Articles
- Page Path
- HOME > J Mov Disord > Volume 13(2); 2020 > Article
-
Original Article
The Non-Motor Symptom Profile of Progressive Supranuclear Palsy -
Sudhakar Pushpa Chaithra1*
 , Shweta Prasad1,2*
, Shweta Prasad1,2* , Vikram Venkappayya Holla1
, Vikram Venkappayya Holla1 , Albert Stezin1,2
, Albert Stezin1,2 , Nitish Kamble1
, Nitish Kamble1 , Ravi Yadav1
, Ravi Yadav1 , Pramod Kumar Pal1
, Pramod Kumar Pal1
-
Journal of Movement Disorders 2020;13(2):118-126.
DOI: https://doi.org/10.14802/jmd.19066
Published online: April 6, 2020
1Department of Neurology, National Institute of Mental Health and Neurosciences, Bangalore, India
2Department of Clinical Neurosciences, National Institute of Mental Health and Neurosciences, Bangalore, India
- Corresponding author: Pramod Kumar Pal, MBBS, MD, DNB, DM Department of Neurology, National Institute of Mental Health and Neurosciences, Hosur Road, Bangalore-560029, Karnataka, India / Tel: +91-80-26995147 / Fax: +91-80-26564830 / E-mail: palpramod@hotmail.com
- *This authors contributed equally to this work.
Copyright © 2020 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Objective
- Non-motor symptoms (NMSs) significantly contribute to increased morbidity and poor quality of life in patients with parkinsonian disorders. This study aims to explore the profile of NMSs in patients with progressive supranuclear palsy (PSP) using the validated Non-Motor Symptom Scale (NMSS).
-
Methods
- Seventy-six patients with PSP were evaluated in this study. Motor symptoms and NMSs were evaluated using the PSP Rating Scale (PSPRS), Unified Parkinson’s Disease Rating Scale-III, Montreal Cognitive Assessment, Hamilton Depression (HAM-D) and Anxiety Rating Scales, Parkinson’s Disease Sleep Scale (PDSS) and NMSS. NMS severity and prevalence were also compared between patients with PSP-Richardson syndrome (PSP-RS) and those with PSP-parkinsonism.
-
Results
- All subjects in this cohort reported at least 2 NMSs. The most prevalent NMSs in patients with PSP were in the domains of sleep/fatigue, mood/cognition, and sexual function. The least prevalent NMSs were in the domains of cardiovascular including falls, and perceptual problems/hallucinations. Significant correlations were observed between the NMSS scores and HAM-D, PDSS, PSPRS scores and PSPRS sub-scores. The severity of NMSs was unrelated to the duration of illness. Patients with PSP-RS reported a higher severity of drooling, altered smell/taste, depression and altered interest in sex and a higher prevalence of sexual dysfunction.
-
Conclusion
- NMSs are commonly observed in patients with PSP, and the domains of sleep, mood and sexual function are most commonly affected. These symptoms contribute significantly to disease morbidity, and clinicians should pay adequate attention to identifying and addressing these symptoms.
- Subject recruitment
- This was a prospective study conducted over 10 months at the National Institute of Mental Health and Neurosciences (NIMHANS), India. Consecutive patients diagnosed with probable or possible PSP, as per the Movement Disorder Society criteria for PSP [16], by a qualified movement disorder specialist were recruited from the general neurology outpatient and movement disorder clinics at NIMHANS. This study was approved by the institute’s ethics committee [NO. NIMH/DO/IEC (BS&NS DIV)/2017-18], and informed consent was obtained from all subjects.
- Assessments
- Demographic details such as age, age at onset and the duration of illness were recorded. The rating of symptom severity and the staging of PSP were performed using the PSP Rating Scale (PSPRS) [17]. The Unified Parkinson’s Disease Rating Scale III (UPDRS-III) was also applied in the off state (at least 12 hours after the last dose of levodopa, 48 hours after the last dose of a dopamine agonist) [18]. The Hamilton Anxiety Rating Scale (HAM-A) and Hamilton Depression Rating Scale (HAM-D) were administered to estimate the presence and severity of anxiety and depression, respectively. The severity of sleep disturbances was assessed using the Parkinson’s Disease Sleep Scale (PDSS), and the Montreal Cognitive Assessment Scale (MoCA) was applied to assess cognitive function. The severity and frequency of NMSs was assessed by using the validated NMSS [15]. This is a 30-item scale covering 9 domains of NMSs: cardiovascular (CVS), including falls; sleep/fatigue; mood/cognition; perceptual problems/hallucinations; attention/memory; gastrointestinal tract; urinary; sexual function; and miscellaneous. The severity (0–3), frequency (1–4) and final score (severity × frequency) of each item are evaluated separately, and the total score for a domain is obtained by the sum of individual item scores. The total NMSS score ranges from 0 to 320, and higher scores are indicative of a higher severity and frequency of NMSs. The NMS burden was estimated using burden grading cut-off scores, i.e., 0: none, 1–20: mild, 21–40: moderate, 41–70: severe, and ≥ 71: very severe [2]. Finally, caregiver burden was estimated using the Zarit Burden interview [19].
- Statistical analysis
- Descriptive statistical analysis was performed for the demographic and clinical features of the entire cohort of patients with PSP. Data were tested for normality, following which the PSP-RS and PSP-P subgroups were compared. The third subgroup comprised other subtypes and was excluded from comparison owing to the small sample size. Partial correlations with age as a covariate were performed between scores of the NMSS domains, clinical parameters, PSPRS scores, PSPRS subscores and scores of other scales. A correlation coefficient of r > 0.5 was considered significant, and linear regressions were performed on these variables to ascertain the strength of association. Statistical significance was set at p < 0.05.
MATERIALS & METHODS
- Demographic and clinical features
- A total of 76 patients with PSP, 53 men and 23 women, were recruited for this study (Table 1). The mean age of the PSP cohort was 62.04 ± 7.10 years, with a mean disease duration of 2.68 ± 2.10 years. The cohort comprised patients with PSP-RS (n = 53, 69.73%), PSP-P (n = 16, 21.05%), PSP-with cognitive or behavioral symptoms (n = 4, 5.26%), PSP with progressive gait freezing (n = 2, 2.63%) and PSP with corticobasal syndrome (n = 1, 1.31%).
- The UPDRS-III off state score was 33.08 ± 14.00, the PSPRS score was 38.97 ± 14.00, and the PSP stage was 3.13 ± 0.94. The mean MoCA score was 19.00 ± 6.60, the HAM-A score was 5.41 ± 6.30, the HAM-D score was 7.47 ± 5.90, and the PDSS score was 128.49 ± 18.10. The caregiver burden score was 21.88 ± 12.5. A comparison of the PSP-RS and PSP-P subgroups revealed a longer duration of illness in the PSP-P subgroup. No other significant differences were observed in the demographic and basic clinical scores of these two groups.
- Non-motor symptom profile
- The total NMSS score was 47.08 ± 32.40, and the NMSS burden was mild in 19.7% (n = 15), moderate in 27.63% (n = 21), severe in 35.5% (n = 27) and very severe in 17.1% (n = 13) of patients with PSP (Table 2). The mean number of NMS domains affected was 4.81 ± 1.65 (2–9 domains), and the mean number of NMSs reported was 9.61 ± 4.84 (2–24 symptoms).
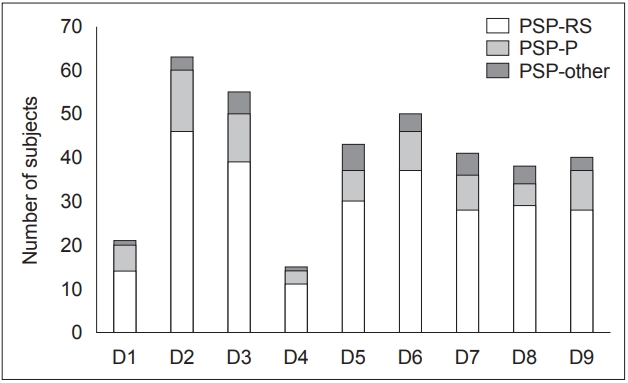
- The most prevalent domains of NMSs were sleep/fatigue (82.90%), mood/cognition (72.40%), and sexual function (70.40%) (Figure 1). Based on mean NMSS domain scores, the most affected domains of NMSs were mood/cognition, sleep/fatigue, and urinary (Table 2). Of the 76 patients with PSP, only 54 patients were sexually active. In the remaining 22 patients, the lack of sexual activity was unrelated to sexual dysfunction. Perceptual problems/hallucinations were the least prevalent NMSs (19.70%), followed by NMSs in the CVS system, including falls, domain (27.60%). The most prevalent item was “Does fatigue or lack of energy limit the patient’s daytime activities?”, which was reported by 65.80% of patients with PSP (Table 2).
- There was no difference in the total NMSS scores between the two subgroups. In the PSP-RS subgroup, the most prevalent domains of NMSs were sleep/fatigue (86.79%), sexual function (80.56%), and mood/cognition (73.58%). Perceptual problems/hallucinations were the least prevalent NMSs (20.75%), followed by NMSs in the CVS system, including falls, domain (26.42%). In the PSP-P subgroup, the most prevalent domains of NMSs were sleep/fatigue (87.50%), mood/cognition (68.75%), and gastrointestinal tract (56.25%). Similar to the PSP-RS subgroup, perceptual problems/hallucinations were the least prevalent NMSs (18.75%), followed by NMSs in the CVS system, including falls, domain (37.50%). In both subgroups, the most prevalent item was “Does fatigue or lack of energy limit the patient’s daytime activities?”, which was reported by 69.81% of patients with PSPRS and 68.75% of patients with PSP-P (Table 2). A comparison of the prevalence of NMSs between patients with PSP-RS and those with PSP-P revealed a higher prevalence of sexual dysfunction and a positive response to “Does the patient have altered interest in sex?” in patients with PSP-RS.
- The severity of NMSs was significantly different for four questions on the NMSS: “Does the patient feel sad or depressed or has he/she reported such feelings?” (PSP-RS > PSP-P), “Does the patient dribble saliva during the day?” (PSP-RS > PSP-P), “Does the patient have altered interest in sex?” (PSP-RS > PSP-P), and “Does the patient report a change in ability to taste or smell?” (PSP-RS > PSP-P). In addition, the total domain score for sexual function was higher in patients with PSP-RS.
- Several significant correlations were observed between clinical scores and the NMSS domain subscores (Table 3). High positive correlations were observed between the domain of attention/memory (D3) and the HAM-D score. The sleep/fatigue domain (D2) showed a high positive correlation with total PSP-RS score, PSP-history score, PSP-bulbar score, and caregiver burden scale score. The gastrointestinal tract domain (D6) also showed a positive correlation with the total PSP-RS score, PSP history score, PSP bulbar score, and caregiver burden scale score. Finally, the sexual function domain (D8) correlated with the caregiver burden scale score. A high negative correlation was observed between the PDSS score and the gastrointestinal tract domain (D6), the total NMSS score, the sexual function domain and the caregiver burden score (r = 0.55, p < 0.01). No correlations were observed between NMSS scores and the duration of illness.
- Linear regressions performed on the above variables revealed the persistence of associations between all variables except for sleep/fatigue and caregiver burden scale score (Table 4).
RESULTS
Complete PSP cohort
PSP-RS vs. PSP-P
Partial correlations and linear regressions
- NMSs significantly contribute to a worsening quality of life and increasing morbidity in patients with parkinsonian disorders. Although extensively explored and established in PD, the exact burden of NMSs in other parkinsonian disorders is uncertain. The present study aimed to explore the profile of NMSs in patients with PSP, to compare the NMSs between patients with PSP-Richardson syndrome and those with PSP-parkinsonism, and to identify the clinical correlates of NMSs in PSP. Additionally, we attempted to ascertain the impact of NMSs on caregiver burden. To the best of our knowledge, this is the largest study to explore the profile of NMSs in patients with PSP and to explore the differences in NMSs between PSP subtypes (Table 5) [7-11]. Although three previous studies [7,9,11] have used the NMSS in PSP, the evaluated sample sizes were relatively smaller (Radicati et al. [7]: 50, Ou et al. [9]: 27, Lee et al. [11]: 14). Other studies, which also have smaller sample sizes, either have utilized a different scale [14] or have primarily focused only on autonomic symptoms [8].
- In the current study, all patients reported the presence of at least one NMS. The most prevalent NMSs were in the domains of sleep/fatigue, mood/cognition, and sexual function (Figure 1, Table 2). The least prevalent NMSs were in the domains of perceptual problems/hallucinations, followed by the CVS system, including falls. Although no differences in the total NMSS score were observed between patients with PSP-RS and those with PSP-P, patients in the PSP-RS subgroup reported higher severity for specific questions in the domains of mood/cognition, sexual function, and miscellaneous (Figure 1). In addition, a significantly higher prevalence of sexual dysfunction was observed in patients with PSP-RS. The following paragraphs will attempt to explain the basis for the observed NMSs in patients with PSP.
- Sleep disturbances comprising daytime sleepiness, fatigue, difficulty falling or staying asleep and restless legs were the most prevalent NMSs in our cohort (Figure 1, Table 2), and “fatigue or lack of energy” was the most prevalent item in this domain (Table 2). This observation of sleep disturbances in PSP has been previously reported as the most prevalent NMS from questionnaire-based studies by Ou et al. [9] and Radicati et al. [7] and has also been reported in polysomnography (PSG)-based studies in patients with PSP [20]. Patients with PSP have been found to have disruptions in sleep architecture and in both sleep wake regulation mechanisms and circadian rhythm activity [21]. These abnormalities may be implicated in the preferential degeneration of pontine tegmental nuclei in PSP [21]. Additionally, the significant axial stiffness and difficulty turning in bed may also contribute to this NMS. This is supported by the significant correlations observed between the total PSPRS score and the sleep/fatigue domain score observed in the study, which suggests that an increase in disease severity tends to contribute to the worsening of symptoms in this domain (Table 3). In addition, we also observed correlations between the PSPRS history and bulbar subscores. These correlations may be due to the presence of a question related to sleep difficulty in the PSP-history subscore, and perhaps bulbar dysfunction may also contribute due to choking or coughing, which may occur due to difficulty swallowing saliva. These observations were supported by high p values obtained from the linear regression models (Table 3).
- NMSs in the mood/cognition domain were the second most prevalent NMSs in this cohort, and a “lack of motivation” was the most reported item in this domain (Figure 1, Table 2). This domain also had the highest score on the NMSS. Neuropsychiatric abnormalities comprising cognitive dysfunction are predominantly frontal dysexecutive syndrome, and behavioral changes such as apathy, depression and impulsivity have been frequently reported in PSP [9,11,13,22,23]. These may be attributable to the degeneration of the medial frontal regions and insular cortex, which is observed in PSP [24].
- Sexual dysfunction in patients with PSP has seldom been discussed or reported. In the present study, sexual function was the third most prevalent NMS domain, and “problems having sex” was the most commonly reported item in this domain (Figure 1, Table 3). The PSP-RS group reported a higher prevalence and severity than the PSP-P group. A reduction in libido has been previously reported in individuals with PSP [25], and previous NMSSbased studies have also reported sexual dysfunction [7,9]. This NMS in individuals with PSP may be attributable to the physical disability produced due to the disease process. Interestingly, this NMS was the only domain that significantly correlated with caregiver burden.
- Gastrointestinal tract symptoms were also reported by a large proportion of patients in this cohort (Figure 1, Table 2). As expected, “dysphagia” was the most prevalent item in this domain (Table 2). Although frequently reported and a recognized symptom of PSP, it is inadequately explored. This is crucial since dysphagia plays a significant role in the development of aspiration pneumonia, which is a predominant cause of death in patients with PSP. In the present study, we observed significant correlations between the total PSPRS score and the domain score for gastrointestinal tract symptoms, suggesting that the severity of this NMS increases as the disease progresses. In addition to the overall PSPRS score, significant correlations were observed between the history and bulbar subscores. These results, especially the latter, suggest a direct association between NMSS scores and symptoms in patients with PSP. In addition, the high p values obtained from the linear regression models also support these findings (Table 3).
- Attention/memory deficits in PSP have been frequently reported and were also observed in our cohort (Figure 1, Table 2). Patients reported maximal difficulty in “sustaining conversations during activities” (Table 2). Patients with PSP are known to have impaired attention and recall and an impairment in social functioning [13]. We observed a strong correlation between HAM-D scores and the attention/memory domain score. Memory disturbances are known to occur in depression, and it is possible that the mild depression observed in these patients contributes to the observed NMSs.
- Urinary symptoms have rarely been studied in PSP, and few studies report the presence of this or clarify the etiology [26]. In the present study, “urinary urgency” was the most prevalent item in this domain (Figure 1, Table 2). In a previous study by Radicati et al. [7], the domain of urinary symptoms was the most prevalent NMS, and nocturia was the most frequent item. It is uncertain why this result varies between the present study and the abovementioned study. Although the cause of urinary dysfunction in patients with PSP is uncertain, detrusor sphincter dyssynergia, detrusor hyperreflexia and forebrain dysfunction may be implicated [27]. Urinary symptoms are commonly associated with multiple system atrophy, and the observation of urinary symptoms in patients with PSP was higher than anticipated. Patients with atypical parkinsonian features who complain of urinary symptoms early in the course of illness should also be evaluated for the possibility of PSP.
- Patients with PSP also reported abnormalities in the domains of miscellaneous NMSs and CVS including falls, and perceptual problems/hallucinations (Figure 1, Table 2). Pain in individuals with PSP may be secondary to altered self-estimation of pain due to frontal cortical degeneration or associated with the degeneration of descending inhibitory control systems within the brainstem [28]. Recent changes in weight may be associated with dysphagia. Unlike in patients with PD, we did not observe a significant prevalence of hyposmia in patients with PSP [6]. Interestingly, a lower prevalence of abnormality in smell/taste was reported in the PSP-P subgroup than in the PSP-RS subgroup.
- The domain of CVS including falls, was found to be the second least prevalent domain. Questions in this domain should be administered with care to aid in the differentiation of falls secondary to motor disturbance and falls due to autonomic dysfunction. Dizziness was the most prevalent item in the domain of CVS including falls (Table 3). Sympathetic and parasympathetic cardiac autonomic dysfunction has been reported in patients with PSP, and this may be implicated in the observed falls [8]. Finally, the least prevalent NMSs in patients with PSP were in the perceptual problems/hallucinations domain, wherein visual hallucinations were the most frequently reported (Table 1). Hallucinations are commonly reported in patients with PD and DLB and are less frequently reported in patients with PSP [29]. A mechanism similar to that of PD, i.e., denervation supersensitivity of mesolimbic and mesocortical dopaminergic receptors and other neurotransmitter imbalances, has been implicated for hallucinations in patients with PSP [29].
- There are several limitations to this study. The NMSS is a scale primarily designed to assess NMSs in patients with PD and has not been validated in patients with PSP. However, in the absence of other validated scales for parkinsonian syndromes, we chose to utilize this scale to assess NMSs in patients with PSP. Furthermore, this scale was also recently used by Radicati et al. [7] in a study on NMSs in patients with PD and PSP. The present study was not a case-control study, and we did not compare the PSP group against healthy controls or individuals with other parkinsonian disorders. We did this with the objective of determining the impact and burden of NMSs in patients with PSP and their caregivers. Despite the use of consecutive sampling to collect data, there was a significant difference in gender in the present cohort, which may be a confounding factor.
- In conclusion, all patients with progressive supranuclear palsy reported a minimum of two NMSs, with the domains of sleep, mood and sexual function being the most commonly affected. The severity of these NMSs was unrelated to the duration of illness. NMSs contribute significantly to disease morbidity, and adequate attention should be paid to the identification and treatment of these symptoms.
DISCUSSION
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Author Contributions
Conceptualisation: all authors. Data curation: Sudhakar Pushpa Chaithra, Shweta Prasad, Vikram Venkappayya Holla, and Albert Stezin. Formal analysis: Sudhakar Pushpa Chaithra, Shweta Prasad, Vikram Venkappayya Holla, and Albert Stezin. Supervision: Nitish Kamble, Ravi Yadav, and Pramod Kumar Pal. Writing—original draft: Sudhakar Pushpa Chaithra, Shweta Prasad, Vikram Venkappayya Holla, and Albert Stezin. Writing—review & editing: Nitish Kamble, Ravi Yadav, and Pramod Kumar Pal.
Notes
- None.
Acknowledgments
| PSP (n = 76) | PSP-RS (n = 53) | PSP-P (n = 16) | p value (PSP-RS vs. PSP-P) | |
|---|---|---|---|---|
| Age (years) | 62.04 ± 7.10 | 62.19 ± 7.86 | 61.19 ± 5.07 | 0.63 |
| Gender (M:W) | 53:23 | 35:18 | 13:03 | 0.35 |
| Age of onset (years) | 59.55 ± 7.10 | 60.17 ± 7.65 | 56.81 ± 5.52 | 0.10 |
| Duration of illness (years) | 2.68 ± 2.10 | 2.19 ± 1.44 | 4.28 ± 3.25 | 0.02* |
| UPDRS-III off | 33.08 ± 14.00 | 34.08 ± 13.76 | 33.09 ± 12.68 | 0.79 |
| PSPRS score | 38.97 ± 14.00 | 40.32 ± 13.94 | 37.00 ± 13.66 | 0.40 |
| PSPRS-history | 7.26 ± 3.82 | 7.47 ± 3.89 | 6.81 ± 3.86 | 0.55 |
| PSPRS-mentation | 2.50 ± 2.68 | 2.57 ± 2.53 | 1.81 ± 2.34 | 0.29 |
| PSPRS-bulbar | 2.52 ± 1.69 | 2.79 ± 1.62 | 2.00 ± 1.54 | 0.08 |
| PSPRS-ocular | 9.93 ± 2.99 | 10.08 ± 3.07 | 9.63 ± 2.60 | 0.59 |
| PSPRS-limb | 5.07 ± 2.51 | 5.09 ± 2.48 | 5.69 ± 2.41 | 0.40 |
| PSPRS-gait | 11.40 ± 4.56 | 12.11 ± 4.18 | 10.31 ± 5.19 | 0.15 |
| PSP staging | 3.13 ± 0.94 | 3.24 ± 0.91 | 2.81 ± 0.98 | 0.10 |
| MoCA | 19.00 ± 6.60 | 19.05 ± 5.81 | 20.50 ± 6.04 | 0.41 |
| HAM-A | 5.41 ± 6.30 | 4.96 ± 6.23 | 6.19 ± 5.86 | 0.48 |
| HAM D | 7.47 ± 5.90 | 7.04 ± 5.15 | 6.81 ± 5.76 | 0.88 |
| PDSS | 128.49 ± 18.10 | 130.36 ± 17.31 | 124.75 ± 17.59 | 0.26 |
| NMSS | 47.08 ± 32.4 | 48.89 ± 35.88 | 37.31 ± 22.29 | 0.22 |
| Caregiver burden | 21.88 ± 12.5 | 21.26 ± 13.02 | 22.89 ± 9.51 | 0.73 |
* significant difference between patients with PSP-RS and those with PSP-P.
HAM-A: Hamilton Anxiety Rating Scale, HAM-D: Hamilton Depression Rating Scale, M: men, MoCA: Montreal Cognitive Assessment, NMSS: Non-Motor Symptom Scale, PDSS: Parkinson’s Disease Sleep Scale, PSP: progressive supranuclear palsy, PSPRS: Progressive Supranuclear Palsy Rating Scale, PSP-P: PSP-parkinsonism, PSP-RS: PSP-Richardson syndrome, UPDRS: Unified Parkinson’s Disease Rating Scale, W: women.
| Domain |
PSP (n = 76) |
PSP-RS (n = 53) |
PSP-P (n = 16) |
PSP-RS vs. PSP-P |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Prevalence (%) | NMSS score | Prevalence (%) | NMSS score | Prevalence (%) | NMSS score | Prevalence p value | NMSS score p value | |||
| D1 | Cardiovascular including falls | 27.60 | 1.40 ± 3.37 | 26.42 | 1.55 ± 3.81 | 37.50 | 1.31 ± 2.27 | 0.53 | 0.81 | |
| 1 | Does the patient experience light-headedness, dizziness, weakness on standing from sitting or lying position? | 27.60 | 1.13 ± 2.27 | 26.42 | 1.15 ± 2.37 | 37.50 | 1.31 ± 2.27 | 0.53 | 0.81 | |
| 2 | Does the patient fall because of fainting or blacking out? | 5.30 | 0.27 ± 1.49 | 7.55 | 0.40 ± 1.78 | 0.00 | 0 ± 0 | 0.53 | 0.37 | |
| D2 | Sleep/fatigue | 82.90 | 7.84 ± 6.44 | 86.79 | 8.23 ± 6.54 | 87.50 | 8.19 ± 5.82 | 1.00 | 0.98 | |
| 3 | Does the patient doze off or fall asleep unintentionally during daytime activities? | 36.80 | 1.52 ± 2.46 | 41.51 | 1.66 ± 2.31 | 31.25 | 1.63 ± 3.28 | 0.56 | 0.96 | |
| 4 | Does fatigue or lack of energy limit the patients daytime activities? | 65.80 | 3.71 ± 3.78 | 69.81 | 4.28 ± 3.91 | 68.75 | 3.25 ± 3.39 | 1.00 | 0.34 | |
| 5 | Does the patient have difficulties falling or staying asleep? | 38.20 | 1.90 ± 3.33 | 35.85 | 1.62 ± 2.91 | 50.00 | 2.44 ± 3.28 | 0.38 | 0.34 | |
| 6 | Does the patient experience an urge to move the legs or restlessness in legs that improves with movement when he/she is sitting or lying down inactive? | 14.50 | 0.56 ± 1.83 | 15.09 | 0.55 ± 1.84 | 18.75 | 0.88 ± 2.18 | 0.70 | 0.55 | |
| D3 | Mood/cognition | 72.40 | 10.43 ± 11.75 | 73.58 | 10.43 ± 12.02 | 68.75 | 8.44 ± 11.03 | 0.75 | 0.55 | |
| 7 | Has the patient lost interest in his/her surroundings? | 53.90 | 2.71 ± 3.49 | 54.72 | 2.66 ± 3.43 | 43.75 | 2.25 ± 3.41 | 0.57 | 0.67 | |
| 8 | Has the patient lost interest in doing things or lack motivation to start new activities? | 55.30 | 2.74 ± 3.51 | 56.60 | 2.70 ± 3.47 | 43.75 | 2.06 ± 3.31 | 0.40 | 0.51 | |
| 9 | Does the patient feel nervous, worried or frightened for no apparent reason? | 28.90 | 0.97 ± 2.13 | 28.30 | 0.91 ± 2.23 | 31.25 | 1.00 ± 1.63 | 1.00 | 0.87 | |
| 10 | Does the patient feel sad or depressed or has he/she reported such feelings? | 42.10 | 1.37 ± 2.26 | 45.28 | 1.51 ± 2.45 | 31.25 | 0.63 ± 1.02 | 0.39 | 0.04* | |
| 11 | Does the patient have flat moods without the normal “highs” and “lows”? | 28.90 | 1.33 ± 2.70 | 30.19 | 1.40 ± 2.81 | 31.25 | 1.44 ± 2.78 | 1.00 | 0.95 | |
| 12 | Does the patient have difficulty in experiencing pleasure from their usual activities or report that they lack pleasure? | 32.90 | 1.37 ± 2.62 | 33.96 | 1.34 ± 2.51 | 18.75 | 1.06 ± 3.08 | 0.35 | 0.71 | |
| D4 | Perceptual problem/hallucination | 19.70 | 1.45 ± 4.42 | 20.75 | 1.64 ± 4.93 | 18.75 | 0.69 ± 2.02 | 1.00 | 0.45 | |
| 13 | Does the patient indicate that he/she sees things that are not there? | 10.50 | 0.57 ± 2.27 | 9.43 | 0.58 ± 2.54 | 12.50 | 0.38 ± 1.08 | 0.65 | 0.75 | |
| 14 | Does the patient have beliefs that you know are not true? | 5.30 | 0.41 ± 1.88 | 3.77 | 0.40 ± 2.04 | 6.25 | 0.25 ± 1.00 | 0.55 | 0.78 | |
| 15 | Does the patient experience double vision? | 9.20 | 0.47 ± 1.89 | 11.32 | 0.66 ± 2.24 | 6.25 | 0.06 ± 0.25 | 1.00 | 0.06 | |
| D5 | Attention/memory | 56.60 | 4.91 ± 7.08 | 56.60 | 4.60 ± 6.64 | 43.75 | 3.69 ± 7.67 | 0.16 | 0.64 | |
| 16 | Does the patient have problems sustaining conversation during activities? | 47.40 | 2.42 ± 3.31 | 49.06 | 2.47 ± 3.29 | 37.50 | 1.56 ± 2.55 | 0.56 | 0.31 | |
| 17 | Does the patient forget things that he/she has been told a short time ago or events that happened in the last few days? | 30.30 | 1.38 ± 2.84 | 28.30 | 1.11 ± 2.41 | 12.50 | 1.00 ± 3.09 | 0.32 | 0.87 | |
| 18 | Does the patient forget to do things? | 27.60 | 1.11 ± 2.46 | 26.42 | 1.02 ± 2.30 | 12.50 | 1.13 ± 3.26 | 0.32 | 0.88 | |
| D6 | Gastrointestinal tract | 65.80 | 6.41 ± 7.60 | 69.81 | 7.36 ± 8.34 | 56.25 | 4.31 ± 5.36 | 0.37 | 0.17 | |
| 19 | Does the patient dribble saliva during the day? | 36.80 | 1.67 ± 2.75 | 41.51 | 1.92 ± 2.98 | 18.75 | 0.69 ± 1.74 | 0.13 | 0.04* | |
| 20 | Does the patient have difficulty swallowing? | 48.70 | 2.86 ± 3.81 | 50.94 | 3.15 ± 4.05 | 43.75 | 2.50 ± 3.54 | 0.77 | 0.56 | |
| 21 | Does the patient suffer from constipation? | 34.20 | 1.88 ± 3.31 | 39.62 | 2.28 ± 3.64 | 18.75 | 1.13 ± 2.52 | 0.14 | 0.24 | |
| D7 | Urinary | 53.90 | 7.34 ± 9.45 | 52.83 | 7.38 ± 9.66 | 50.00 | 5.50 ± 8.18 | 1.00 | 0.48 | |
| 22 | Does the patient have difficulty holding urine? | 44.70 | 2.28 ± 3.18 | 45.28 | 2.43 ± 3.48 | 37.50 | 1.50 ± 2.12 | 0.77 | 0.31 | |
| 23 | Does the patient have to void within 2 hours of last voiding? | 34.20 | 2.34 ± 3.91 | 33.96 | 2.32 ± 3.93 | 25.00 | 1.75 ± 3.49 | 0.55 | 0.60 | |
| 24 | Does the patient have to get up regularly at night to pass urine? | 42.10 | 2.72 ± 3.87 | 41.51 | 2.62 ± 3.75 | 31.25 | 2.25 ± 3.78 | 0.56 | 0.72 | |
| D8 | Sexual function† | 70.40 | 4.20 ± 4.94 | 80.56 | 5.11 ± 5.34 | 41.66 | 1.67 ± 3.42 | 0.02* | 0.04* | |
| 25 | Does the patient have altered interest in sex? | 53.70 | 1.48 ± 2.02 | 63.89 | 1.86 ± 2.20 | 25.00 | 0.50 ± 1.16 | 0.04* | 0.01* | |
| 26 | Does the patient have problems having sex? | 61.70 | 3.13 ± 3.84 | 58.33 | 3.77 ± 4.15 | 41.66 | 1.27 ± 2.37 | 0.34 | 0.06 | |
| D9 | Miscellaneous | 52.60 | 3.42 ± 5.77 | 52.83 | 3.96 ± 6.57 | 56.25 | 2.19 ± 2.73 | 0.78 | 0.29 | |
| 27 | Does the patient suffer from pain not explained by other known conditions? | 30.30 | 1.43 ± 2.64 | 30.19 | 1.43 ± 2.66 | 25.00 | 1.38 ± 2.80 | 0.76 | 0.93 | |
| 28 | Does the patient report a change in ability to taste or smell? | 11.80 | 0.72 ± 2.37 | 16.98 | 1.04 ± 2.78 | 0.00 | 0.00 ± 0.00 | 0.10 | 0.01* | |
| 29 | Does the patient report a recent change in weight? | 26.30 | 0.63 ± 1.24 | 30.19 | 0.75 ± 1.34 | 25.00 | 0.50 ± 1.09 | 0.76 | 0.49 | |
| 30 | Does the patient experience excessive sweating? | 15.80 | 0.63 ± 1.95 | 16.98 | 0.74 ± 2.21 | 12.50 | 0.31 ± 1.01 | 1.00 | 0.46 | |
| D1 | D2 | D3 | D4 | D5 | D6 | D7 | D8 | D9 | NMSS total | |
|---|---|---|---|---|---|---|---|---|---|---|
| AAO | 0.04 | -0.30 | -0.08 | 0.19 | 0.00 | -0.12 | 0.04 | 0.00 | 0.06 | -0.08 |
| Duration | -0.08 | 0.30 | 0.19 | -0.14 | 0.14 | 0.06 | -0.15 | 0.01 | -0.08 | 0.08 |
| UPDRS off | -0.25 | 0.38 | 0.04 | -0.31 | -0.15 | 0.31 | -0.18 | 0.25 | -0.06 | 0.07 |
| MoCA | 0.00 | 0.06 | -0.34 | -0.28 | -0.29 | -0.09 | 0.41 | -0.16 | -0.20 | -0.10 |
| HAM-A | 0.31 | 0.26 | 0.19 | 0.00 | -0.02 | 0.36 | 0.48 | -0.12 | 0.14 | 0.35 |
| HAM-D | 0.36 | 0.24 | 0.41 | 0.22 | 0.54* | 0.27 | 0.28 | -0.04 | 0.24 | 0.45 |
| PDSS | -0.22 | -0.46 | -0.48 | -0.09 | -0.19 | -0.56* | -0.25 | -0.39 | -0.28 | -0.58* |
| Total PSPRS | -0.06 | 0.54* | 0.29 | -0.14 | 0.02 | 0.52* | -0.20 | 0.40 | 0.10 | 0.33 |
| PSPRS-history | -0.05 | 0.59* | 0.32 | -0.20 | 0.05 | 0.53* | -0.04 | 0.25 | 0.10 | 0.38 |
| PSPRS-mentation | 0.09 | 0.07 | 0.35 | 0.20 | 0.35 | 0.12 | -0.32 | -0.02 | 0.07 | 0.14 |
| PSPRS-bulbar | -0.12 | 0.55* | 0.26 | -0.08 | -0.11 | 0.68* | 0.12 | 0.23 | 0.22 | 0.43 |
| PSPRS-ocular | -0.02 | 0.28 | 0.37 | 0.15 | 0.17 | 0.37 | -0.10 | 0.37 | 0.12 | 0.33 |
| PSPRS-limb | -0.03 | 0.32 | 0.13 | -0.13 | -0.04 | 0.16 | -0.05 | 0.40 | -0.11 | 0.15 |
| PSPRS-gait | -0.04 | 0.35 | -0.02 | -0.21 | -0.13 | 0.30 | -0.32 | 0.32 | 0.07 | 0.07 |
| PSP staging | -0.11 | 0.18 | 0.00 | -0.23 | -0.01 | 0.18 | -0.25 | 0.28 | -0.01 | 0.02 |
| Caregiver burden scale | 0.19 | 0.52* | 0.32 | 0.00 | 0.22 | 0.19 | 0.14 | 0.60* | 0.00 | 0.41 |
D1: cardiovascular, including falls, D2: sleep/fatigue, D3: mood/cognition, D4: perceptual problems/hallucinations, D5: attention/memory, D6: gastrointestinal tract, D7: urinary, D8: sexual function, D9: miscellaneous.
* correlation coefficient greater than 0.5.
AAO: age at onset, HAM-A: Hamilton Anxiety Rating Scale, HAM-D: Hamilton Depression Rating Scale, MoCA: Montreal Cognitive Assessment, PDSS: Parkinson’s Disease Sleep Scale, PSP: progressive supranuclear palsy, PSPRS: Progressive Supranuclear Palsy Rating Scale, UPDRS: Unified Parkinson’s Disease Rating Scale.
For each regression model, the total domain score was the dependent variable, and each clinical score was the independent variable. No covariates were used in the regression analysis. GIT: gastrointestinal tract, HAM-D: Hamilton Depression Rating Scale, NMSS: Non-Motor Symptom Scale, PDSS: Parkinson’s Disease Sleep Scale, PSPRS: Progressive Supranuclear Palsy Rating Scale.
| Study | Sample size | Method of estimation | Main NMSs domains (prevalence) |
|---|---|---|---|
| Schmidt et al. [8] | PSP: 32 | Structured questionnaire | Urogenital dysfunction (93%) |
| PD: 26 | For autonomic dysfunction | Gastrointestinal symptoms (89%) | |
| HC: 27 | |||
| Colosimo et al. [14] (PRIAMO study) | PSP: 30 | Structured questionnaire | Gastrointestinal symptoms (80%) |
| MSA: 34 | Fatigue (80%) | ||
| DLB: 14 | |||
| CBD: 11 | |||
| VP: 83 | |||
| Lee et al. [11] | PSP: 14 | NMSS | Attention/memory* |
| MSA-P: 31 | Mood/cognition | ||
| MSA-C: 12 | |||
| Ou et al. [9] | PSP: 27 | NMSS | Sleep/fatigue (100%) |
| PD: 27 | Mood/apathy (100%) | ||
| HC: 27 | |||
| Radicati et al. [7] | PSP: 50 | NMSS | Sleep/fatigue (92%) |
| PD: 100 | Urinary dysfunction (92%) |
The results were represented as the percentage of the total NMSS score.
* prevalence rates unavailable.
CBD: corticobasal degeneration, DLB: dementia with Lewy bodies, HC: healthy controls, MSA: multiple system atrophy, MSA-C: MSA with predominant cerebellar ataxia, MSA-P: MSA with predominant parkinsonism, NMSs: non-motor symptoms, NMSS: Non-Motor Symptom Scale, PRIAMO: Parkinson and non-motor symptoms, PSP: progressive supranuclear palsy, VP: vascular parkinsonism.
- 1. Chaudhuri KR, Healy DG, Schapira AH. Non-motor symptoms of Parkinson’s disease: diagnosis and management. Lancet Neurol 2006;5:235–245.ArticlePubMed
- 2. Martinez-Martin P, Rodriguez-Blazquez C, Kurtis MM, Chaudhuri KR. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord 2011;26:399–406.ArticlePubMed
- 3. Chaudhuri KR, Rojo JM, Schapira AHV, Brooks DJ, Stocchi F, Odin P, et al. A proposal for a comprehensive grading of Parkinson’s disease severity combining motor and non-motor assessments: meeting an unmet need. PLoS One 2013;8:e57221. ArticlePubMedPMC
- 4. Muzerengi S, Contrafatto D, Chaudhuri KR. Non-motor symptoms: identification and management. Parkinsonism Relat Disord 2007;13 Suppl 3:S450–S456.ArticlePubMed
- 5. Zis P, Martinez-Martin P, Sauerbier A, Rizos A, Sharma JC, Worth PF, et al. Non-motor symptoms burden in treated and untreated early Parkinson’s disease patients: argument for non-motor subtypes. Eur J Neurol 2015;22:1145–1150.ArticlePubMed
- 6. Poewe W. Non-motor symptoms in Parkinson’s disease. Eur J Neurol 2008;15 Suppl 1:14–20.ArticlePubMed
- 7. Radicati FG, Martin PM, Fossati C, Chaudhuri KR, Torti M, Blazquez CR, et al. Non motor symptoms in progressive supranuclear palsy: prevalence and severity. NPJ Parkinsons Dis 2017;3:35.ArticlePubMedPMCPDF
- 8. Schmidt C, Herting B, Prieur S, Junghanns S, Schweitzer K, Reichmann H, et al. Autonomic dysfunction in patients with progressive supranuclear palsy. Mov Disord 2008;23:2083–2089.ArticlePubMed
- 9. Ou R, Song W, Wei Q, Chen K, Cao B, Hou Y, et al. Characteristics of nonmotor symptoms in progressive supranuclear palsy. Parkinsons Dis 2016;2016:9730319.ArticlePubMedPMCPDF
- 10. Barone P, Antonini A, Colosimo C, Marconi R, Morgante L, Avarello TP, et al. The PRIAMO study: a multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov Disord 2009;24:1641–1649.ArticlePubMed
- 11. Lee C-N, Kim M, Lee HM, Jang J-W, Lee S-M, Kwon D-Y, et al. The interrelationship between non-motor symptoms in atypical parkinsonism. J Neurol Sci 2013;327:15–21.ArticlePubMed
- 12. Litvan I, Agid Y, Calne D, Campbell G, Dubois B, Duvoisin RC, et al. Clinical research criteria for the diagnosis of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome): report of the NINDS-SPSP international workshop. Neurology 1996;47:1–9.ArticlePubMed
- 13. Gerstenecker A, Duff K, Mast B, Litvan I; ENGENE-PSP Study Group. Behavioral abnormalities in progressive supranuclear palsy. Psychiatry Res 2013;210:1205–1210.ArticlePubMed
- 14. Colosimo C, Morgante L, Antonini A, Barone P, Avarello TP, Bottacchi E, et al. Non-motor symptoms in atypical and secondary parkinsonism: the PRIAMO study. J Neurol 2010;257:5–14.ArticlePubMedPDF
- 15. Chaudhuri KR, Martinez-Martin P, Brown RG, Sethi K, Stocchi F, Odin P, et al. The metric properties of a novel non-motor symptoms scale for Parkinson’s disease: results from an international pilot study. Mov Disord 2007;22:1901–1911.ArticlePubMed
- 16. Hoglinger GU, Respondek G, Stamelou M, Kurz C, Josephs KA, Lang AE, et al. Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord 2017;32:853–864.ArticlePubMedPMC
- 17. Golbe LI, Ohman-Strickland PA. A clinical rating scale for progressive supranuclear palsy. Brain 2007;130:1552–1565.ArticlePubMedPDF
- 18. Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. Mov Disord 2003;18:738–750.ArticlePubMed
- 19. Zarit SH, Reever KE, Bach-Peterson J. Relatives of the impaired elderly: correlates of feelings of burden. Gerontologist 1980;20:649–655.ArticlePubMedPDF
- 20. Gross RA, Spehlmann R, Daniels JC. Sleep disturbances in progressive supranuclear palsy. Electroencephalogr Clin Neurophysiol 1978;45:16–25.ArticlePubMed
- 21. Abbott SM, Videnovic A. Sleep disorders in atypical parkinsonism. Mov Disord Clin Pract 2014;1:89–96.ArticlePubMedPMC
- 22. Bloise MC, Berardelli I, Roselli V, Pasquini M, Stirpe P, Colosimo C, et al. Psychiatric disturbances in patients with progressive supranuclear palsy: a case-control study. Parkinsonism Relat Disord 2014;20:965–968.ArticlePubMed
- 23. Gerstenecker A, Mast B, Duff K, Ferman TJ, Litvan I; ENGENE-PSP Study Group. Executive dysfunction is the primary cognitive impairment in progressive supranuclear palsy. Arch Clin Neuropsychol 2013;28:104–113.ArticlePubMedPDF
- 24. Stanton BR, Leigh PN, Howard RJ, Barker GJ, Brown RG. Behavioural and emotional symptoms of apathy are associated with distinct patterns of brain atrophy in neurodegenerative disorders. J Neurol 2013;260:2481–2490.ArticlePubMedPDF
- 25. Bae HJ, Cheon SM, Kim JW. Autonomic dysfunctions in parkinsonian disorders. J Mov Disord 2009;2:72–77.ArticlePubMedPMCPDF
- 26. Sakakibara R, Hattori T, Tojo M, Yamanishi T, Yasuda K, Hirayama K. Micturitional disturbance in progressive supranuclear palsy. J Auton Nerv Syst 1993;45:101–106.ArticlePubMed
- 27. Griffiths DJ, Fowler CJ. The micturition switch and its forebrain influences. Acta Physiol (Oxf) 2013;207:93–109.ArticlePubMed
- 28. Stamelou M, Dohmann H, Brebermann J, Boura E, Oertel WH, Höglinger G, et al. Clinical pain and experimental pain sensitivity in progressive supranuclear palsy. Parkinsonism Relat Disord 2012;18:606–608.ArticlePubMed
- 29. Papapetropoulos S, Mash DC. Visual hallucinations in progressive supranuclear palsy. Eur Neurol 2005;54:217–219.ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Autonomic dysfunction in progressive supranuclear palsy
Francesca Baschieri, Maria Vitiello, Pietro Cortelli, Giovanna Calandra-Buonaura, Francesca Morgante
Journal of Neurology.2023; 270(1): 109. CrossRef - PDQ-8: A Simplified and Effective Tool Measuring Life Quality in Progressive Supranuclear Palsy
Xin-Yi Li, Ming-Jia Chen, Xiao-Niu Liang, Rui-Xin Yao, Bo Shen, Bin Wu, Gen Li, Yi-Min Sun, Jian-Jun Wu, Feng-Tao Liu, Yu-Jie Yang, Jian Wang
Journal of Parkinson's Disease.2023; 13(1): 83. CrossRef - Non-motor symptoms in multiple system atrophy: A comparative study with Parkinson's disease and progressive supranuclear palsy
Wen-Zheng Hu, Ling-Xiao Cao, Jin-Hui Yin, Xue-Song Zhao, Ying-Shan Piao, Wei-Hong Gu, Jing-Hong Ma, Zhi-Rong Wan, Yue Huang
Frontiers in Neurology.2023;[Epub] CrossRef - Neurological update: the palliative care landscape for atypical parkinsonian syndromes
Noreen O’Shea, Shane Lyons, Stephen Higgins, Sean O’Dowd
Journal of Neurology.2023; 270(4): 2333. CrossRef - Structural correlates of survival in progressive supranuclear palsy
Duncan Street, W Richard Bevan-Jones, Maura Malpetti, P Simon Jones, Luca Passamonti, Boyd CP. Ghosh, Timothy Rittman, Ian TS. Coyle-Gilchrist, Kieren Allinson, Catherine E. Dawson, James B. Rowe
Parkinsonism & Related Disorders.2023; 116: 105866. CrossRef - Migraine and Tension-type Headache in Parkinson’s Disease and Progressive Supranuclear Palsy/Corticobasal Syndrome
Vinayak Jatale, Ashutosh Tiwari, Mritunjai Kumar, Ravi Gupta, Niraj Kumar
Annals of Indian Academy of Neurology.2023; 26(5): 708. CrossRef - A Systematic Review of Apathy and Depression in Progressive Supranuclear Palsy
Joshua Flavell, Peter J. Nestor
Journal of Geriatric Psychiatry and Neurology.2022; 35(3): 280. CrossRef - The Burden of Progressive Supranuclear Palsy on Patients, Caregivers, and Healthcare Systems by PSP Phenotype: A Cross-Sectional Study
Demetris Pillas, Alexander Klein, Teresa Gasalla, Andreja Avbersek, Alexander Thompson, Jack Wright, Jennifer Mellor, Anna Scowcroft
Frontiers in Neurology.2022;[Epub] CrossRef - Fatigue in hypokinetic, hyperkinetic, and functional movement disorders
Ilaria Antonella Di Vico, Giovanni Cirillo, Alessandro Tessitore, Mattia Siciliano, Massimo Venturelli, Cristian Falup-Pecurariu, Gioacchino Tedeschi, Francesca Morgante, Michele Tinazzi
Parkinsonism & Related Disorders.2021; 86: 114. CrossRef - Prevalence and Characteristics of Polyneuropathy in Atypical Parkinsonian Syndromes: An Explorative Study
Rachel Rohmann, Eva Kühn, Raphael Scherbaum, Lovis Hilker, Saskia Kools, Leonard Scholz, Katharina Müller, Sophie Huckemann, Christiane Schneider-Gold, Ralf Gold, Kalliopi Pitarokoili, Lars Tönges, Eun Hae Kwon
Brain Sciences.2021; 11(7): 879. CrossRef - Understanding fatigue in progressive supranuclear palsy
Jong Hyeon Ahn, Joomee Song, Dong Yeong Lee, Jinyoung Youn, Jin Whan Cho
Scientific Reports.2021;[Epub] CrossRef - “Parkinson’s disease” on the way to progressive supranuclear palsy: a review on PSP-parkinsonism
Ján Necpál, Miroslav Borsek, Bibiána Jeleňová
Neurological Sciences.2021; 42(12): 4927. CrossRef - Clinical progression of progressive supranuclear palsy: impact of trials bias and phenotype variants
Duncan Street, Maura Malpetti, Timothy Rittman, Boyd C P Ghosh, Alexander G Murley, Ian Coyle-Gilchrist, Luca Passamonti, James B Rowe
Brain Communications.2021;[Epub] CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite

