Articles
- Page Path
- HOME > J Mov Disord > Volume 11(1); 2018 > Article
-
Original Article
Alteration in the Local and Global Functional Connectivity of Resting State Networks in Parkinson’s Disease - Maryam Ghahremani1, Jaejun Yoo1, Sun Ju Chung2, Kwangsun Yoo1, Jong C. Ye1, Yong Jeong1,3
-
Journal of Movement Disorders 2018;11(1):13-23.
DOI: https://doi.org/10.14802/jmd.17061
Published online: January 23, 2018
1Department of Bio and Brain Engineering, Korea Advanced Institute of Science and Technology, Daejeon, Korea
2Department of Neurology, Asan Medical Center, Ulsan University College of Medicine, Seoul, Korea
3KI for Health Science and Technology, Korea Advanced Institute of Science and Technology, Daejeon, Korea
- Corresponding author: Jong C. Ye, Department of Bio and Brain Engineering, Korea Advanced Institute of Science and Technology, 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Korea / Tel: +82-42-350-4320 / Fax: +82-42-350-4310 / E-mail: jong.ye@kaist.ac.kr
- Corresponding author: Yong Jeong, Department of Bio and Brain Engineering, Korea Advanced Institute of Science and Technology, 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Korea / Tel: +82-42-350-4324 / Fax: +82-42-350-4310 / E-mail: yong@kaist.ac.kr
• Received: August 30, 2017 • Revised: November 25, 2017 • Accepted: December 11, 2017
Copyright © 2018 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Objective
- Parkinson’s disease (PD) is a neurodegenerative disorder that mainly leads to the impairment of patients’ motor function, as well as of cognition, as it progresses. This study tried to investigate the impact of PD on the resting state functional connectivity of the default mode network (DMN), as well as of the entire brain.
-
Methods
- Sixty patients with PD were included and compared to 60 matched normal control (NC) subjects. For the local connectivity analysis, the resting state fMRI data were analyzed by seed-based correlation analyses, and then a novel persistent homology analysis was implemented to examine the connectivity from a global perspective.
-
Results
- The functional connectivity of the DMN was decreased in the PD group compared to the NC, with a stronger difference in the medial prefrontal cortex. Moreover, the results of the persistent homology analysis indicated that the PD group had a more locally connected and less globally connected network compared to the NC.
-
Conclusion
- Our findings suggest that the DMN is altered in PD, and persistent homology analysis, as a useful measure of the topological characteristics of the networks from a broader perspective, was able to identify changes in the large-scale functional organization of the patients’ brain.
- Patients with PD were recruited from the Department of Neurology at Asan Medical Center in Seoul, Korea between March 2010 and June 2012. The diagnosis of PD was made based on the United Kingdom Parkinson’s Disease Society Brain Bank clinical diagnostic criteria [16], and the mean disease duration of all the PD patients was 4.0 (± 4.1) years. Every participant underwent MRI scans, clinical interviews, and neurological examinations, including the Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS). The MDS-UPDRS and fMRI data were obtained during the practically off period. Age-matched NC are also recruited from the Department of Neurology at Samsung Medical Center in Seoul, Korea. They consisted of individuals who had no history of neurological or psychiatric disorders and no cardiovascular risk factors, such as hypertension, diabetes mellitus, hyperlipidemia, and ischemic heart disease, and who exhibited normal performances on neuropsychological tests.
- To analyze the effects of PD on the resting state networks of the brain, resting state fMRI datasets of 60 PD patients and 60 matched normal subjects were included. The PD subjects were aged 62.3 years on average and included 20 males and 40 females, with an average Mini-Mental State Examination (MMSE) of 25.2. The NC data included 14 male subjects and 46 female subjects aged 63.2 years on average, with an MMSE score of 28.8 on average. The resting state fMRI data were acquired using a 3 Tesla MRI system at the Asan Medical Center and Samsung Medical Center, Seoul, Korea for the PD patients and NC, respectively, using identical system specifications and protocols. The datasets included 100 volumes of resting state fMRI data with a repetition time of 3 seconds and echo time of 35 milliseconds, along with matching structural T1-weighted images with a repetition time of 1.114 seconds and an echo time of 10 milliseconds. The data were acquired while the patients were resting quietly but in a wakeful state with their eyes closed. This study was approved by Institutional Review Boards of both medical centers, and all the subjects gave written informed consent in advance (SMC 2006-03-011-001, AMC 2010-0138).
- Data analysis
- To estimate the spatiotemporal characteristics of the resting state fMRI data, the conventional seed-based correlation analysis [17,18] was initially applied using the Conn connectivity toolbox [19]. running on MATLAB (MathWorks Inc., Natick, MA, USA). As a primary step, the fMRI data were preprocessed using the SPM8 toolbox (Department of Imaging Neuroscience, Institute of Neurology, UCL, London, UK) as reported previouly [20]. Band-pass filtering was also applied through the Conn toolbox with a frequency window of 0.008 to 0.09 Hz. The seed-based correlation analysis was then implemented on the preprocessed data using the general linear model. Consequently, the PCC (x, y, z: -6, -52, 40 mm), mPFC (x, y, z: -1, 49, -5 mm), and the medial temporal and lateral parietal lobes as the other major contributing areas of the DMN, were selected as regions of interests (ROIs) [1,17]. The resulting correlation maps were averaged across all subjects of each group. All the DMN maps were thresholded at p < 0.001, the false-discovery rate (FDR) correction was applied for multiple comparisons correction, and the results were visualized on the common MNI brain template. To identify any statistically significant differences between the PD and NC groups, a two-sample t-test was implemented for each of the group averaged PCC and mPFC functional connectivity maps across the PD and NC subjects. The highlighted regions referred to the difference in the functional connectivity of the DMN regions between the two groups with the connectivity of the PD group being subtracted from that of the NC. Furthermore, a connectome ring was obtained from the functional connectivity of the PCC and the mPFC, with 100 selected regions from all over the brain, including the DMN nodes, using the ROI-to-ROI results explorer of the Conn toolbox with a threshold value of 0.05 [20], FDR-corrected at the seed level.
- The second part of this study aimed to investigate functional connectivity changes in PD from a broader perspective using the persistent homology analysis, which, as opposed to the seed-based analysis, does not require a fixed threshold value. The persistent homology analysis is a multiscale network modeling approach that traces whole brain network changes over different threshold values. If there are p ROI selected in the brain, each region can represent a node in the network, and these nodes are a certain functional distance away from each other. This distance is defined in terms of the correlation between the nodes, such that the higher the pairwise correlation is, the shorter the distance is between them. To form a functional network, the nodes must be connected with edges, conditional on the fact that the distance between them is less than or equal to a certain threshold value ε [14]. This implies that, after the definition of network nodes, the association between pairs of nodes is estimated based on a certain threshold that will allow them to get connected if the correlation between them is at a certain level. Any subset of the set of measurements at each node can determine a so-called n-simplex, with the elements of the set representing the vertices of the simplex. Any set of such simplices is known as a simplicial family [14]. Homology groups provide information about the properties of chains that are formed from simplices. A k-chain is defined as a linear combination of k-dimensional simplices. Thus, if k is zero, there is a sum of 0-simplices, which are the nodes or vertices of the graph, and similarly with k = 1, we would have a summation of 1-simplices, or edges and so on. A single k-chain can be mathematically explained as the following:
- where αi is an integer coefficient, σik are the k-simplices and nk is the number of k-simplices in the complex and a summation of all k-chains together, forms a group. What persistent homology is trying to do is to encode networks into simplicial complexes and then examine the network topological features which are persistent over a sequence of simplicial complexes of different sizes [21,22]. This sequence reflects the changes of the network as nodes connect together. A Rips complex is also a simplicial complex whose k-simplexes correspond to (k + 1) points which are pairwise within distance ε. By changing the threshold value, a sequence of networks is formed, each corresponding to a specific threshold, with the following relationship between them: as ε increases the subsequent Rips complexes will be bigger than all the previous ones. Thus, a nested sequence of Rips complexes is formed, each being a subset of the next on. This nested sequence of Rips complexes is called Rips Filtration, corresponding to the multiscale networks. As the filtration value changes, the subsets of connected nodes as well as the number of connected components of the network change, which implies that the topological characteristic of Rips complexes will change. These changes are visualized using a barcode diagram. A barcode plots the changes in the topological features of the network over different filtration values [14,23]. The topological features are the 0th Betti number (β0) which counts the number of connected components in the network. Therefore, the barcode plots the number of connected components versus different filtration values and it is a decreasing function that illustrates the pattern of connected components merging to form a bigger component. It reflects the basic aim of persistent homology which is to measure life-time of certain topological properties of the network over a certain filtration range [24]. However, the barcode does not have any information about where the changes occur, and it does not provide any geometric information of node positions. If the information about node position (index) is also incorporated to the barcode, a dendrogram will be obtained.14,25 If the bars in barcode are rearranged and connected according to node indexing and Rips filtration, then a single linkage dendrogram (SLD) is obtained [14,25]. Barcode provides information about the global topological characteristics when the components merge together, while the SLD reflects the local network characteristics of what subnetworks are clustered together [14].
- The implementation of the persistent homology analysis was done through MATLAB. As the initial step, 90 ROIs were selected from Automated Anatomical Labeling template, excluding the cerebellum [23], and the individual voxel time series signals were extracted and averaged across each region for each subject. The correlation coefficient among them was calculated by Pearson’s correlation coefficient [20,26], and the resulting correlation matrices were then averaged across each group. Then, the persistent homology analysis was carried out by first calculating the functional distance using the correlation matrices obtained. Based on that, the minimum spanning trees [27] were estimated, giving rise to the barcodes that displayed the topological changes over the filtration values for the PD and NC groups. After obtaining this parameter, the barcodes for each group were superimposed onto one graph, and conclusions were drawn based on the observations of the shapes and slopes [28,29]. Using the obtained barcodes, dendrograms were also produced to represent all subclusters that eventually constituted one giant network. The lines were color-coded based on the distance from the giant component. For visualization purposes, the correlations between the 90 ROIs were also displayed on a 3D human glass brain at three selected filtration values representing the transitions of the barcode slope. The nodes of the network were represented as spheres and the pairwise correlations as edges, which were color-coded based on the strength of the nodal measurement and the connection.
MATERIALS & METHODS
- In the first part of this study, the functional connectivity maps of the DMN were investigated for possible network alterations caused by PD. The PCC and the mPFC, as well as the medial temporal and lateral parietal lobes, were selected as the ROI, with the PCC (Figure 1 left column) and mPFC (Figure 1 right column) as the seed regions. With the PCC as a seed, there existed a considerable difference in the functional connectivity between the two groups, with PD having a lower connectivity. Similar decreased patterns were also observed when placing the seed at the mPFC, which is an important constituent component of the DMN. The third row of Figure 1 displays the difference maps across the two groups for both the PCC seed maps and the mPFC seed maps. The highlighted regions refer to the difference in the functional connectivity of the DMN regions between the two groups, with the connectivity of the PD group being subtracted from that of the NC (Figure 1 3rd row). Thus, also from a statistical viewpoint, the functional connectivity of the DMN was lower in PD.
- Furthermore, the connectivity of the PCC and the mPFC were also evaluated with other selected regions from all over the brain using the ROI-to-ROI results explorer of the Conn toolbox, with a threshold value of 0.05 (Figure 2). The PCC connectome ring demonstrated that in the case of PD, the PCC was less connected to the rest of the brain, and certain connections were missing when compared to the NC. Some of the significant areas that were missing in the PD connectome ring included area 38 across the temporal cortex, area 46 of the frontal cortex and area 3 of the lateral parietal lobe (Figure 2A). The mPFC connectome ring also depicted a considerable number of functional connections that were missing in the PD group compared to the NC group. These regions included the inferior parietal lobe, anterior and posterior superior temporal gyrus, cingulate gyrus, visual areas 17, 18, 19, and 22, motor and somatosensory areas 3, 4, 5, and 6, visuomotor area 7, auditory areas 41 and 42, and inferior frontal gyrus area 45 (Figure 2B).
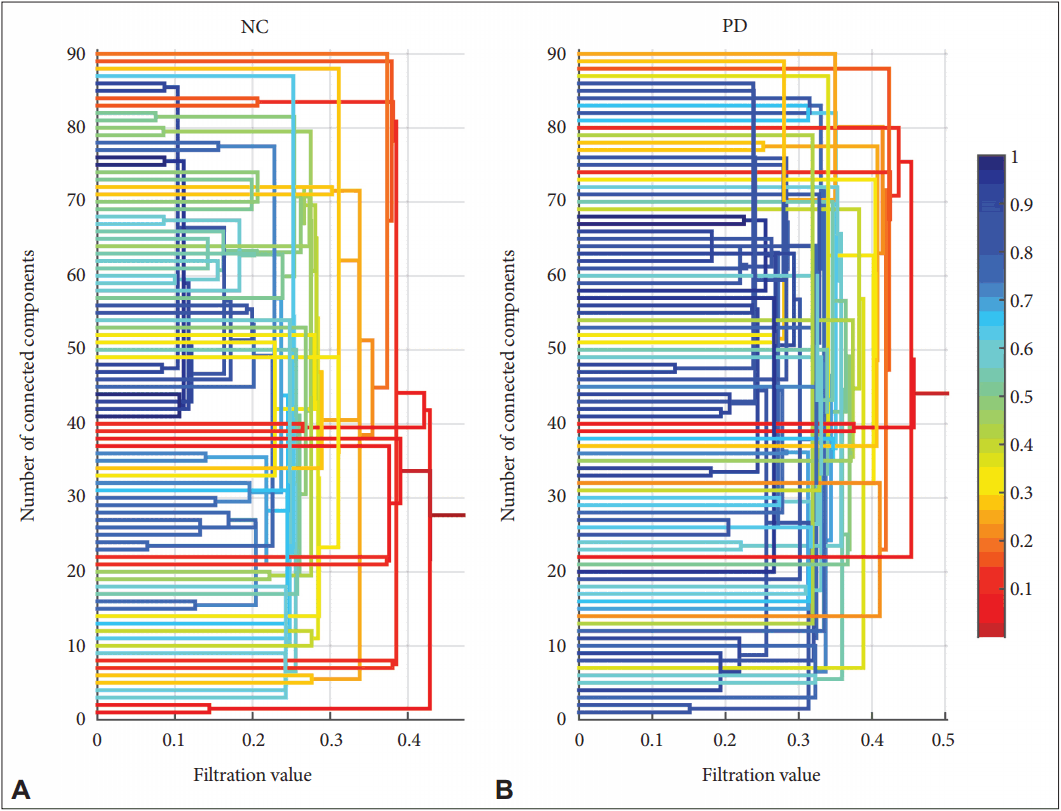
- For the second part of this study, the persistent homology analysis was performed to examine the topological characteristics of the networks from a broader perspective. Ninety ROIs were selected, and the number of connected components among these regions on a range of filtration values was placed under consideration. The resulting barcode diagrams and 3D-visualized connectivity maps are illustrated in Figure 3. This figure depicts the pattern of how smaller components connect and merge across the filtration values until they form one giant component. In this figure, the blue curve, corresponding to the PD group, demonstrates a slower slope and a longer tail at the end than the green curve, which belongs to the NC, indicating that PD formed a giant component at a later stage.
- Furthermore, a dendrogram was also obtained after including the geometrical information of where the change represented in the barcode was occurring (Figure 4). The coloring of the lines is related to the distance from the giant component, which was obtained at the end of the barcode where the whole network is globally connected. Compared to the NC (Figure 4A), it was observed that the nodal connections were colored more in dark blue, indicating a wider span of filtration values in PD (Figure 4B). Moreover, the network became one giant component at larger filtration values as displayed by the shades of red at the far right of the graph.
RESULTS
- In the first part of this study, a functional connectivity analysis was performed to evaluate the DMN connectivity in PD compared to NC. As shown in Figure 1, there was a considerable change in the connectivity of the DMN in PD. The PD group showed lower functional connectivity than the NC did among the DMN regions. Both the PCC and the mPFC, two fundamental components of the DMN, demonstrated reduced functional connectivity with DMN constituents, including their pairwise connectivity. This result was in line with previous reports [11,30]; however, it is not clear why the DMN connectivity is impaired in PD. One study observed the restoration of the DMN after levodopa administration and postulated that the dopaminergic system modulates the DMN in Parkinson’s disease [31]. Indeed, the change can be attributed to the loss of dopaminergic neurons in the substantia nigra, which is connected to the mPFC via the striatum. Thus, the impact of PD progression can indirectly cause certain functional disconnections in this circuit and may cause a possible impairment in the activity of the frontal regions of the brain [32]. Considering the fact that the mPFC is a major part of the DMN, it has been claimed that the impaired connectivity of the mPFC with the striatum may influence the integrity of the DMN and the strength of functional connections between the mPFC and the other DMN regions. Looking at it from a broader perspective, the connectivity of the PCC was further examined with 100 ROI from all over the brain, including the DMN regions, using the ROI-to-ROI explorer (Figure 2). In the NC group, the PCC seemed to be more uniformly and globally connected with other regions of the brain (Figure 2B), whereas in the PD group, the PCC was connected in a more clustered fashion, and it seemed that certain connections were missing between identical brain regions as selected in the NC analysis (Figure 2A). Across the temporal cortex, the temporopolar area, area 38 was among the areas missing its connection in PD at the selected threshold value. Studies have shown that this region is also heavily involved in other neurological disorders, such as Alzheimer’s disease, temporal lobe epilepsy, and schizophrenia [33]. Across the frontal cortex, area 46 was also disconnected in the PD group at the selected threshold value. This area roughly corresponds to the dorsolateral prefrontal cortex (DLPFC) that is thought to be involved in cognitive control of motor behavior through topdown modulations of information processing as a result of motor tasks involving, for instance, the premotor and posterior parietal associative cortices [34]. It also plays a central role in a variety of cognitive tasks, including working memory and attention [35]. Regarding resting-state networks, the DLPFC is a prominent node of both the central executive network [36,37] and the dorsal attention network [38,39], which have also been reported to be affected in PD, especially with cognitive impairments [40]. Moreover, the functional connectivity with area 3 was missing in the PD group, which might have been related to the motor deficits associated with the disease. Located in the postcentral gyrus of the lateral parietal lobe, this region corresponds to the primary somatosensory cortex and receives extensive thalamocortical projections from sensory input fields [41]. The connectome ring of the mPFC also demonstrated missing functional connections with several brain areas across the entire brain in PD (Figure 2B). Motor and somatosensory areas 3, 4, 5, and 6 were among the missing regions in the mPFC connectome ring, which could have possibly explained the motor and somatosensory deficits associated with PD. The loss of connections with the primary (17), secondary (18), and associative (19) visual areas may be related to the visual hallucinations that develop in some PD patients. These hallucinations are known to be more common at an advanced stage and are heavily associated with cognitive impairments [42,43]. These areas have been commonly included in studies examining visual hallucinations in PD patients [44,45]. One study compared PD patients with visual hallucinations and reported a consistent reduction in the gray matter volume of the occipitoparietal areas, which are related to visual functions [46]. Overall, these results may provide indications of possible damage to the functional connectivity with other brain areas, apart from the DMN constituents, that can be related to a series of symptoms that develop over the course of PD. It should be noted that these results were all observed at a fixed selected threshold value.
- We applied persistent homology to obtain a more comprehensive overview of the network connections and their topology over a range of filtration values. It is obvious that the connections, as well as their strengths, change depending on what threshold value one chooses. Persistent homology allows us to see the changes in connectivity along with the threshold changes. As shown in Figure 3, the PD group had a slower slope and reaches zero in a slower fashion than the NC did. The slope also had a longer tail at the end, meaning that it reached the state of one giant connected component at bigger filtration values than the NC. For example, if we took the number of connected components at a filtration value of 0.3, in the case of PD group, there were more than 50 connected components at this filtration value, while in the case of normal group, there were less than 20 connected components formed. This implied that, in the case of PD, the network is connected in a more clustered and local fashion than the NC. Furthermore, as shown in Figure 4, a dendrogram was also produced based on the barcode results with the additional geometrical information of node positions. The colors represent the distance from the giant component [14,25]. In the case of PD, as shown in (b), the connections of the nodes are colored in blue for larger filtration values, implying a larger number of locally connected clusters; then, the components start globally connecting and becoming a giant component at larger filtration values, as represented in red. On the other hand, the dendrogram for the NC group, as displayed in (a), shows a lesser proportion of shades of blue and a faster transition to shades of red, implying that the network is less locally connected and more globally connected; it reaches the one giant connected component at lower filtration values. These results indicated that the network in PD has local over-connectivity and global underconnectivity, which can imply a deficit in the global integrity of the large-scale networks that can affect information processing and neural communication across the entire cortex. Such a deficiency can be associated with functional deficits that arise in the course of PD, and the persistent homology was able to reveal these hidden network topologies through changing filtration values. This finding may be able to explain why deep brain stimulation of the subthalamus, which is based on a local perspective of PD pathogenesis, does not have an effect on all the signs and symptoms of the disease [47].
- Overall, by applying seed-based analysis and persistent homology, this study showed that the PD patients had more locally connected networks than the normal group did; this goes beyond the scope of a single network. This indicates that PD is not a disease that only involves limited areas or circuits of the brain. It impacts the functional organization of the entire brain, resulting in decreased connectivity of resting state networks, including the DMN. This study had certain limitations that need to be considered for future work. As indicated by the MMSE records, it appeared that the PD patients of this study had a lower score compared to the NC, indicating that some patients in this group might have developed cognitive impairment, even though the MMSE score by itself is not sufficient to rule out any cognitive impairment. Several studies have demonstrated that cognitive impairments that develop in brain disorders have a considerable impact on the functional connectivity of the DMN [48,49]. Consequently, the results of the present study may not clarify whether the network changes are from pure PD pathologies or PD with cognitive impairments. The second limitation of this study is the possibility of the effects of dopaminergic drugs on functional connectivity. Even though the UPDRS and fMRI data were all acquired during the practical off-medication period, there remains the chance that the effects may persist due to the long duration response of the dopamine replacement therapy. Further studies are needed to evaluate the longitudinal changes of the networks, in addition to changes in the networks followed by medication [50] or other treatment methods, such as deep brain stimulation [51].
DISCUSSION
- This research was supported by the Brain Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (2016 M3C7A1913844).
Acknowledgments
Figure 1.Functional connectivity of the DMN with the PCC (left) and mPFC (right) as the seed regions in the NC (first row), PD group (second row), and the difference between them (third row). PCC: posterior cingulate cortex, mPFC: medial prefrontal cortex, NC: normal control, PD: Parkinson’s disease, DMN: default mode network.
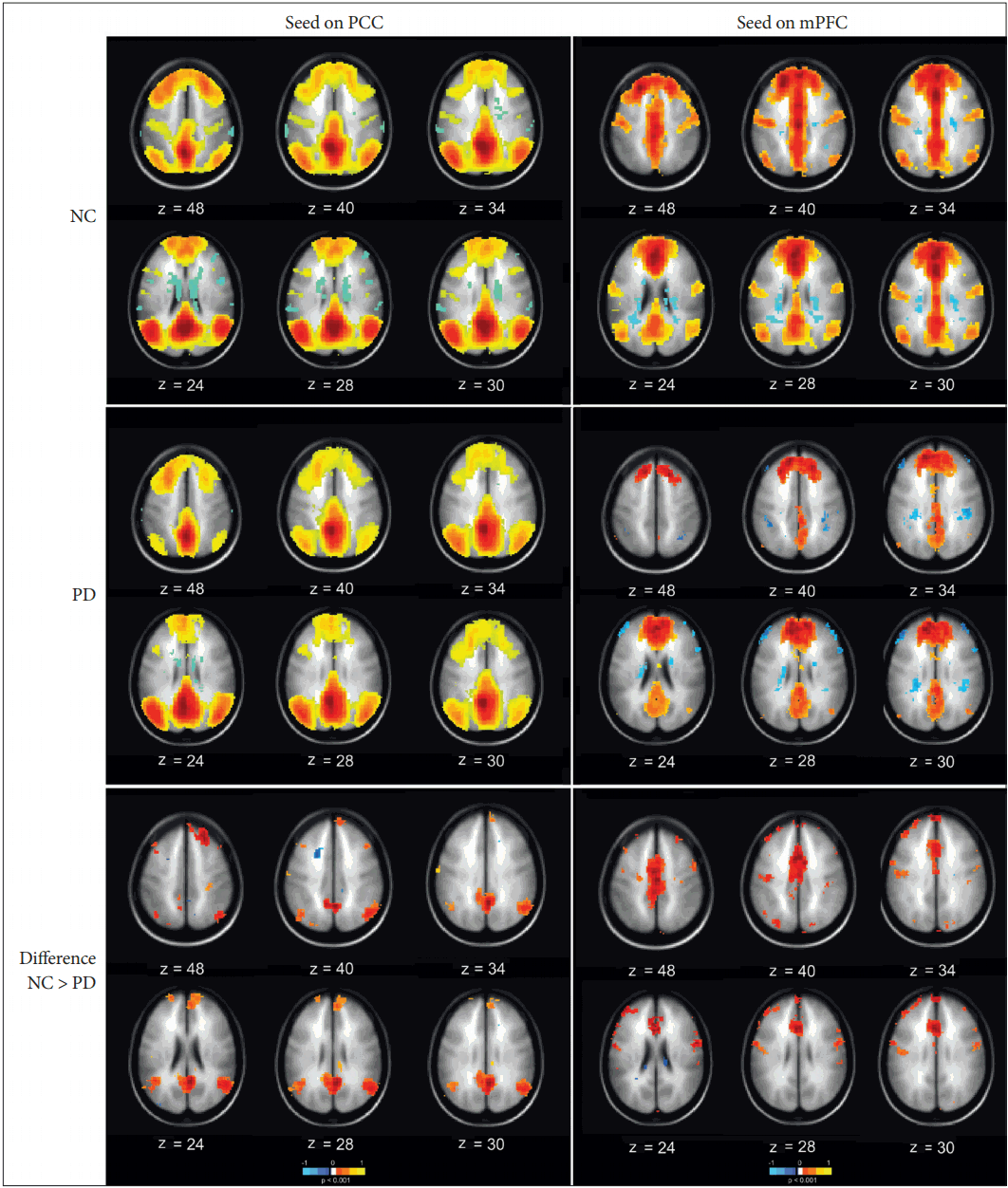

Figure 2.Connectome rings of the PCC (A and B) and mPFC (C and D) with the rest of the brain in the PD (A and C) and NC (B and D) groups. PD: Parkinson’s disease, NC: normal control, PCC: posterior cingulate cortex, mPFC: medial prefrontal cortex.


Figure 3.Barcode diagram and the brain connectivity visualization at filtrations values 0.2, 0.3, and 0.4 for the NC (green curve) and PD (blue curve) groups. The vertical axis represents the number of connected components (0th Betti number), while the horizontal axis corresponds to the filtration values. The 3D brain visualization represents the ROIs as spherical nodes and their pairwise connections as edges to represent added connection with changing filtration values; color-coded based on the connection strength. NC: normal control, PD: Parkinson’s disease, ROI: regions of interest.
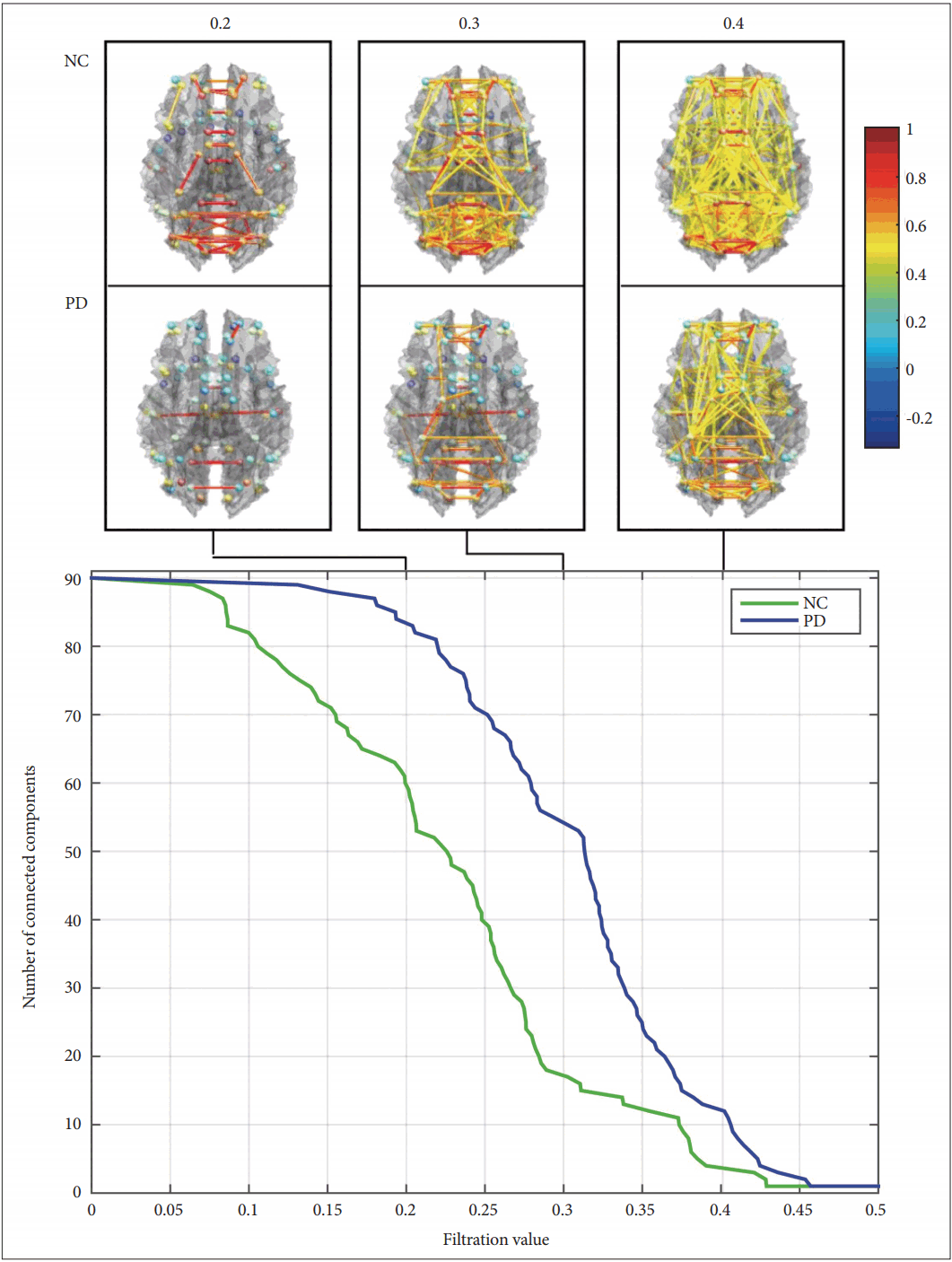

Figure 4.Dendrogram for the NC (A) and PD group (B) based on the barcode diagram. The vertical axis represents the node index over the 90 regions of interest, and the horizontal axis represents the filtration values. The coloring of the lines is based on the distance from the giant component. NC: normal control, PD: Parkinson’s disease.


- 1. Kalia LV, Lang AE. Parkinson’s disease. Lancet 2015;386:896–912.ArticlePubMed
- 2. Mantini D, Perrucci MG, Del Gratta C, Romani GL, Corbetta M. Electrophysiological signatures of resting state networks in the human brain. Proc Natl Acad Sci U S A 2007;104:13170–13175.ArticlePubMedPMC
- 3. Rosazza C, Minati L. Resting-state brain networks: literature review and clinical applications. Neurol Sci 2011;32:773–785.ArticlePubMed
- 4. Yoo K, Chung SJ, Kim HS, Choung OH, Lee YB, Kim MJ, et al. Neural substrates of motor and non-motor symptoms in Parkinson’s disease: a resting FMRI study. PLoS One 2015;10:e0125455. ArticlePubMedPMC
- 5. Biswal BB, Van Kylen J, Hyde JS. Simultaneous assessment of flow and BOLD signals in resting-state functional connectivity maps. NMR Biomed 1997;10:165–170.ArticlePubMed
- 6. De Luca M, Beckmann CF, De Stefano N, Matthews PM, Smith SM. fMRI resting state networks define distinct modes of long-distance interactions in the human brain. Neuroimage 2006;29:1359–1367.ArticlePubMed
- 7. Damoiseaux JS, Rombouts SA, Barkhof F, Scheltens P, Stam CJ, Smith SM, et al. Consistent resting-state networks across healthy subjects. Proc Natl Acad Sci U S A 2006;103:13848–13853.ArticlePubMedPMC
- 8. van den Heuvel MP, Hulshoff Pol HE. Exploring the brain network: a review on resting-state fMRI functional connectivity. Eur Neuropsychopharmacol 2010;20:519–534.ArticlePubMed
- 9. Wu T, Wang L, Chen Y, Zhao C, Li K, Chan P. Changes of functional connectivity of the motor network in the resting state in Parkinson’s disease. Neurosci Lett 2009;460:6–10.ArticlePubMed
- 10. Baudrexel S, Witte T, Seifried C, von Wegner F, Beissner F, Klein JC, et al. Resting state fMRI reveals increased subthalamic nucleus-motor cortex connectivity in Parkinson’s disease. Neuroimage 2011;55:1728–1738.ArticlePubMed
- 11. Tessitore A, Esposito F, Vitale C, Santangelo G, Amboni M, Russo A, et al. Default-mode network connectivity in cognitively unimpaired patients with Parkinson disease. Neurology 2012;79:2226–2232.ArticlePubMed
- 12. van Wijk BC, Stam CJ, Daffertshofer A. Comparing brain networks of different size and connectivity density using graph theory. PLoS One 2010;5:e13701. ArticlePubMedPMC
- 13. Bohland JW, Bokil H, Allen CB, Mitra PP. The brain atlas concordance problem: quantitative comparison of anatomical parcellations. PLoS One 2009;4:e7200. ArticlePubMedPMC
- 14. Lee H, Kang H, Chung MK, Kim BN, Lee DS. Persistent brain network homology from the perspective of dendrogram. IEEE Trans Med Imaging 2012;31:2267–2277.ArticlePubMed
- 15. Ferrarini L, Veer IM, Baerends E, van Tol MJ, Renken RJ, van der Wee NJ, et al. Hierarchical functional modularity in the resting-state human brain. Hum Brain Mapp 2009;30:2220–2231.ArticlePubMed
- 16. Hughes AJ, Daniel SE, Kilford L, Lees AJ. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinicopathological study of 100 cases. J Neurol Neurosurg Psychiatry 1992;55:181–184.ArticlePubMedPMC
- 17. Fox MD, Raichle ME. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci 2007;8:700–711.ArticlePubMedPDF
- 18. Wu JT, Wu HZ, Yan CG, Chen WX, Zhang HY, He Y, et al. Aging-related changes in the default mode network and its anti-correlated networks: a resting-state fMRI study. Neurosci Lett 2011;504:62–67.ArticlePubMed
- 19. Whitfield-Gabrieli S, Nieto-Castanon A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect 2012;2:125–141.ArticlePubMed
- 20. Friston KJ, Ashburner JT, Kiebel SJ, Nichols TE, Penny WD. Statistical Parametric Mapping: the Analysis of Functional Brain Images. 1st ed. London: Academic Press; 2006.
- 21. Hatcher A. Algebraic Topology. 1st ed. Cambridge: Cambridge University Press; 2001.
- 22. Edelsbrunner H, Morozov D. Persistent homology: theory and practice. Proc ECM 2010.Article
- 23. Edelsbrunner H, Letscher D, Zomorodian A. Topological persistence and simplification. Discrete Comput Geom 2002;28:511–533.Article
- 24. Ghrist R. Barcodes: the persistent topology of data. Bull Amer Math Soc 2008;45:61–75.ArticlePDF
- 25. Carlsson G, Memoli F. Persistent Clustering and a Theorem of J. Kleinberg. [cited 2008 Aug 16] Available from: https://arxiv.org/abs/0808.2241v1.
- 26. Rice JA. Mathematical Statistics and Data Analysis. 3rd ed. Belmont, CA: Thomson Higher Education; 2006.
- 27. Pe’er D, Wigderson A. On Minimum Spanning Trees. Jerusalem: Hebrew University; 1998.
- 28. Lee H, Chung MK, Kang H, Kim BN, Lee DS. Computing the shape of brain networks using graph filtration and Gromov-Hausdorff metric. Med Image Comput Comput Assist Interv 2011;14:302–309.ArticlePubMed
- 29. Lee H, Chung MK, Kang H, Kim BN, Lee DS. Discriminative persistent homology of brain networks. IEEE International Symposium; 2011 Mar 30-April 2; Chicago, IL, USA. 2011. 841–844.Article
- 30. Rektorova I, Krajcovicova L, Marecek R, Mikl M. Default mode network and extrastriate visual resting state network in patients with Parkinson’s disease dementia. Neurodegener Dis 2012;10:232–237.ArticlePubMed
- 31. Delaveau P, Salgado-Pineda P, Fossati P, Witjas T, Azulay JP, Blin O. Dopaminergic modulation of the default mode network in Parkinson’s disease. Eur Neuropsychopharmacol 2010;20:784–792.ArticlePubMed
- 32. Jenkins IH, Fernandez W, Playford ED, Lees AJ, Frackowiak RS, Passingham RE, et al. Impaired activation of the supplementary motor area in Parkinson’s disease is reversed when akinesia is treated with apomorphine. Ann Neurol 1992;32:749–757.ArticlePubMed
- 33. Ding SL, Van Hoesen GW, Cassell MD, Poremba A. Parcellation of human temporal polar cortex: a combined analysis of multiple cytoarchitectonic, chemoarchitectonic, and pathological markers. J Comp Neurol 2009;514:595–623.ArticlePubMedPMC
- 34. MacDonald AW 3rd, Cohen JD, Stenger VA, Carter CS. Dissociating the role of the dorsolateral prefrontal and anterior cingulate cortex in cognitive control. Science 2000;288:1835–1838.ArticlePubMed
- 35. Cieslik EC, Zilles K, Caspers S, Roski C, Kellermann TS, Jakobs O, et al. Is there “one” DLPFC in cognitive action control? Evidence for heterogeneity from co-activation-based parcellation. Cereb Cortex 2013;23:2677–2689.ArticlePubMedPDF
- 36. Heinonen J, Numminen J, Hlushchuk Y, Antell H, Taatila V, Suomala J. Default mode and executive networks areas: association with the serial order in divergent thinking. PLoS One 2016;11:e0162234. ArticlePubMedPMC
- 37. Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 2007;27:2349–2356.ArticlePubMedPMC
- 38. Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, Raichle ME. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci U S A 2005;102:9673–9678.ArticlePubMedPMC
- 39. Corbetta M, Shulman GL. Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci 2002;3:201–215.ArticlePubMedPDF
- 40. Baggio HC, Segura B, Sala-Llonch R, Marti MJ, Valldeoriola F, Compta Y, et al. Cognitive impairment and resting-state network connectivity in Parkinson’s disease. Hum Brain Mapp 2015;36:199–212.ArticlePubMed
- 41. Viaene AN, Petrof I, Sherman SM. Synaptic properties of thalamic input to layers 2/3 and 4 of primary somatosensory and auditory cortices. J Neurophysiol 2011;105:279–292.ArticlePubMed
- 42. Barnes J, David AS. Visual hallucinations in Parkinson’s disease: a review and phenomenological survey. J Neurol Neurosurg Psychiatry 2001;70:727–733.ArticlePubMedPMC
- 43. Sanchez-Ramos JR, Ortoll R, Paulson GW. Visual hallucinations associated with Parkinson disease. Arch Neurol 1996;53:1265–1268.ArticlePubMed
- 44. Sanchez-Castaneda C, Rene R, Ramirez-Ruiz B, Campdelacreu J, Gascon J, Falcon C, et al. Frontal and associative visual areas related to visual hallucinations in dementia with Lewy bodies and Parkinson’s disease with dementia. Mov Disord 2010;25:615–622.ArticlePubMed
- 45. Nagano-Saito A, Washimi Y, Arahata Y, Iwai K, Kawatsu S, Ito K, et al. Visual hallucination in Parkinson’s disease with FDG PET. Mov Disord 2004;19:801–806.ArticlePubMed
- 46. Ramírez-Ruiz B, Martí MJ, Tolosa E, Giménez M, Bargalló N, Valldeoriola F, et al. Cerebral atrophy in Parkinson’s disease patients with visual hallucinations. Eur J Neurol 2007;14:750–756.ArticlePubMed
- 47. Kim HJ, Jeon BS, Paek SH. Nonmotor symptoms and subthalamic deep brain stimulation in Parkinson’s disease. J Mov Disord 2015;8:83–91.ArticlePubMedPMC
- 48. Disbrow EA, Carmichael O, He J, Lanni KE, Dressler EM, Zhang L, et al. Resting state functional connectivity is associated with cognitive dysfunction in non-demented people with Parkinson’s disease. J Parkinsons Dis 2014;4:453–465.ArticlePubMed
- 49. Lee ES, Yoo K, Lee YB, Chung J, Lim JE, Yoon B, et al.; Alzheimer’s Disease Neuroimaging Initiative. Default mode network functional connectivity in early and late mild cognitive impairment: results from the Alzheimer’s disease neuroimaging initiative. Alzheimer Dis Assoc Disord 2016;30:289–296.ArticlePubMed
- 50. Marsden CD, Parkes JD. “On-off” effects in patients with Parkinson’s disease on chronic levodopa therapy. Lancet 1976;1:292–296.ArticlePubMed
- 51. Limousin P, Pollak P, Benazzouz A, Hoffmann D, Le Bas JF, Broussolle E, et al. Effect of parkinsonian signs and symptoms of bilateral subthalamic nucleus stimulation. Lancet 1995;345:91–95.ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Topological disruption of high‐order functional networks in cognitively preserved Parkinson's disease
Song'an Shang, Siying Zhu, Jingtao Wu, Yao Xu, Lanlan Chen, Weiqiang Dou, Xindao Yin, Yu‐Chen Chen, Dejuan Shen, Jing Ye
CNS Neuroscience & Therapeutics.2023; 29(2): 566. CrossRef - IABC: A Toolbox for Intelligent Analysis of Brain Connectivity
Yuhui Du, Yanshu Kong, Xingyu He
Neuroinformatics.2023; 21(2): 303. CrossRef - Topological data analysis in biomedicine: A review
Yara Skaf, Reinhard Laubenbacher
Journal of Biomedical Informatics.2022; 130: 104082. CrossRef - Altered Long- and Short-Range Functional Connectivity Density in Patients With Thyroid-Associated Ophthalmopathy: A Resting-State fMRI Study
Wen-Hao Jiang, Huan-Huan Chen, Wen Chen, Qian Wu, Lu Chen, Jiang Zhou, Xiao-Quan Xu, Hao Hu, Fei-Yun Wu
Frontiers in Neurology.2022;[Epub] CrossRef - Modulations of static and dynamic functional connectivity among brain networks by electroacupuncture in post-stroke aphasia
Minjie Xu, Ying Gao, Hua Zhang, Binlong Zhang, Tianli Lyu, Zhongjian Tan, Changming Li, Xiaolin Li, Xing Huang, Qiao Kong, Juan Xiao, Georg S. Kranz, Shuren Li, Jingling Chang
Frontiers in Neurology.2022;[Epub] CrossRef - Multi-dimensional persistent feature analysis identifies connectivity patterns of resting-state brain networks in Alzheimer’s disease
Jin Li, Chenyuan Bian, Haoran Luo, Dandan Chen, Luolong Cao, Hong Liang
Journal of Neural Engineering.2021; 18(1): 016012. CrossRef - Characterizing resting‐state networks in Parkinson’s disease: A multi‐aspect functional connectivity study
Mahdieh Ghasemi, Ali Foroutannia, Abbas Babajani‐Feremi
Brain and Behavior.2021;[Epub] CrossRef - The role of the medial prefrontal cortex in cognition, ageing and dementia
Dan D Jobson, Yoshiki Hase, Andrew N Clarkson, Rajesh N Kalaria
Brain Communications.2021;[Epub] CrossRef - Image Target Recognition Model of Multi- Channel Structure Convolutional Neural Network Training Automatic Encoder
Sen Zhang, Qiuyun Cheng, Dengxi Chen, Haijun Zhang
IEEE Access.2020; 8: 113090. CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite