of spectral data. BMC Bioinformatics 2009;10:213.ArticlePubMedPMCPDF
- 36. Huang S, Cai N, Pacheco PP, Narrandes S, Wang Y, Xu W. Applications of support vector machine (SVM) learning in cancer genomics. Cancer Genomics Proteomics 2018;15:41–51.PubMed
- 37. Badrkhahan SZ, Sikaroodi H, Sharifi F, Kouti L, Noroozian M. Validity and reliability of the Persian version of the Montreal Cognitive Assessment (MoCA-P) scale among subjects with Parkinson’s disease. Appl Neuropsychol Adult 2020;27:431–439.ArticlePubMed
- 38. Tan JP, Li N, Gao J, Wang LN, Zhao YM, Yu BC, et al. Optimal cutoff scores for dementia and mild cognitive impairment of the Montreal Cognitive Assessment among elderly and oldest-old Chinese population. J Alzheimers Dis 2015;43:1403–1412.ArticlePubMed
- 39. Williams-Gray CH, Evans JR, Goris A, Foltynie T, Ban M, Robbins TW, et al. The distinct cognitive syndromes of Parkinson’s disease: 5 year follow-up of the CamPaIGN cohort. Brain 2009;132(Pt 11):2958–2969.ArticlePubMed
- 40. Aarsland D, Batzu L, Halliday GM, Geurtsen GJ, Ballard C, Ray Chaudhuri K, et al. Parkinson disease-associated cognitive impairment. Nat Rev Dis Primers 2021;7:47.ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite