Articles
- Page Path
- HOME > J Mov Disord > Volume 13(2); 2020 > Article
-
Review Article
COVID-19: An Early Review of Its Global Impact and Considerations for Parkinson’s Disease Patient Care -
Roongroj Bhidayasiri1
 , Sasivimol Virameteekul1, Jong-Min Kim2, Pramod Kr. Pal3
, Sasivimol Virameteekul1, Jong-Min Kim2, Pramod Kr. Pal3 , Sun-Ju Chung4
, Sun-Ju Chung4
-
Journal of Movement Disorders 2020;13(2):105-114.
DOI: https://doi.org/10.14802/jmd.20042
Published online: April 30, 2020
1Chulalongkorn Centre of Excellence for Parkinson’s Disease & Related Disorders, Department of Medicine, Faculty of Medicine, Chulalongkorn University and King Chulalongkorn Memorial Hospital, Thai Red Cross Society, Bangkok, Thailand
2Department of Neurology, Seoul National University College of Medicine, Seoul National University Bundang Hospital, Seongnam, Korea
3Department of Neurology, National Institute of Mental Health and Neurosciences, Bengaluru, India
4Department of Neurology, Asan Medical Centre, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding author: Roongroj Bhidayasiri, MD, FRCP, FRCPI Chulalongkorn Center of Excellence for Parkinson’s Disease & Related Disorders, Chulalongkorn University Hospital, 1873 Rama 4 Road, Bangkok 10330, Thailand / Tel: +662-256-4000 / Fax: +662-256-4630 / E-mail: rbh@chulapd.org
Copyright © 2020 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- ABSTRACT
- ARE PARKINSON’S DISEASE PATIENTS SUSCEPTIBLE TO SEVERE COVID-19?
- DO PARKINSON’S DISEASE PATIENTS DETERIORATE DUE TO THE COVID-19 PANDEMIC?
- ARE THERE POTENTIAL CONCERNS FOR POSTENCEPHALITIC PARKINSONISM WITH COVID-19?
- MANAGEMENT OF PARKINSON’S DISEASE DURING THE COVID-19 OUTBREAK
- CONCLUSION
- Notes
- Acknowledgments
- REFERENCES
ABSTRACT
- While many infectious disorders are unknown to most neurologists, COVID-19 is very different. It has impacted neurologists and other health care workers, not only in our professional lives but also through the fear and panic within our own families, colleagues, patients and their families, and even in the wider public. COVID-19 affects all sorts of individuals, but the elderly with underlying chronic conditions are particularly at risk of severe disease, or even death. Parkinson’s disease (PD) shares a common profile as an age-dependent degenerative disorder, frequently associated with comorbidities, particularly cardiovascular diseases, so PD patients will almost certainly fall into the high-risk group. Therefore, the aim of this review is to explore the risk of COVID-19 in PD based on the susceptibility to severe disease, its impact on PD disease severity, potential long-term sequelae, and difficulties of PD management during this outbreak, where neurologists face various challenges on how we can maintain effective care for PD patients without exposing them, or ourselves, to the risk of infection. It is less than six months since the identification of the original COVID-19 case on New Year’s Eve 2019, so it is still too early to fully understand the natural history of COVID-19 and the evidence on COVID-19-related PD is scant. Though the possibilities presented are speculative, they are theory-based, and supported by prior evidence from other neurotrophic viruses closely related to SARS-CoV-2. Neurologists should be on high alert and vigilant for potential acute and chronic complications when encountering PD patients who are suspected of having COVID-19.
- Clinical vignette
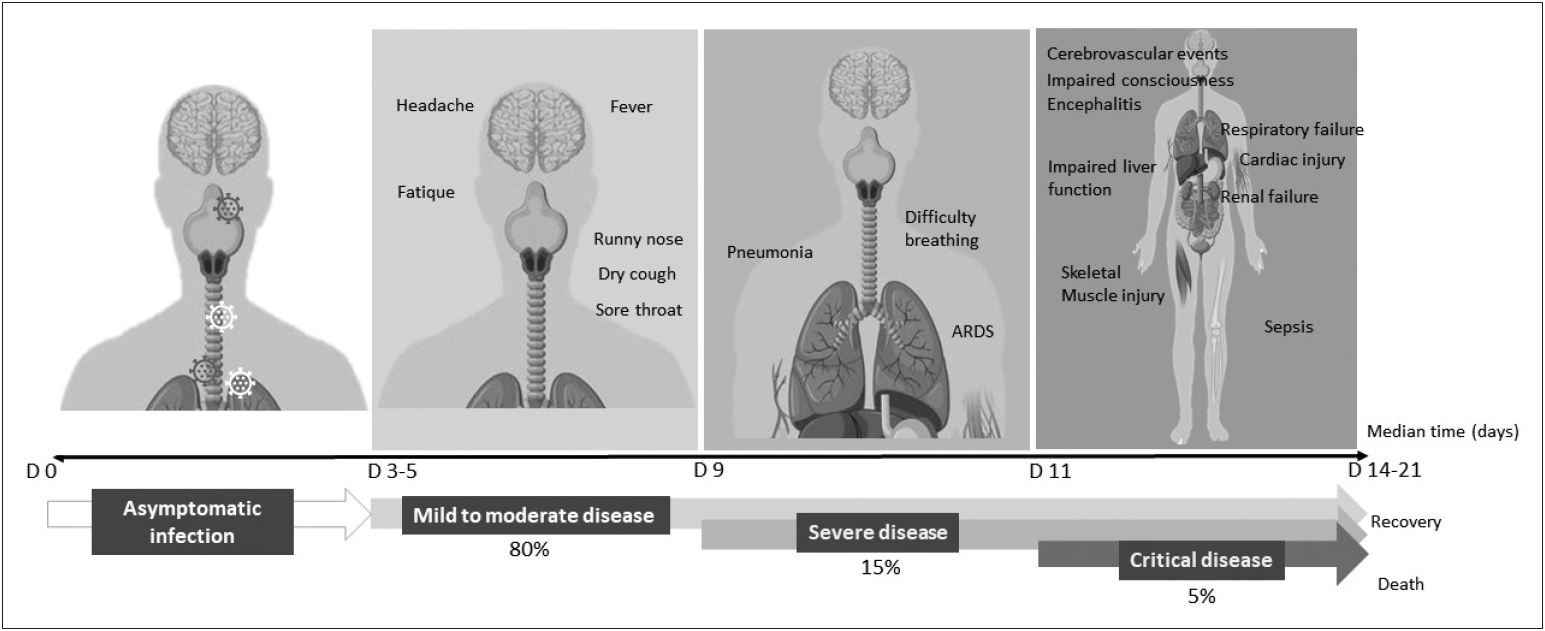
- A 77-year-old Korean man with Parkinson’s disease dementia was admitted to the emergency room with alteration of consciousness on February 23, 2020. His past medical history was significant for coronary artery disease (CAD). At admission, he was minimally responsive and hypotensive (61/35, PR = 79/min) with a body temperature of 36°C. He had tachypnea (RR = 26/min) with oxygen saturation of 90% on room air. A diagnosis of septicemia with acute respiratory distress syndrome (ARDS) was suspected and he was treated with inotropes, antibiotics, and respiratory support.
- Chest computed tomography revealed diffuse bilateral ground glass opacities and RT-PCR assays of the throat and nasal swab samples were reported two days later as positive for SARS-CoV-2, confirming the diagnosis of COVID-19. The family decided against resuscitation and he was given supportive treatment, without a ventilator, in a negative pressure isolation room. He died six days after admission from respiratory failure (Personal communication with Prof. Ho-Won Lee, Kyungpook National University Chilgok Hospital, South Korea).
- Presence of comorbidities, especially cardiovascular disease
- This case illustrates an example of a critical COVID-19 patient who fulfilled the risk criteria for severe disease, including old age and cardiovascular disease. This case also provides perspectives that we can all learn from in terms of the understanding and treatment of this disease, as well as preventive precautions for family members and wider society. From the underlying disease perspective, a cardiovascular disorder comorbidity is considered a high risk for severe disease, with the prevalence of cardiovascular disease reported as between 2.5–12.1% of patients with COVID-19 [3,22]. Compared to the overall CFR of 3.8%, the presence of comorbid conditions significantly increases the CFR, with the highest for cardiovascular disease (13.2%), followed by diabetes (9.2%), hypertension (8.4%), chronic respiratory disorder (8.0%), and cancer (7.6%) [4]. CFR can also increase with age, with the highest mortality observed among people over 80 years (CFR 21.9%) as well as male sex (CFR 4.7%) and retirees (CFR 8.9%) [4]. Although PD is not included as a risk factor for severe disease, PD was mentioned as an underlying disorder associated with mortality in some case series, such as in our case vignette [23].
- Whether PD itself poses a risk for severe disease is not entirely clear. Based on published literature since the identification of COVID-19, there is no conclusive evidence of such a risk in PD. The experience from the three most heavily affected regions in Italy does not show an apparent increased risk, although there are no systematic data available [24]. While the risk cannot be confirmed for the whole PD population, there are several theoretical possibilities that a risk for severe disease may still occur in certain PD subgroups. Indirect risk can be estimated through the strong link between PD and a number of comorbidities, particularly cardiovascular disease. Several cardiac abnormalities have been observed in PD patients, including cardiac autonomic dysfunction, cardiomyopathy, CAD, arrhythmias, and sudden cardiac death [25]. While the associations between cardiovascular risk factors and PD are not straightforward, the link between cardiovascular disease and PD may be related to a shared sedentary lifestyle in both conditions [26]. In addition, diabetes and cerebrovascular disease are also identified as frequent comorbidities in PD patients and are recognized risk factors for severe disease with COVID-19 [27,28].
- Subgroups of Parkinson’s disease patients with underlying respiratory dysfunction and increased risk for aspiration pneumonia
- Direct risk for a severe disease with COVID-19 is likely to occur in a subgroup of PD patients, particularly those with respiratory dysfunction. Indeed, dyspnea is a common symptom in PD, observed in 39.2% of patients without a history of lung or heart disease [29]. With frequent cardio-pulmonary comorbidities among PD patients, the number of PD patients with dyspnea is likely to be higher in clinical practice than that observed in clinical trials. When comorbidities are excluded, dyspnea in PD is primarily associated with advanced disease with contributing factors from respiratory muscle weakness, axial manifestations, and abnormal posture (e.g., camptocormia, stoop), causing respiratory muscle rigidity and poor respiratory excursions, finally resulting in ventilatory failure [29,30]. In PD, dyspnea may also occur as part of nonmotor fluctuations, frequently associated with anxiety and depression [31]. Furthermore, the presence of restrictive or obstructive respiratory dysfunction can potentially predispose PD patients to sleep apnea, further compromising these patients’ night-time breathing function [32]. While inspiratory muscle weakness may be clinically evident in advanced stage patients, it has recently been found to be present at a subclinical level in early stage patients whose pulmonary function tests did not disclose any restrictive or obstructive disorders [33]. Central effects of respiratory dysfunction in PD cannot be discounted as an early stage of neurodegeneration in PD also involving brainstem nuclei, affecting the respiratory center in the medulla, which is also targeted by SARS-CoV-2 infection [8]. Therefore, PD patients with underlying respiratory dysfunction could be at significant risk of respiratory failure if infected by SARS-CoV-2.
- Pneumonia was identified as the foremost reason for hospital admissions and the most common cause of death among PD patients [34,35]. The inverse association also demonstrates that PD by itself confers a higher probability of dying from pneumonia [36]. As PD progresses, mastication is affected and swallowing is impaired by bradykinesia, rigidity, and dyskinesia, causing pooling of saliva in the mouth, leading to drooling or aspiration of secretions [37,38]. This is important as aspiration of saliva may also occur in between meals. In advanced patients who are weak and frail, their cough becomes weaker because of chest wall rigidity and decreases in the sensory component of the cough reflex. This combination represents a perfect scenario for aspiration pneumonia. As COVID-19 is mainly transmitted through respiratory droplets from coughing and sneezing ultimately infecting alveolar epithelial cells, PD patients may predispose themselves to severe pneumonia by aspirating infected secretions into the lower respiratory tract. However, while this mechanism may seem plausible, there is no published literature supporting this association in PD patients with COVID-19.
- Possible altered immune responses in Parkinson’s disease patients
- Relative to viruses that cause human upper respiratory tract infection, viruses that cause lower respiratory tract infection are smaller and their infections are mostly limited to tracheobronchitis in healthy individuals but can cause viral pneumonias in immunocompromised patients. Before the outbreak of COVID-19, the most frequent virus that caused acute lower respiratory infections in China was respiratory syncytial virus (RSV), but influenza viruses were the main cause in adults and the elderly [39]. Human coronavirus represented only a minority (1.4%) within this Chinese cohort. If we assume that PD represents a risk for severe disease consisting of pneumonia and ARDS with COVID-19, it is important to determine the immune status responses in general PD populations or certain PD subgroups to identify if they are immunocompromised. Indeed, the concern whether PD can lower immunity in affected patients, making them more susceptible to COVID-19, was recently shared by PD patients and caregivers [40]. Importantly, evidence of dysregulation of immune responses in patients with severe COVID-19 infection is emerging, characterized by elevated levels of infection-related biomarkers and inflammatory cytokines, including a low number of T cells in more debilitating and severe cases [41]. Both helper T cells and suppressor T cells in patients with COVID-19 were found to be below normal levels, with the lowest level of helper T cells in the severe group. In severe cases, the percentage of memory helper and regulatory T cells was also decreased. As PD typically affects people from the sixth decade of life, it is also important to determine a state of immunosenescence, which is associated with dramatic changes in the distribution and competence of immune cells, leading to the loss of adaptive immunity and the gain of nonspecific innate immunity, leaving older individuals susceptible to infection and cancer and unprotected from chronic tissue inflammation [42]. The cooccurrence of weakened adaptive immunity with a bias toward nonspecific tissue inflammation (‘inflammaging’) probably explains why the elderly, particularly those above 80 years, are highly susceptible to severe COVID-19.
ARE PARKINSON’S DISEASE PATIENTS SUSCEPTIBLE TO SEVERE COVID-19?
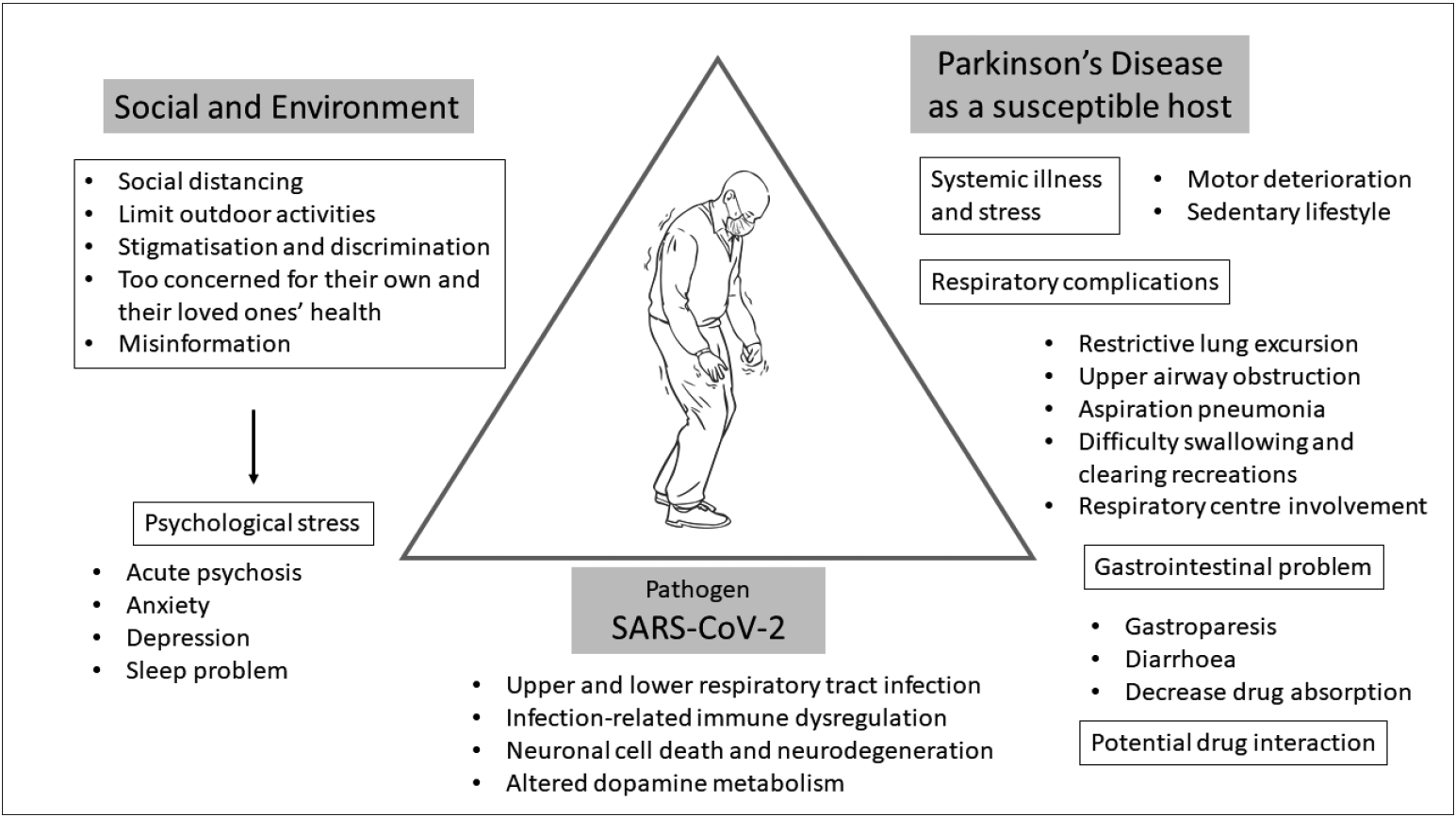
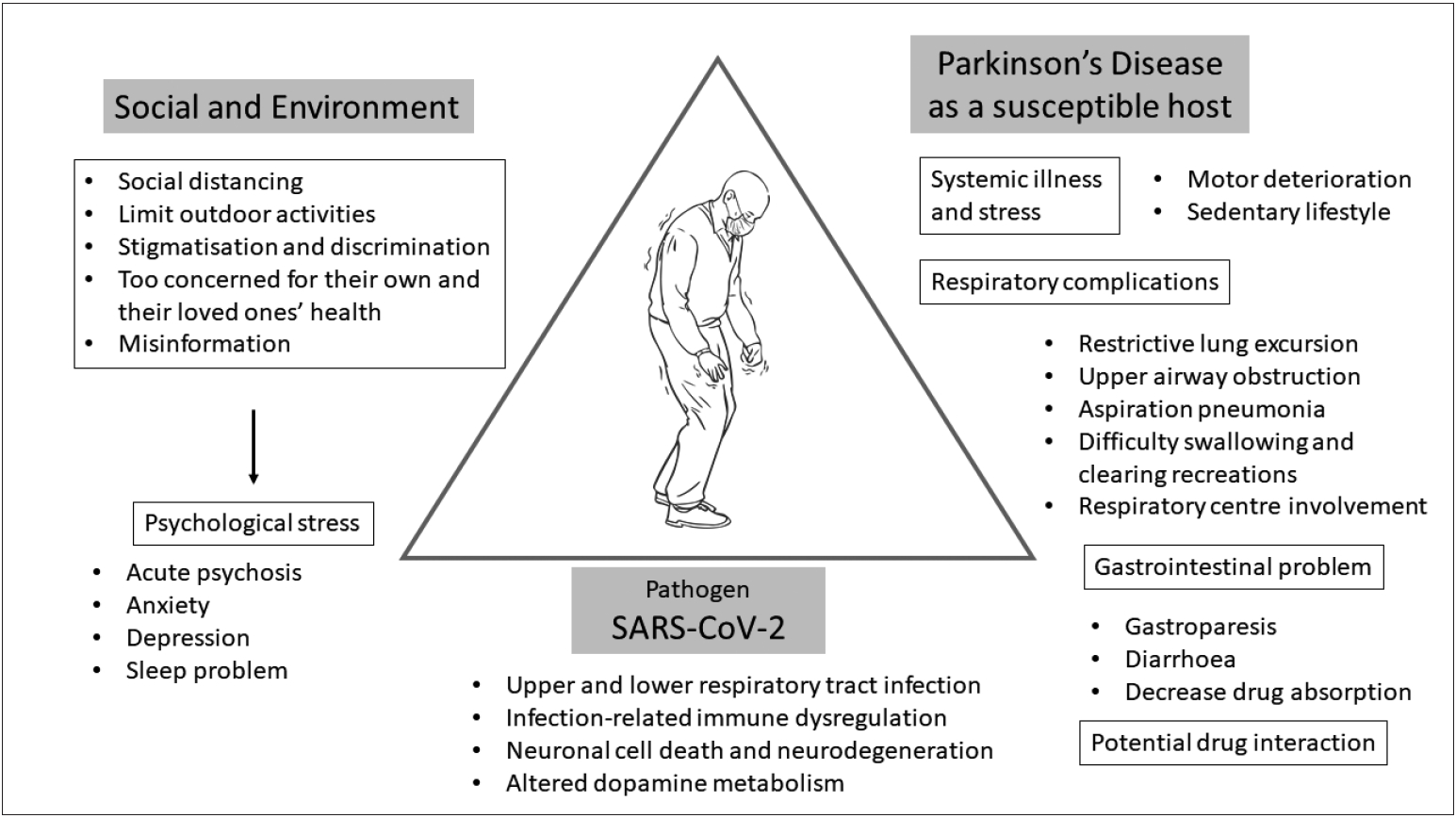
- To adequately address this question, we need to determine various aspects of how COVID-19 by itself and by environmental and social factors could have an effect on PD patients (Figure 3). At this moment, there are no available data that provide sufficient information on how COVID-19 itself has an effect on parkinsonian symptoms except for scattered mortality case reports, which include PD among many other underlying disorders. While this lack of information may be interpreted as there being no significant effect of COVID-19 on the overall symptoms of PD patients, it is probably too early to estimate this association and neurologists should be on high alert to evaluate parkinsonian symptoms comprehensively with a particular emphasis on respiratory function for those infected with SARS-CoV-2.
- Possible direct impact of COVID-19 on Parkinson’s disease patients with COVID-19
- Despite a lack of specific data on COVID-19, the available evidence indicates that PD patients can deteriorate following systemic infections [43]. The clinical spectrum of this phenomenon is wide, ranging from mild worsening to akinetic crisis [43]. Fever and delirium are two of the most important factors found to be associated with motor deterioration and respiratory tract infection found to be the most common contributor [44,45]. As fever is the most common symptom of COVID-19, observed in 87.9% of affected patients and up to 12.3% experience a high fever of more than 39.0°C, there is a strong possibility of subacute worsening of parkinsonism for PD patients with COVID-19 [3,4]. The prevalence of this phenomenon was found to be between 25–32.5% with advanced disease, characterized by long disease duration, high Hoehn & Yahr (HY) stage, cognitive dysfunction, and high dopaminergic medication dosages as potential risk factors [44,45]. The combination of fever and altered dopaminergic medication intake can potentially predispose PD patients to Parkinsonism-hyperpyrexia syndrome, which is a movement disorder emergency, requiring immediate and aggressive management [46]. Despite improvement of the underlying infection, not all PD patients with this phenomenon fully recover, and some are left with significant disability. Fatal cases have also been reported [45]. The mechanisms underlying this motor deterioration are likely to be complex with variable contributions from individual cases, but include altered medication intake during systemic illnesses, changes in the pharmacodynamics of dopaminergic medications, altered dopamine metabolism in the brain, peripheral inflammatory processes or circulating toxins or cytokines [43]. Therefore, PD patients who possess these risks should be carefully observed and monitored for motor deterioration during a period of active SARS-CoV-2 infection, which should also be extended into the convalescence period.
- Possible indirect impact of COVID-19 on Parkinson’s disease patients without COVID-19
- The impact of universal preventative measures for COVID-19 exposure on symptoms, functions, activities and stress of PD patients should not be ignored [47]. Advice for everybody to ‘stay-athome’ can limit patient’s outdoor physical activities and mobility, resulting in a sedentary lifestyle for some patients, potentially leading to worsening outcomes. Social distancing limits face-to-face family visits to patients who live alone, and may result in feelings of loneliness and depression. When patients stay at home, they are more likely to spend more time watching the television, listening to the radio and using social media with their 24-hour broadcasting of COVID-19 situations, potentially increasing psychological stress, worsening anxiety and depression that are already common nonmotor symptoms in PD patients. With high levels of fear, PD patients and their families may not think clearly and rationally when reacting to COVID-19, potentially leading to other psychosocial challenges, including stigmatization and discrimination [48]. Psychological stress can affect various motor symptoms, such as tremor, gait, and dyskinesia, as well as reduce the efficacy of dopaminergic medications [49,50].
DO PARKINSON’S DISEASE PATIENTS DETERIORATE DUE TO THE COVID-19 PANDEMIC?
- So far, no reports of acute parkinsonism following COVID-19 have been reported in the literature. It remains to be seen if SARS-CoV-2 can induce parkinsonism following an episode of encephalitis. The issue of a viral etiology of encephalitis lethargica and postencephalitic parkinsonism continues to be a matter of debate. Much of the linkage of parkinsonism with influenza and many other viruses stem from an outbreak of encephalitis lethargica (EL), also known as von Economo’s disease, and postencephalitic parkinsonism that occurred subsequent to the 1918 pandemic influenza outbreak caused by a type A H1N1 influenza virus [51]. The cause of EL and the link to subsequent postencephalitic parkinsonism remains controversial with clinical features showing both similarities and distinct symptomatology to PD. Following an acute episode, the chronic phase of postencephalitic parkinsonism developed one to five years later, but it could also follow immediately, or more than a decade later, with typical clinical presentations involving upper limb bradykinesia and stiffness associated with frequent episodes of kinesia paradoxica, oculogyric crisis, and psychiatric disturbances [52]. Perhaps, EL is not necessarily a prerequisite to developing postencephalitic parkinsonism, but merely a contributing factor [52]. Although direct evidence for influenza virus has never been substantiated in these cases, proving a negative is also difficult. Despite the etiological controversy, people born during the time of the pandemic influenza outbreak of 1918 have a two-to-three fold-increased risk of PD than those born prior to 1888 or after 1924 [53-55]. While EL and COVID-19 involve different primary organs for their clinical manifestations, they are both pandemics affecting more than one million people with a link to neurotrophic viruses. In addition to parkinsonism, there is a theoretical possibility that SARS-CoV-2 may contribute to accelerated CNS aging in survivors, which could manifest months or years after the infection [56]. Therefore, the medical community should be cautious of neurological comorbidities of COVID-19 that may occur following an outbreak.
ARE THERE POTENTIAL CONCERNS FOR POSTENCEPHALITIC PARKINSONISM WITH COVID-19?
- As the outbreak of COVID-19 continues globally, neurologists face challenges with new tasks related to the care of COVID-19 patients and existing tasks in providing a continuum of care for their neurological patients, but in a very different situation from their usual clinical practice. Most of the adjustments were not planned in advance and involve maximizing and reallocating human, clinical, and research resources to the priority list of taking care of COVID-19 patients. Undoubtedly, these changes not only affect the routine care of PD patients but also highlight a number of important issues that neurologists should be aware of or consider implementing during this outbreak.
- Concerns on dopaminergic medications and device-aided therapies
- As mentioned above, PD patients may experience a subacute worsening of their condition, especially motor symptoms, during systemic infections, potentially as a result of altered medication intake and changes in the pharmacodynamics of dopaminergic medications. For those with mild symptoms of COVID-19, patients should be advised to observe their symptoms closely and to seek consultation if their condition, particularly respiratory symptoms, significantly worsen. While most dopaminergic medications can be continued during a period of systemic infections without any absolute contraindications, caution should be noted with the use of cough syrup containing dextromethorphan and cyclobenzaprine or nasal decongestants containing pseudoephedrine, phenylephrine, and phenylpropanolamine with a monoamine oxidase inhibitor (e.g., selegiline and rasagiline) for potential drug interactions that can intensify sympathomimetic activities [57].
- Antiviral (e.g., favipiravir, atazanavir, iopinavir/ritonavir) and anti-malarial (chloroquine and hydroxychloroquine) drugs are being tested for COVID-19 without any specific interactions with dopaminergic medications reported. Therapeutic implications of amantadine, an agent that can block a pore in the envelope protein of SARS-CoV and is no longer used as an antiinfluenza agent due to its high resistance, remain unexplored as a potential treatment of COVID-19 [58].
- Patients with device-aided therapies, including deep brain stimulation (DBS), apomorphine and levodopa-carbidopa intestinal gel infusions may encounter problems related to hardware, acute adverse events, or severe symptom fluctuations during this outbreak [59-61]. While the recommendations are to postpone device-aided therapies for all new cases, acute problems may arise among those who are already on one of these treatments where immediate interventions may be necessary. For example, if the battery for a DBS system stops working completely and the DBS is no longer effective, patients may experience a significant return of PD symptoms. Although most DBS procedures are considered elective, it is not an elective procedure if it is the end of battery life. In these situations, decisions should be made individually by taking into consideration the risk of complications, exposure risk to COVID-19 for both patients and health care workers, and the availability of all resources to treat that particular complication during this outbreak. In places where there are no limitations of resources, elective procedures can be resumed but full consultations with various experts and appropriate health authorities should be undertaken to ensure that it is safe to do so.
- Concerns on outpatient and ambulatory care settings of PD: emerging roles of telemedicine
- Another challenge for us as neurologists is how to maintain effective care for PD patients without exposing them to the risk of COVID-19 in the hospital. CDC recommends healthcare facilities delay elective ambulatory provider visits and adjust the way they triage, assess, and care for patients by using methods that do not rely on face-to-face care, such as the use of telemedicine [62]. Telemedicine encompasses a broad range of health care tools, including real-time interactive or synchronous audio and video communications between a patient and a provider (e.g., video conferencing, smartphone health care applications), and other telehealth services, such as simple telephone communication, email and text messages, and remote monitoring of patient data from wearables [20]. Previous studies have suggested that telemedicine for PD is feasible when delivered either in the home or in an off-site clinic or nursing home setting [63]. Most studies have also reported that an adequate and reliable motor examination can be performed via telemedicine with the motor section of the Unified Parkinson’s Disease Rating Scale (UPDRS-III) [64-66]. However, limitations remain for the examination of rigidity, postural instability, and cognitive dysfunction [67]. Benefits with telemedicine are also extended to cost savings, reduction in travel distance, and a reduction in time spent attending appointments [63]. Moreover, rehabilitation and allied health services can be delivered remotely with success stories demonstrated on the Lee Silverman Voice Treatment and physical therapy assessments [68,69]. In general, patient’s views on telemedicine have been favorable for the main advantages, including access to specialists and convenience, but tempered by the concern of loss of hands on care [70]. However, issues with laws, regulations, policies and patient’s confidentiality with telemedicine need to be addressed by the parties involved.
MANAGEMENT OF PARKINSON’S DISEASE DURING THE COVID-19 OUTBREAK
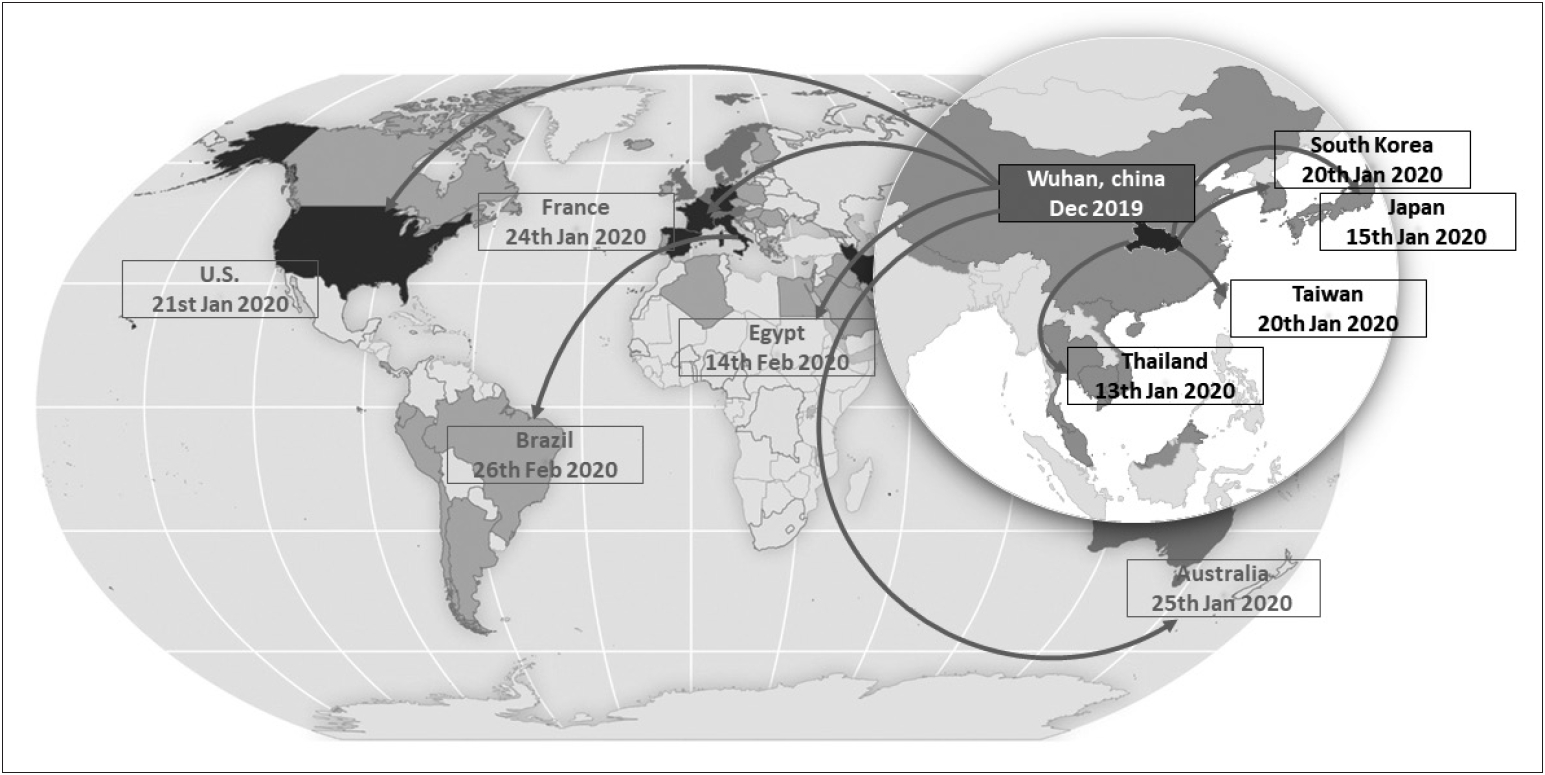
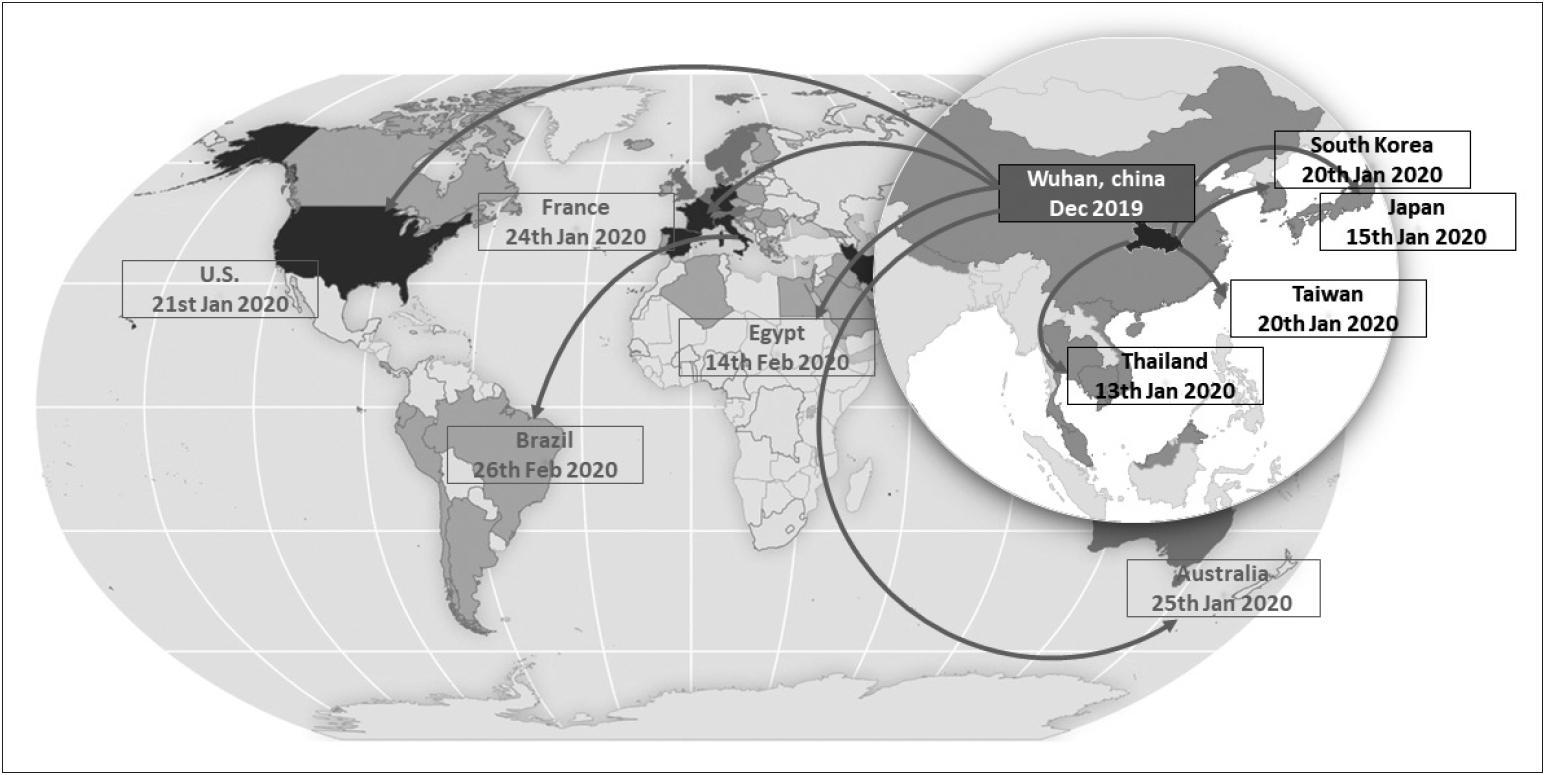
- While many infectious disorders are unknown to most neurologists, COVID-19 is very different. It has made itself known to neurologists and other health care workers not only in their professions but also because of the fear and panic caused within our own families, colleagues, patients and their families, and the wider public. Since the identification of SARS-CoV-2 on New Year’s Eve 2019, it has spread to 213 countries on almost all continents in less than six months and was declared by WHO as a global pandemic. During this time period, COVID-19 has shown that it is not just a respiratory disorder as neurological complications start to emerge, ranging from mild headache and dizziness to acute encephalitis and loss of consciousness. COVID-19 affects all individuals, but it causes high mortality among the elderly and those with various underlying chronic medical conditions. PD patients are probably included in this at-risk group and the whole PD community should act together to understand, prevent, and protect themselves from COVID-19. If prevention is not possible and PD patients get infected, they should be closely monitored for worsening of their parkinsonian symptoms and signs of severe disease, especially with respect to respiratory function. It is likely that advanced PD patients with existing underlying respiratory dysfunction are particularly at-risk for severe disease and eventually respiratory and multiorgan failure. Those elderly PD patients with comorbidities (e.g., cardiovascular disease, diabetes) will have a mortality risk that is even higher. While no specific treatments are yet available, prompt supportive management of respiratory function is likely to influence outcomes. Based on its nature of neurotropism, sequelae may start to reveal themselves with time. Some early evidence of hyposmia and encephalitis are important clues that neurologists should continue to follow COVID-19 patients even though they are seronegative and discharged home since long-term complications, including parkinsonism, are all possible.
CONCLUSION
- Roongroj Bhidayasiri is supported by a Thailand Science Research and Innovation grant (RTA6280016), International Research Network grant (IRN59W0005) of the Thailand Research Fund, Chulalongkorn Academic Advancement Fund into Its 2nd Century Project of Chulalongkorn University (2300042200), and Center of Excellence grant to Chulalongkorn University (GCE 6100930004-1).
Acknowledgments
- 1. Coronavirus disease (COVID-19) Pandemic. World Health Organization; 2020 [updated 2020; cited 2020 9 April]; Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019.
- 2. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med 2020;382:727–733.ArticlePubMedPMC
- 3. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med 2020.Article
- 4. Report of the WHO-China Joint Mission on Coronavisus Disease 2019 (COVID-19). World Health Organization; 2020.
- 5. The L. COVID-19: protecting health-care workers. Lancet 2020;395:922.Article
- 6. Ferioli M, Cisternino C, Leo V, Pisani L, Palange P, Nava S. Protecting healthcare workers from SARS-CoV-2 infection: practical indications. Eur Respir Rev 2020;29:Article
- 7. Lechien JR, Chiesa-Estomba CM, De Siati DR, Horoi M, Le Bon SD, Rodriguez A, et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur Arch Otorhinolaryngol 2020.Article
- 8. Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol 2020.ArticlePDF
- 9. Coronavirus disease 2019 (COVID-19): Groups at Higher Risk for Severe Illness. Centers for Disease Control and Prevention (CDC); 2020 [updated 2020; cited]; Available from: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/groups-at-higher-risk.html.
- 10. Zhou L, Zhang M, Wang J, Gao J. Sars-Cov-2: Underestimated damage to nervous system. Travel Med Infect Dis 2020;101642.ArticlePubMedPMC
- 11. Moriguchi T, Harii N, Goto J, Harada D, Sugawara H, Takamino J, et al. A first Case of Meningitis/Encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis 2020.Article
- 12. Korean Society of Infectious D, Korea Centers for Disease C, Prevention. Analysis on 54 Mortality Cases of Coronavirus Disease 2019 in the Republic of Korea from January 19 to March 10, 2020. J Korean Med Sci 2020;35:e132. ArticlePubMedPMCPDF
- 13. Zhao H, Shen D, Zhou H, Liu J, Chen S. Guillain-Barre syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol 2020.Article
- 14. Poyiadji N, Shahin G, Noujaim D, Stone M, Patel S, Griffith B. COVID19-associated Acute Hemorrhagic Necrotizing Encephalopathy: CT and MRI Features. Radiology 2020;201187.ArticlePubMed
- 15. Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, et al. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol 2020.ArticlePDF
- 16. Helms J, Kremer S, Merdji H, Clere-Jehl R, Schenck M, Kummerlen C, et al. Neurologic Features in Severe SARS-CoV-2 Infection. N Engl J Med 2020.Article
- 17. Nath A. Neurologic complications of coronavirus infections. Neurology 2020.Article
- 18. Baig AM, Khaleeq A, Ali U, Syeda H. Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host-Virus Interaction, and Proposed Neurotropic Mechanisms. ACS Chem Neurosci 2020;11:995–998.ArticlePubMedPMC
- 19. Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020;367:1444–1448.ArticlePubMedPMC
- 20. Klein BC, Busis NA. COVID-19 is catalyzing the adoption of teleneurology. Neurology 2020.Article
- 21. Waldman G, Mayeux R, Claassen J, Agarwal S, Willey J, Anderson E, et al. Preparing a neurology department for SARS-CoV-2 (COVID-19): Early experiences at Columbia University Irving Medical Center and the New York Presbyterian Hospital in New York City. Neurology 2020.Article
- 22. Emami A, Javanmardi F, Pirbonyeh N, Akbari A. Prevalence of Underlying Diseases in Hospitalized Patients with COVID-19: a Systematic Review and Meta-Analysis. Arch Acad Emerg Med 2020;8:e35. PubMedPMC
- 23. Adhikari SP, Meng S, Wu YJ, Mao YP, Ye RX, Wang QZ, et al. Epidemiology, causes, clinical manifestation and diagnosis, prevention and control of coronavirus disease (COVID-19) during the early outbreak period: a scoping review. Infect Dis Poverty 2020;9:29.ArticlePubMedPMCPDF
- 24. Papa SM, Brundin P, Fung VSC, Kang UJ, Burn DJ, Colosimo C, et al. Impact of the COVID-19 pandemic on Parkinson’s disease and movement disorders. Mov Disord 2020.
- 25. Scorza FA, Fiorini AC, Scorza CA, Finsterer J. Cardiac abnormalities in Parkinson’s disease and Parkinsonism. J Clin Neurosci 2018;53:1–5.ArticlePubMed
- 26. Potashkin J, Huang X, Becker C, Chen H, Foltynie T, Marras C. Understanding the links between cardiovascular disease and Parkinson’s disease. Mov Disord 2020;35:55–74.ArticlePubMed
- 27. Yue X, Li H, Yan H, Zhang P, Chang L, Li T. Risk of Parkinson Disease in Diabetes Mellitus: An Updated Meta-Analysis of Population-Based Cohort Studies. Medicine (Baltimore) 2016;95:e3549. ArticlePubMedPMC
- 28. Hong CT, Hu HH, Chan L, Bai CH. Prevalent cerebrovascular and cardiovascular disease in people with Parkinson’s disease: a meta-analysis. Clin Epidemiol 2018;10:1147–1154.ArticlePubMedPMC
- 29. Baille G, Chenivesse C, Perez T, Machuron F, Dujardin K, Devos D, et al. Dyspnea: An underestimated symptom in Parkinson’s disease. Parkinsonism Relat Disord 2019;60:162–166.ArticlePubMed
- 30. Baille G, Perez T, Devos D, Machuron F, Dujardin K, Chenivesse C, et al. Dyspnea Is a Specific Symptom in Parkinson’s Disease. J Parkinsons Dis 2019;9:785–791.ArticlePubMed
- 31. Witjas T, Kaphan E, Azulay JP, Blin O, Ceccaldi M, Pouget J, et al. Nonmotor fluctuations in Parkinson’s disease: frequent and disabling. Neurology 2002;59:408–413.ArticlePubMed
- 32. Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci 1998;5:131–135.PubMed
- 33. Baille G, Perez T, Devos D, Deken V, Defebvre L, Moreau C. Early occurrence of inspiratory muscle weakness in Parkinson’s disease. PLoS One 2018;13:e0190400. ArticlePubMedPMC
- 34. Okunoye O, Kojima G, Marston L, Walters K, Schrag A. Factors associated with hospitalisation among people with Parkinson’s disease - A systematic review and meta-analysis. Parkinsonism Relat Disord 2020;71:66–72.ArticlePubMed
- 35. Pennington S, Snell K, Lee M, Walker R. The cause of death in idiopathic Parkinson’s disease. Parkinsonism Relat Disord 2010;16:434–437.ArticlePubMed
- 36. Bugalho P, Ladeira F, Barbosa R, Marto JP, Borbinha C, Salavisa M, et al. Motor and non-motor function predictors of mortality in Parkinson’s disease. J Neural Transm (Vienna) 2019;126:1409–1415.ArticlePubMedPDF
- 37. Leopold NA, Kagel MC. Laryngeal deglutition movement in Parkinson’s disease. Neurology 1997;48:373–376.ArticlePubMed
- 38. Curtis JA, Molfenter S, Troche MS. Predictors of Residue and Airway Invasion in Parkinson’s Disease. Dysphagia 2020;35:220–230.ArticlePubMedPDF
- 39. Feng L, Li Z, Zhao S, Nair H, Lai S, Xu W, et al. Viral etiologies of hospitalized acute lower respiratory infection patients in China, 2009-2013. PLoS One 2014;9:e99419. ArticlePubMedPMC
- 40. Prasad S, Holla VV, Neeraja K, Surisetti BK, Kamble N, Yadav R, et al. Parkinson’s disease and COVID-19: Perceptions and implications in patients and caregivers. Mov Disord 2020.
- 41. Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis 2020.Article
- 42. Weyand CM, Goronzy JJ. Aging of the Immune System. Mechanisms and Therapeutic Targets. Ann Am Thorac Soc 2016;13 Suppl 5:S422–S428.ArticlePubMed
- 43. Brugger F, Erro R, Balint B, Kagi G, Barone P, Bhatia KP. Why is there motor deterioration in Parkinson’s disease during systemic infections-a hypothetical view. NPJ Parkinsons Dis 2015;1:15014.ArticlePubMedPMCPDF
- 44. Umemura A, Oeda T, Tomita S, Hayashi R, Kohsaka M, Park K, et al. Delirium and high fever are associated with subacute motor deterioration in Parkinson disease: a nested case-control study. PLoS One 2014;9:e94944. ArticlePubMedPMC
- 45. Zheng KS, Dorfman BJ, Christos PJ, Khadem NR, Henchcliffe C, Piboolnurak P, et al. Clinical characteristics of exacerbations in Parkinson disease. Neurologist 2012;18:120–124.ArticlePubMedPMC
- 46. Rajan S, Kaas B, Moukheiber E. Movement Disorders Emergencies. Semin Neurol 2019;39:125–136.ArticlePubMedPDF
- 47. Helmich RC, Bloem BR. The Impact of the COVID-19 Pandemic on Parkinson’s Disease: Hidden Sorrows and Emerging Opportunities. J Parkinsons Dis 2020;10:351–354.ArticlePubMedPMC
- 48. Ahorsu DK, Lin CY, Imani V, Saffari M, Griffiths MD, Pakpour AH. The Fear of COVID-19 Scale: Development and Initial Validation. Int J Ment Health Addict 2020;1–9.ArticlePDF
- 49. Hemmerle AM, Herman JP, Seroogy KB. Stress, depression and Parkinson’s disease. Exp Neurol 2012;233:79–86.ArticlePubMed
- 50. Zach H, Dirkx MF, Pasman JW, Bloem BR, Helmich RC. Cognitive Stress Reduces the Effect of Levodopa on Parkinson’s Resting Tremor. CNS Neurosci Ther 2017;23:209–215.ArticlePubMedPMC
- 51. Taubenberger JK. The origin and virulence of the 1918 “Spanish” influenza virus. Proc Am Philos Soc 2006;150:86–112.PubMedPMC
- 52. Hoffman LA, Vilensky JA. Encephalitis lethargica: 100 years after the epidemic. Brain 2017;140:2246–2251.ArticlePubMedPDF
- 53. Maurizi CP. Why was the 1918 influenza pandemic so lethal? The possible role of a neurovirulent neuraminidase. Med Hypotheses 1985;16:1–5.ArticlePubMed
- 54. Poskanzer DC, Schwab RS. Cohort Analysis of Parkinson’s Syndrome: Evidence for a Single Etiology Related to Subclinical Infection About 1920. J Chronic Dis 1963;16:961–973.ArticlePubMed
- 55. Ravenholt RT, Foege WH. 1918 influenza, encephalitis lethargica, parkinsonism. Lancet 1982;2:860–864.ArticlePubMed
- 56. Lippi A, Domingues R, Setz C, Outeiro TF, Krisko A. SARS-CoV-2: at the crossroad between aging and neurodegeneration. Mov Disord 2020.Article
- 57. Pray S. Interactions Between Nonprescription Products and Psychotropic Medications. U.S. Pharmacist; 2007 [updated 2007; cited]; Available from: https://www.uspharmacist.com/article/interactions-between-nonprescription-products-and-psychotropic-medications.
- 58. Torres J, Maheswari U, Parthasarathy K, Ng L, Liu DX, Gong X. Conductance and amantadine binding of a pore formed by a lysine-flanked transmembrane domain of SARS coronavirus envelope protein. Protein Sci 2007;16:2065–2071.ArticlePubMedPMC
- 59. Jitkritsadakul O, Bhidayasiri R, Kalia SK, Hodaie M, Lozano AM, Fasano A. Systematic review of hardware-related complications of Deep Brain Stimulation: Do new indications pose an increased risk? Brain Stimul 2017;10:967–976.ArticlePubMed
- 60. Bhidayasiri R, Garcia Ruiz PJ, Henriksen T. Practical management of adverse events related to apomorphine therapy. Parkinsonism Relat Disord 2016;33 Suppl 1:S42–S48.ArticlePubMed
- 61. Lang AE, Rodriguez RL, Boyd JT, Chouinard S, Zadikoff C, Espay AJ, et al. Integrated safety of levodopa-carbidopa intestinal gel from prospective clinical trials. Mov Disord 2016;31:538–546.ArticlePubMed
- 62. Coronavirus Disease 2019: Outpatient and ambulatory care settings: Responding to community transmission of COVID-19 in the United States. Centers for Disease Control and Prevention (CDC); 2020 [updated 2020; cited]; Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/ambulatory-care-settings.html.
- 63. Schneider RB, Biglan KM. The promise of telemedicine for chronic neurological disorders: the example of Parkinson’s disease. Lancet Neurol 2017;16:541–551.ArticlePubMed
- 64. Dorsey ER, Deuel LM, Voss TS, Finnigan K, George BP, Eason S, et al. Increasing access to specialty care: a pilot, randomized controlled trial of telemedicine for Parkinson’s disease. Mov Disord 2010;25:1652–1659.ArticlePubMed
- 65. Cubo E, Gabriel-Galan JM, Martinez JS, Alcubilla CR, Yang C, Arconada OF, et al. Comparison of office-based versus home Web-based clinical assessments for Parkinson’s disease. Mov Disord 2012;27:308–311.ArticlePubMed
- 66. Abdolahi A, Scoglio N, Killoran A, Dorsey ER, Biglan KM. Potential reliability and validity of a modified version of the Unified Parkinson’s Disease Rating Scale that could be administered remotely. Parkinsonism Relat Disord 2013;19:218–221.ArticlePubMed
- 67. Abdolahi A, Bull MT, Darwin KC, Venkataraman V, Grana MJ, Dorsey ER, et al. A feasibility study of conducting the Montreal Cognitive Assessment remotely in individuals with movement disorders. Health Informatics J 2016;22:304–311.ArticlePubMed
- 68. Dias AE, Limongi JC, Barbosa ER, Hsing WT. Voice telerehabilitation in Parkinson’s disease. Codas 2016;28:176–181.ArticlePubMedPDF
- 69. van der Kolk NM, de Vries NM, Kessels RPC, Joosten H, Zwinderman AH, Post B, et al. Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: a double-blind, randomised controlled trial. Lancet Neurol 2019;18:998–1008.ArticlePubMed
- 70. Spear KL, Auinger P, Simone R, Dorsey ER, Francis J. Patient Views on Telemedicine for Parkinson Disease. J Parkinsons Dis 2019;9:401–404.ArticlePubMed
- 71. Scholthof KB. The disease triangle: pathogens, the environment and society. Nat Rev Microbiol 2007;5:152–156.ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

- Covid-19 in Parkinson's Disease treated by drugs or brain stimulation
M. Salari, M. Etemadifar, A. Zali, Z. Aminzade, I. Navalpotro-Gomez, S. Tehrani Fateh
Neurología.2024; 39(3): 254. CrossRef - Calidad de vida de las personas con enfermedad de Parkinson durante el confinamiento por la pandemia por covid-19
A.M. Fernández-Elgueta, H.F. Retamal-Matus, C. Núñez-Espinosa, P. Barria Aburto
Rehabilitación.2024; 58(1): 100821. CrossRef - Efficacy of non-immersive virtual reality-based telerehabilitation on postural stability in Parkinson's disease: a multicenter randomized controlled trial
Michela GOFFREDO, Francesca BAGLIO, Roberto DE ICCO, Stefania PROIETTI, Giorgio MAGGIONI, Andrea TUROLLA, Sanaz POURNAJAF, Johanna JONSDOTTIR, Federica ZENI, Sara FEDERICO, Luisa CACCIANTE, Matteo CIOETA, Cristina TASSORELLI, Marco FRANCESCHINI, Rocco S.
European Journal of Physical and Rehabilitation Medicine.2024;[Epub] CrossRef - Covid-19 in Parkinson's Disease treated by drugs or brain stimulation
M. Salari, M. Etemadifar, A. Zali, Z. Aminzade, I. Navalpotro-Gomez, S. Tehrani Fateh
Neurología (English Edition).2024; 39(3): 254. CrossRef - COVID-19 and Parkinson’s disease: a systematic review and meta-analysis
Maryam Afraie, Ghobad Moradi, Pardis Mohammadzedeh, Mobin Azami, Sevda Riyahifar, Yousef Moradi
Acta Neurologica Belgica.2023; 123(4): 1209. CrossRef - Survey on the Impact of the COVID-19 Pandemic on Patients with Parkinson’s Disease and Their Caregivers in Japan
Nobutaka Hattori, Yoshiko Okada, Yayoi Kawata, Yoshihiko Furusawa, Takumi Imai, Hisako Yoshida, Mihoko Ota, Masaki Arai, Ayumi Shintani, Jovelle Fernandez
Patient Preference and Adherence.2023; Volume 17: 1221. CrossRef - COVID-19 and neurological disorders: what might connect Parkinson’s disease to SARS-CoV-2 infection
Salvatore Iacono, Giuseppe Schirò, Chiara Davì, Sergio Mastrilli, Michelle Abbott, Fabrizio Guajana, Valentina Arnao, Paolo Aridon, Paolo Ragonese, Cesare Gagliardo, Claudia Colomba, Nicola Scichilone, Marco D’Amelio
Frontiers in Neurology.2023;[Epub] CrossRef - Worsening of Parkinson’s Disease After Termination of COVID-19 Quarantine Cannot Be Reversed Despite Resumption of Physiotherapy
Christian Ineichen, Heide Vogel-Baumann, Matthias Sitzler, Günther Deuschl, Christian R. Baumann
Journal of Parkinson's Disease.2023; 13(5): 845. CrossRef - The Impact of COVID-19 on Parkinson’s Disease: A Case-Controlled Registry and Questionnaire Study on Clinical Markers and Patients’ Perceptions
Gustav Cedergren Weber, Jonathan Timpka, Filip Bergquist, David Bäckström, Nil Dizdar, Karin Gunnarsson, Dag Nyholm, Per Svenningsson, Per Odin, Gessica Sala
Acta Neurologica Scandinavica.2023; 2023: 1. CrossRef - Remote Assessment of Parkinson’s Disease Patients Amidst the COVID-19 Lockdown in Mexico
Rodrigo León-García, Emmanuel Ortega-Robles, Oscar Arias-Carrión
Brain Sciences.2023; 13(7): 1114. CrossRef - Social and economic development impact of elderly health care products based on design ethics
Na Qi, Xun Zhang, Muhammad Attique Khan, Gaurav Dhiman, Sathishkumar VE
Intelligent Data Analysis.2023; 27: 137. CrossRef - The effects of an individualized smartphone-based exercise program on self-defined motor tasks in Parkinson’s disease: a long-term feasibility study
Lisa Lützow, Isabelle Teckenburg, Veronika Koch, Franz Marxreiter, Jelena Jukic, Sabine Stallforth, Martin Regensburger, Jürgen Winkler, Jochen Klucken, Heiko Gaßner
Journal of Patient-Reported Outcomes.2023;[Epub] CrossRef - Doctor Hope; Calming and Comforting Loneliness in Parkinson’s Disease
Sasivimol Virameteekul, Roongroj Bhidayasiri
Clinical Parkinsonism & Related Disorders.2022; 6: 100131. CrossRef - Neurological toll of COVID-19
Shivam Bhola, Jhillika Trisal, Vikram Thakur, Parneet Kaur, Saurabh Kulshrestha, Shashi Kant Bhatia, Pradeep Kumar
Neurological Sciences.2022; 43(4): 2171. CrossRef - Resilience and Trauma among Patients with Parkinson’s Disease during the COVID-19 Pandemic
Roberto Erro, Sofia Cuoco, Emanuele Nigro, Raffaele Ragone, Paolo Barone
Journal of Movement Disorders.2022; 15(1): 77. CrossRef - Implication of Covid-19 on Neurological Complications with Specific Emphasis
on Alzheimer’s and Parkinson’s Disease
Ankita Sood, Ravi Goyal, Harshdeep Singh, Tapan Behl, Sandeep Arora, Balraj Saini, Rajwinder Kaur
CNS & Neurological Disorders - Drug Targets.2022; 21(3): 235. CrossRef - The use of amantadine in the prevention of progression and treatment of COVID-19 symptoms in patients infected with the SARS-CoV-2 virus (COV-PREVENT): Study rationale and design
Konrad Rejdak, Piotr Fiedor, Robert Bonek, Aleksander Goch, Agnieszka Gala-Błądzińska, Waldemar Chełstowski, Jacek Łukasiak, Sławomir Kiciak, Piotr Dąbrowski, Mateusz Dec, Zbigniew J. Król, Ewa Papuć, Adriana Zasybska, Agnieszka Segiet, Paweł Grieb
Contemporary Clinical Trials.2022; 116: 106755. CrossRef - Neuropathological Aspects of SARS-CoV-2 Infection: Significance for Both Alzheimer’s and Parkinson’s Disease
Jaime Silva, Felipe Patricio, Aleidy Patricio-Martínez, Gerardo Santos-López, Lilia Cedillo, Yousef Tizabi, Ilhuicamina Daniel Limón
Frontiers in Neuroscience.2022;[Epub] CrossRef - Online dance classes during the Covid-19 pandemic: new challenges and teaching strategies for the ‘Dance & Parkinson’s at home’ project
Marcela Dos Santos Delabary, Isadora Loch Sbeghen, Mariana Wolffenbuttel, Djefri Ramon Pereira, Aline Nogueira Haas
Research in Dance Education.2022; : 1. CrossRef - Impact of the COVID-19 Pandemic on Chronic Neurological Disorders: Focus on Patients with Dementia
Maria Antonietta Barbieri, Gianluca Bagnato, Carmelo Ioppolo, Antonio Giovanni Versace, Natasha Irrera
CNS & Neurological Disorders - Drug Targets.2022; 21(10): 1017. CrossRef - Factors Related to Experiencing Pain in Patients Affected by Chronic Inflammatory Rheumatic Disease During the COVID-19 Pandemic
Tfarah El Joumani, Hanan Rkain, Fatima Zahrae Taik, Kenza Hassouni, Redouane Abouqal, Sara Bahloul, Nada Alami, Latifa Tahiri, Najia Hajjaj-Hassouni, Fadoua Allali
The Open Pain Journal.2022;[Epub] CrossRef - A “new” role of amantadines in COVID-19 in patients with Parkinson’s disease: results of own comparative study
Z. A. Zalyalova, S. E. Munasipova, D. M. Khasanova, G. R. Ilyina, Z. G. Khayatova, N. I. Bagdanova
Neurology, Neuropsychiatry, Psychosomatics.2022; 14(6): 40. CrossRef - Attitudes toward telemedicine of patients with Parkinson’s disease during the COVID‐19 pandemic
Kanako Kurihara, Koichi Nagaki, Kotoe Inoue, Sumiko Yamamoto, Takayasu Mishima, Shinsuke Fujioka, Shinji Ouma, Yoshio Tsuboi
Neurology and Clinical Neuroscience.2021; 9(1): 77. CrossRef - Risk of Hospitalization and Death for COVID‐19 in People with Parkinson's Disease or Parkinsonism
Luca Vignatelli, Corrado Zenesini, Laura M.B. Belotti, Elisa Baldin, Giuseppe Bonavina, Giovanna Calandra‐Buonaura, Pietro Cortelli, Carlo Descovich, Giovanni Fabbri, Giulia Giannini, Maria Guarino, Roberta Pantieri, Giuseppe Samoggia, Cesa Scaglione, Sus
Movement Disorders.2021; 36(1): 1. CrossRef - Impact of COVID-19 in the Mental Health in Elderly: Psychological and Biological Updates
Roberta Eduarda Grolli, Maiqueli Eduarda Dama Mingoti, Amanda Gollo Bertollo, Adriana Remião Luzardo, João Quevedo, Gislaine Zilli Réus, Zuleide Maria Ignácio
Molecular Neurobiology.2021; 58(5): 1905. CrossRef - COVID-19: Implications for Sudden Death in Parkinson’s Disease
Ana Claudia Fiorini, Marcelo Cunio Machado Fonseca, Carla Alessandra Scorza, Josef Finsterer, Antônio Márcio Rodrigues, Antônio-Carlos Guimarães de Almeida, Fulvio Alexandre Scorza
Journal of Movement Disorders.2021; 14(1): 78. CrossRef - Impact and Challenges of the COVID-19 Pandemic on Patients Requiring Botulinum Toxin A Treatment
Azalea Tenerife Pajo, Adrian Isidoro Espiritu, Roland Dominic Go Jamora
Journal of Movement Disorders.2021; 14(1): 29. CrossRef - COVID-19 in age-related neurodegenerative diseases: is there a role for vitamin D3 as a possible therapeutic strategy?
Milena de Barros Viana, Bárbara dos Anjos Rosário, Maria de Fátima Santana de Nazaré, Débora Estadella, Daniel Araki Ribeiro, Glauce Socorro de Barros Viana
Reviews in the Neurosciences.2021; 32(2): 235. CrossRef - How COVID-19 will boost remote exercise-based treatment in Parkinson’s disease: a narrative review
Agnes Langer, Lucia Gassner, Anna Flotz, Sebastian Hasenauer, Jakob Gruber, Laurenz Wizany, Rochus Pokan, Walter Maetzler, Heidemarie Zach
npj Parkinson's Disease.2021;[Epub] CrossRef - Neurological Manifestation of SARS-CoV-2 Induced Inflammation and Possible Therapeutic Strategies Against COVID-19
Dipak Kumar, Sadaf Jahan, Andleeb Khan, Arif Jamal Siddiqui, Neeru Singh Redhu, Wahajuddin, Johra Khan, Saeed Banwas, Bader Alshehri, Mohammed Alaidarous
Molecular Neurobiology.2021; 58(7): 3417. CrossRef - Auswirkungen der COVID-19 Pandemie auf die medizinische Versorgung von Patienten mit angeborenen Blutungsneigungen
Martin Olivieri, Susan Halimeh, Cornelia Wermes, Wolf Hassenpflug, Katharina Holstein, Sylvia von Mackensen
Das Gesundheitswesen.2021; 83(04): 282. CrossRef - Parkinsonism hyperpyraexia syndrome in Parkinson's disease patients undergoing deep brain stimulation: An indirect consequence of COVID-19 lockdowns
Onanong Phokaewvarangkul, Sasivimol Virameteekul, Roongroj Bhidayasiri
Parkinsonism & Related Disorders.2021; 87: 39. CrossRef - Clinical Profiles and Mortality of COVID‐19 Inpatients with Parkinson's Disease in Germany
Raphael Scherbaum, Eun Hae Kwon, Daniel Richter, Dirk Bartig, Ralf Gold, Christos Krogias, Lars Tönges
Movement Disorders.2021; 36(5): 1049. CrossRef - Chronic pain experience and health inequities during the COVID-19 pandemic in Canada: qualitative findings from the chronic pain & COVID-19 pan-Canadian study
Lise Dassieu, M. Gabrielle Pagé, Anaïs Lacasse, Maude Laflamme, Vickie Perron, Audrée Janelle-Montcalm, Maria Hudspith, Gregg Moor, Kathryn Sutton, James M Thompson, Manon Choinière
International Journal for Equity in Health.2021;[Epub] CrossRef - The Influence of Coronavirus Disease-2019 (COVID-19) On Parkinson’s Disease: An Updated Systematic Review
Vikash Jaiswal, Danah Alquraish, Zouina Sarfraz, Azza Sarfraz, Shavy Nagpal, Prakriti Singh Shrestha, Dattatreya Mukherjee, Prathima Guntipalli, Diana F. Sánchez Velazco, Arushee Bhatnagar, Saloni Savani, Elmjedina Halilaj, Samir Ruxmohan, Wilson Cueva
Journal of Primary Care & Community Health.2021; 12: 215013272110397. CrossRef - Personalized Medicine in Parkinson’s Disease: New Options for Advanced Treatments
Takayasu Mishima, Shinsuke Fujioka, Takashi Morishita, Tooru Inoue, Yoshio Tsuboi
Journal of Personalized Medicine.2021; 11(7): 650. CrossRef - Parkinson's disease patients may have higher rates of Covid-19 mortality in Iran
Mehri Salari, Masoud Etemadifar, Farzad Ashrafi, Davood Ommi, Zahra Aminzade, Sepand Tehrani Fateh
Parkinsonism & Related Disorders.2021; 89: 90. CrossRef - COVID-19 social distancing: negative effects on people with Parkinson disease and their associations with confidence for self-management
Galit Yogev-Seligmann, Michal Kafri
BMC Neurology.2021;[Epub] CrossRef - Management of Parkinson’s Disease in the COVID-19 Pandemic and Future Perspectives in the Era of Vaccination
Yue Hui Lau, Keng Ming Lau, Norlinah Mohamed Ibrahim
Journal of Movement Disorders.2021; 14(3): 177. CrossRef - Parkinson’s Disease and the COVID-19 Pandemic: A Review Article on the Association between SARS-CoV-2 and α-Synucleinopathy
Smriti Sinha, Swati Mittal, Rupali Roy
Journal of Movement Disorders.2021; 14(3): 184. CrossRef - Impact of COVID‐19 on Patient Care, Training, and Research in Movement Disorders in MDS‐AOS Region
Shweta Prasad, Hrishikesh Kumar, Roongroj Bhidayasiri, Pramod Kumar Pal
Movement Disorders.2021; 36(11): 2457. CrossRef - Secondary Impact of COVID-19 Pandemic on People with Parkinson’s Disease—Results of a Polish Online Survey
Karolina Krzysztoń, Beata Mielańczuk-Lubecka, Jakub Stolarski, Anna Poznańska, Katarzyna Kępczyńska, Agata Zdrowowicz, Izabela Domitrz, Jan Kochanowski
Brain Sciences.2021; 12(1): 26. CrossRef - Dance as Lifeline: Transforming Means for Engagement and Connection in Times of Social Isolation
Maria Portman Kelly, David Leventhal
Health Promotion Practice.2021; 22(1_suppl): 64S. CrossRef - Neurological consultations and diagnoses in a large, dedicated COVID-19 university hospital
Adalberto STUDART-NETO, Bruno Fukelmann GUEDES, Raphael de Luca e TUMA, Antonio Edvan CAMELO FILHO, Gabriel Taricani KUBOTA, Bruno Diógenes IEPSEN, Gabriela Pantaleão MOREIRA, Júlia Chartouni RODRIGUES, Maíra Medeiros Honorato FERRARI, Rafael Bernhart CAR
Arquivos de Neuro-Psiquiatria.2020; 78(8): 494. CrossRef - Potential impact and challenges associated with Parkinson’s disease patient care amidst the COVID-19 global pandemic
Ali Elbeddini, Anthony To, Yasamin Tayefehchamani, Cindy Wen
Journal of Clinical Movement Disorders.2020;[Epub] CrossRef - COVID-19 and possible links with Parkinson’s disease and parkinsonism: from bench to bedside
David Sulzer, Angelo Antonini, Valentina Leta, Anna Nordvig, Richard J. Smeyne, James E. Goldman, Osama Al-Dalahmah, Luigi Zecca, Alessandro Sette, Luigi Bubacco, Olimpia Meucci, Elena Moro, Ashley S. Harms, Yaqian Xu, Stanley Fahn, K. Ray Chaudhuri
npj Parkinson's Disease.2020;[Epub] CrossRef - Impact of Prolonged Lockdown due to COVID-19 in Patients with Parkinson's Disease
Shweta Prasad, VikramVenkappayya Holla, Koti Neeraja, BharathKumar Surisetti, Nitish Kamble, Ravi Yadav, PramodKumar Pal
Neurology India.2020; 68(4): 792. CrossRef - The Effect of the COVID-19 Pandemic on People with Parkinson’s Disease
Ethan G. Brown, Lana M. Chahine, Samuel M. Goldman, Monica Korell, Emerald Mann, Daniel R. Kinel, Vanessa Arnedo, Kenneth L. Marek, Caroline M. Tanner
Journal of Parkinson's Disease.2020; 10(4): 1365. CrossRef - Impact of COVID‐19 pandemic on mental health of patients with inherited bleeding disorders in Germany
Sylvia von Mackensen, Susan Halimeh, Manuela Siebert, Cornelia Wermes, Wolf Hassenpflug, Katharina Holstein, Martin Olivieri
Haemophilia.2020;[Epub] CrossRef - Impact of Coronavirus Disease 2019 Pandemic on Parkinson's Disease: A Cross‐Sectional Survey of 568 Spanish Patients
Diego Santos‐García, Mila Oreiro, Patricia Pérez, Gemma Fanjul, Jose Manuel Paz González, María José Feal Painceiras, Carlos Cores Bartolomé, Lorena Valdés Aymerich, Carlos García Sancho, Maria del Mar Castellanos Rodrigo
Movement Disorders.2020; 35(10): 1712. CrossRef - The Impact of COVID‐19 on Access to Parkinson's Disease Medication
Julia Ling‐Yu Cheong, Zhao Hang Keith Goh, Connie Marras, Caroline M. Tanner, Meike Kasten, Alastair J. Noyce
Movement Disorders.2020; 35(12): 2129. CrossRef - Deep Brain Stimulation Battery Exhaustion during the COVID-19 Pandemic: Crisis within a Crisis
Vikram Venkappayya Holla, Koti Neeraja, Bharath Kumar Surisetti, Shweta Prasad, Nitish Kamble, Dwarakanath Srinivas, Ravi Yadav, Pramod Kumar Pal
Journal of Movement Disorders.2020; 13(3): 218. CrossRef - Clinical Characteristics of Coronavirus Disease 2019 (COVID-19) among Patients at a Movement Disorders Center
Joy Antonelle de Marcaida, Jeffrey Lahrmann, Duarte Machado, Lawrence Bluth, Michelle Dagostine, Maria Moro-de Casillas, Elena Bortan, Sulada Kanchana, Mark Alberts
Geriatrics.2020; 5(3): 54. CrossRef - Update on Neurological Manifestations of SARS-CoV-2
Hisham Valiuddin, Almir Kalajdzic, James Rosati, Kevin Boehm, Dominique Hill
Western Journal of Emergency Medicine.2020;[Epub] CrossRef - Effects of COVID-19 Lockdown on Movement Disorders Patients With Deep Brain Stimulation: A Multicenter Survey
Carla Piano, Francesco Bove, Tommaso Tufo, Isabella Imbimbo, Danilo Genovese, Alessandro Stefani, Massimo Marano, Antonella Peppe, Livia Brusa, Rocco Cerroni, Francesco Motolese, Enrico Di Stasio, Marianna Mazza, Antonio Daniele, Alessandro Olivi, Paolo C
Frontiers in Neurology.2020;[Epub] CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite