Articles
- Page Path
- HOME > J Mov Disord > Volume 13(2); 2020 > Article
-
Original Article
Long-term Effects of Bilateral Subthalamic Deep Brain Stimulation on Postural Instability and Gait Difficulty in Patients with Parkinson’s Disease -
Hae-Won Shin1
 , Mi Sun Kim2
, Mi Sun Kim2 , Sung Reul Kim3
, Sung Reul Kim3 , Sang Ryong Jeon4
, Sang Ryong Jeon4 , Sun Ju Chung2
, Sun Ju Chung2
-
Journal of Movement Disorders 2020;13(2):127-132.
DOI: https://doi.org/10.14802/jmd.19081
Published online: May 29, 2020
1Department of Neurology, Chung-Ang University College of Medicine, Seoul, Korea
2Department of Neurology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
3College of Nursing, Korea University, Seoul, Korea
4Department of Neurological Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding author: Sun Ju Chung, MD, PhD Department of Neurology, Asan Medical Center, Ulsan University College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Korea / Tel: +82-2-3010-3440 / Fax: +82-2-474-4691 / E-mail: sjchung@amc.seoul.kr
Copyright © 2020 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Objective
- The long-term effects of bilateral subthalamic nucleus deep brain stimulation (STN-DBS) on postural instability and gait difficulty (PIGD) in patients with Parkinson’s disease (PD) remain unclear. In this study, we aimed to evaluate the longterm effects of STN-DBS surgery on PIGD symptoms in patients with advanced-stage PD.
-
Methods
- This study included 49 consecutively included patients with PD who underwent bilateral STN-DBS. The Unified Parkinson’s Disease Rating Scale (UPDRS) scores and subscores for PIGD were assessed at baseline and at 1, 3, and 5 years postoperatively. The PIGD subscore was divided into PIGD-motor and PIGD-activities of daily living (ADL) scores according to parts III and II of the UPDRS, respectively.
-
Results
- The PIGD-motor and PIGD-ADL scores at the “medication-off” state improved at 3 and 5 years, respectively. Overall, the UPDRS III and II scores at “medication-off” improved at 5 years. The UPDRS IV score also significantly improved and the levodopa equivalent daily dosage decreased at all follow-ups. Finally, the PIGD-motor score at baseline was able to predict long-term improvement in the PIGD-motor score at the 5-year follow-up.
-
Conclusion
- The STN-DBS has both short- and long-term effects on PIGD, as well as overall motor function, in patients with advanced PD. The degree of PIGD at the preoperative evaluation can be used to predict long-term outcomes after STN-DBS surgery.
- Participants
- We retrospectively reviewed 49 consecutively included patients with PD who underwent bilateral STN-DBS at the Asan Medical Center from 2005 to 2010. All patients with PD underwent DBS surgery according to the institutional guidelines for DBS surgery [13]. All patients showed improvement by more than 30% in the UPDRS III score in a preoperative levodopa test following a suprathreshold dose based on the levodopa equivalent daily dosage (LEDD). STN-DBS surgery was performed in all patients [14]. All patients underwent STN-DBS on the more severely affected side first, followed by STN-DBS on the contralateral side three months later. Stereotaxic surgery using the Leksell G frame was performed with the patients under local anesthesia after the overnight withdrawal of any anti-Parkinsonian medications. We located the STN using MRI, electrophysiological recordings, and stimulation. Quadripolar electrodes (model 3389, Medtronic Inc., Minneapolis, MN, USA) were implanted and connected to a subcutaneous programmable pulse generator (Soletra TM; Medtronic). All patients underwent brain CT scans postoperatively to detect surgical complications [14]. The electrical parameters (voltage, pulse width, and frequency) and medication dosages were adjusted at every visit. The stimulation frequency and pulse width were set to be 130 Hz and 60 μs, respectively. The amplitude of the stimulation voltage and stimulation contacts were adjusted so that there was optimal clinical improvement at each visit.
- This study was approved by the institutional Review Board of Asan Medical Center (2014-0332). The board waived the requirement for informed consent from patient because this study was the retrospective and observational study.
- Clinical evaluations
- The patients’ demographic and clinical characteristics were recorded. The patients were assessed at baseline and postoperatively at 1, 3, and 5 years. All patients’ clinical symptoms were assessed using the UPDRS during the “medication-off ” and “medication-on” states preoperatively and during the “medication-off and stimulation-on” and “medication-on and stimulation-on” states postoperatively. All anti-Parkinsonian medications were discontinued for 12 h to determine UPDRS scores during the “medication-off” condition. The “medication-on” condition was considered the period after patients took 1.5 times their usual dose of levodopa. The specific PIGD-motor subscore was defined as the sum of the subscores for arising from chair (UPDRS part III item 27), posture (UPDRS part III item 28), gait (UPDRS III item 29), and postural instability (UPDRS III item 30). The PIGD-activities of daily living (PIGD-ADL) score was defined as the sum of the subscores for episodes of freezing (UPDRS II item 13), falls (UPDRS II item 14), and walking ability (UPDRS II item 15) during ADL.
- Statistical analyses
- The PIGD scores from the same patient were assumed to be correlated and were considered repeated measures. Therefore, a linear mixed model with the Dunnett correction for multiple comparisons was performed to examine the changes in the mean scores for PIGD across time points. A multivariate stepwise regression analysis was performed to predict the longterm effect of STN-DBS on the percent change in the PIGDmotor and ADL scores from baseline to the 5-year follow-up. All data were analyzed using SAS 9.4 (SAS Institute, Cary, NC, USA) with a two-sided test, and the level of statistical significance was set to be p < 0.05.
MATERIALS & METHODS
- Baseline characteristics
- The mean age of the study subjects at the time of STN-DBS surgery was 59.5 years (range 38–76 years). The mean durations of PD and levodopa treatment at the operation were 15.9 ± 4.2 years and 10.2 ± 3.2 years, respectively (Table 1). All 49 patients were evaluated preoperatively and at 1 year; 37 and 21 patients were evaluated at 3 and 5 years, respectively. Seven patients died within 5 years of the treatment, and 20 patients were evaluated at all four time points. There were no significant differences in any of the clinical characteristics between the patients who completed all the follow-ups and those who were lost to follow-up.
- Clinical outcomes
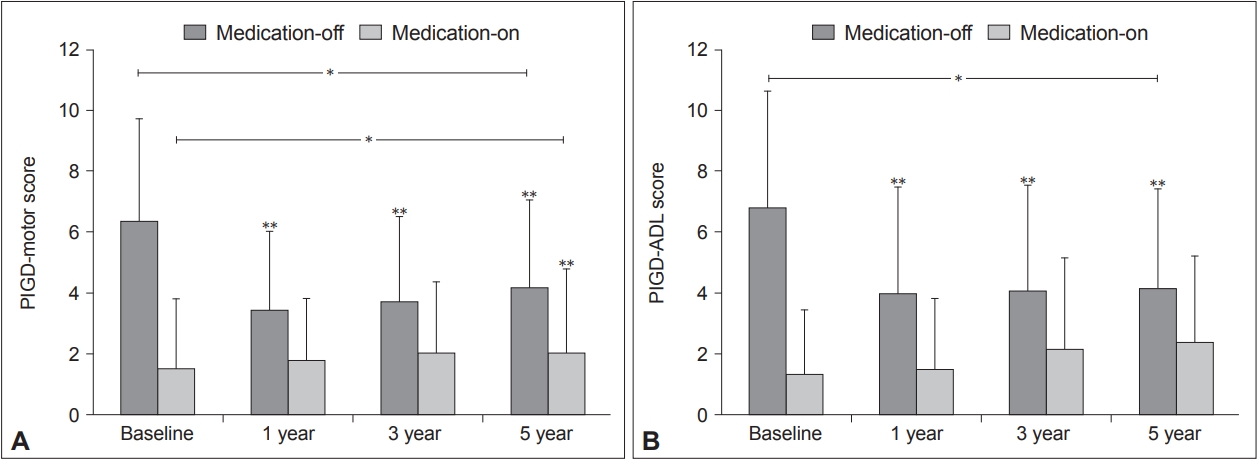
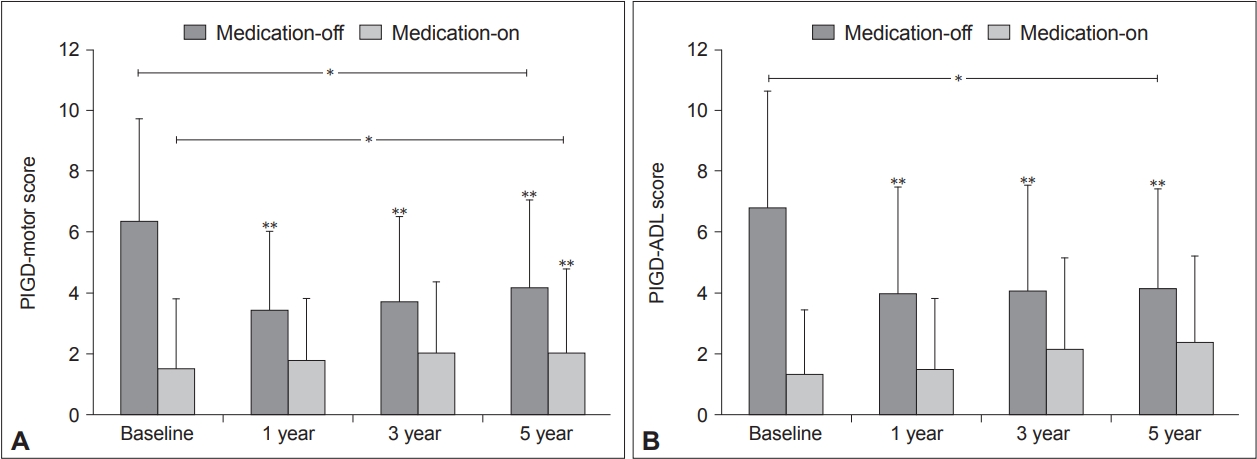
- A linear mixed model for repeated measures showed that the mean PIGD-motor and PIGD-ADL scores in the “medication-off ” state improved with time (p < 0.0001) (Figure 1). Both the PIGD-motor and ADL scores in the “medication-off” state showed significant improvement at postoperative 1, 3, and 5 years compared to baseline (p < 0.0001, p < 0.0001, and p = 0.0165, respectively, for the PIGD-motor score; p < 0.0001, p < 0.0001, and p = 0.0004, respectively, for the PIGD-ADL score) (Figure 1). The mean PIGD-motor scores in the “medication-on” state worsened with time (p = 0.0012) (Figure 1A), whereas the PIGD-ADL scores were comparable at all time points (p = 0.1337) (Figure 1B). When the PIGD-motor scores were separated into postural instability (items 27 and 30), gait (item 29), and postural scores (item 28), the postural score in the “medication-on” state appeared to worsen with time, but the other scores did not appear to differ across the follow-ups.
- The total UPDRS III and II scores in the “medication-off” state showed significant improvement with time (p < 0.0001) (Table 2). However, the UPDRS III and II scores in the “medication-on” state worsened with time (p < 0.0001, p = 0.0004, respectively) (Table 2). The UPDRS IV scores improved over the follow-up period (p < 0.0001). A separate analysis of the dyskinesia part of the UPDRS IV demonstrated that the scores decreased with time (p < 0.0001), and the scores at postoperative 5 years were comparable with the baseline scores (p = 0.1771) (Table 2). In addition, the LEDD significantly decreased with time postoperatively p < 0.0001) (Table 2).
- Factor related to improvement in PIGD
- There were significant correlations between the amount of improvement in the PIGD-motor score at 5 years and that in the PIGD-motor (rho = 0.551, p = 0.01) and levodopa responsiveness scores in the UPDRS III (rho = -0.496. p = 0.022) at baseline. Improvement in the PIGD motor at 5 years was not correlated with any other variables, including the preoperative UPDRS II and III scores, patient age, disease duration, or changes in the levodopa-equivalent dosage. A multivariable stepwise regression analysis revealed that the preoperative PIGD-motor score remained predictive of the long-term effect of STN-DBS on the PIGD-motor score (R2 = 0.424, p = 0.007). The preoperative levodopa responsiveness score in the UPDRS III was excluded from the regression analysis. Improvement in the PIGD-ADL score at 5 years was not significantly correlated with any variable.
RESULTS
- In this study, we evaluated the long-term effects of STN-DBS on PIGD and clinical factors of PIGD outcomes after STN-DBS in patients with PD. The main findings from this study were as follows: 1) STN-DBS improved the PIGD-motor and ADL scores in the “medication-off” state over 5 years in patients with advanced PD. 2) The PIGD-motor scores at baseline predicted the long-term improvement in the PIGD-motor scores at 5 years after STN-DBS surgery. 3) STN-DBS improved the UPDRS II and III scores in the “medication-off” state over 5 years. 4) The PIGD-motor score in the “medication-on” state worsened over time, along with the UPDRS II and III scores.
- These results suggest that STN-DBS may have long-term effects on PIGD symptoms in the “medication-off” state regarding both ADL and motor function. The effects of STN-DBS on PIGD are in line with the long-term improvement observed in the overall motor symptoms and independence in ADL of the patients in the “medication-off” state. A recent meta-analysis showed that bilateral STN-DBS improves gait performance and reduces the severity of freezing of gait (FOG), as measured by item 29 of the UPDRS III (gait) and item 14 of the UPDRS II (freezing when walking), respectively, for as many as 4 years [15]. We used items 27 (arising from chair), 30 (postural instability), 28 (posture) and 29 (gait) from the UPDRS III to assess postural instability. From the UPDRS II, items 13 (falling unrelated to freezing), 14 (freezing when walking), and 15 (walking) were used to calculate the PIGD-ADL score. Improvement in the PIGD-ADL score, as well as the PIGD-motor score, was observed, supporting the hypothesis that STN-DBS is effective in improving mobility in advanced patients with PD. Although the overall motor symptoms and independence in ADL in patients with PD have been evaluated using the UPDRS in most studies, the clinical parameters used to evaluate PIGD symptoms have not been well validated [16-22]. Various clinical and physiological tools have been suggested for measuring PIGD symptoms, including postural instability and FOG.23 Moreover, wearable devices that measure patients’ motor function, including gait function, have been increasingly utilized in research [24]. However, the lack of standardized clinical assessment tools may result in inconsistent findings regarding the effects of STN-DBS on PIGD symptoms in patients with PD [23]. Although the PIGD-motor and ADL scores extracted from the UPDRS scores have not been validated, it may be worthwhile to evaluate the PIGD symptoms because the PIGD-motor score addresses the severity of gait dysfunction (item 29), posture (item 28), and postural instability (items 27 and 30). Moreover, the PIGD-ADL scores address the frequency at which FOG (item 14), gait difficulty (item 15), and falls (item 13) occur in everyday life. Objective tools for measuring PIGD parameters, such as gait parameters, are lacking, which remains a limitation of this study. Therefore, additional research needs to be conducted to establish a tool that can accurately assess PIGD in patients with PD.
- In our study, the PIGD-motor scores at baseline predicted the long-term outcomes of patients, i.e., the PIGD-motor scores at 5 years after STN-DBS surgery. Regarding predictive factors of the effect of STN-DBS on PIGD in patients with PD, the levodopa responsiveness score in the UPDRS III scale at baseline was suggested to be the most significant predictor of gait performance and FOG in a previous meta-analysis study [15]. Another study showed that the preoperative PIGD scores and dosages of medication differed between patients with and without PIGD symptoms that progressed within 8 years after surgery [11]. Because the consensus guidelines for DBS surgery in patients with PD do not specify selection criteria in terms of PIGD [3], our results may provide useful evidence for the selection of patients for DBS surgery and may aid in planning the long-term treatment of patients with PD who undergo STN-DBS surgery.
- In our study, the PIGD-motor scores in the “medication-on” state worsened over the follow-up period. Worsening of the scores was observed at postoperative 3 and 5 years, whereas no changes were observed at postoperative 1 year. Each item representing postural instability (items 27 and 30), gait (item 29), and posture (item 28) showed that the postural score only worsened with time in the “medication-on” state, whereas the scores for gait and postural instability did not change across the follow-ups. Deficits in posture can be complicated by other factors, including levodopa-induced axial dystonia, and other musculoskeletal problems can possibly affect the postural score. Thus, we suggest that the worsening of the PIGD-motor symptoms in the “medication-on” state can be related to levodopa-induced axial dystonia. The PIGD-ADL score in the “medication-on” state did not change during the follow-up period. The UPDRS III and II scores representing overall motor function worsened in the “medication-on” state after STN-DBS surgery. It is believed that STN-DBS acts on the dopaminergic motor system in a similar manner as dopaminergic medication [2,25]. Accordingly, the additive benefits of dopaminergic medications and STN-DBS surgery have not been expected, and motor function in the “medication-on and stimulation- on” state has not been found to change postoperatively.2 In the patients in our study, the LEDD decreased by 21%, 24% and 25% at 1, 3 and 5 years, respectively, compared to baseline. The patients in our study had relatively long durations of the disease (mean 15.9 years), so the reduction in the LEDD might affect the PIGD-motor and UPDRS III and II scores more in these patients than in patients who have shorter disease durations [26]. Indeed, the UPDRS IV scores, particularly those concerning dyskinesia, were significantly lower by approximately 50% at all follow-ups than at baseline. This result suggests that the reduction in the LEDD in the patients in our study might not have been sufficient to induce a fully “medication-on” state, resulting in the worsening of the PIGD-motor and UPDRS III and II scores in the “medication-on” state.
- Our findings are consistent with the results of previous studies, which reported that bilateral STN-DBS has long-term favorable effects on PIGD or axial symptoms, in addition to long-term beneficial effects on patients’ overall motor symptoms [9-12,15,27]. Contrary to our study, other studies have reported that bilateral STN-DBS improves PIGD or axial levodopa-responsive symptoms for as long as 1 year but that these effects diminish in the long term [4,5,7,8]. Although it is well known that dopaminergic treatment positively affects appendicular parkinsonian symptoms, PIGD has been found to be resistant to dopaminergic medication [28]. In addition to dopaminergic neuronal degeneration, cholinergic neuronal loss in the pedunculopontine nucleus has been suggested to play a major role in PIGD in patients with PD [29]. As PD progresses, neurodegeneration beyond the dopaminergic neurons becomes prominent, which can explain why levodopa is less effective in reducing PIGD symptoms in advanced-stage PD patients than in early-stage PD patients [30]. The mechanism of STN-DBS involves the dopaminergic motor system [2], and patients with levodopa-resistant PIGD symptoms may show limited benefits from STN-DBS surgery. All the PD patients in our study showed improvement in the PIGD-motor scores after consuming levodopa in the preoperative examination. Considering the mechanism of action of STN-DBS, the preoperative levodopa responsiveness of PIGD symptoms in our study population can be attributable to the favorable outcomes of STN-DBS surgery in the PD patients in our study. A study with a follow-up period of more than 5 years showed that STN-DBS improves PIGD for 5 years, at which point the effect declines [10]. Follow-up periods of more than 5 years are necessary to determine the outcomes of the patients in our study.
- This study has some notable limitations. First, all patients who underwent STN-DBS surgery did not undergo long-term evaluations. Patients who had relatively better mobility were more likely to participate in the evaluation than those with poorer mobility, which may have affected the proportion of favorable outcomes after STN-DBS surgery regarding PIGD that was observed in our study. The lack of differences in the clinical characteristics before surgery between groups who did and did not complete the follow-ups can account for this limitation. Second, PIGD was assessed using only UPDRS subscores. A specific assessment tool for PIGD symptoms and objective gait analysis with a detailed computational program may be necessary for future studies. Last, we did not analyze PIGD symptoms separately according to their levodopa responsiveness; instead, we grouped all symptoms into one parameter using the UPDRS score. Therefore, this study can be considered a preliminary study, and the effects of STN-DBS on levodopa-responsive or nonresponsive PIGD symptoms should be assessed in the future.
DISCUSSION
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Author Contributions
Conceptualization: Hae-Won Shin and Sun Ju Chung. Data curation: Mi Sun Kim and Sung Reul Kim. Funding acquisition: Sun Ju Chung. Investigation: Mi Sun Kim, Sung Reul Kim, Sang Ryong Jeon and Sun Ju Chung. Methodology: Sang Ryong Jeon and Sun Ju Chung. Project administration: Sang Ryong Jeon and Sun Ju Chung. Writing—original draft: Hae-Won Shin. Writing—review & editing: Hae-Won Shin and Sun Ju Chung.
Notes
- This study was supported by a grant from the Korea Healthcare Technology R & D Project, Ministry of Health & Welfare, Republic of Korea (HI19C0256). We thank Ji Sung Lee, PhD, Department of Clinical Epidemiology and Biostatistics, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea, who assisted in the statistical analysis.
Acknowledgments
|
Baseline (n = 49) |
1 year (n = 49) |
3 years (n = 37) |
5 years (n = 21) |
p value* | ||||
|---|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | p value | Mean ± SD | p value | Mean ± SD | p value | ||
| Medication-off | ||||||||
| UPDRS II | 22.49 ± 7.68 | 13.92 ± 5.97 | <0.0010 | 13.92 ± 7.73 | <0.0010 | 15.10 ± 6.87 | 0.0050 | <0.0001 |
| UPDRS III | 39.70 ± 10.65 | 21.62 ± 9.81 | <0.0010 | 23.49 ± 10.24 | <0.0010 | 19.43 ± 13.12 | <0.0010 | <0.0001 |
| Medication-on | ||||||||
| UPDRS II | 5.23 ± 4.49 | 7.65 ± 4.91 | 0.0120 | 7.76 ± 5.19 | 0.0128 | 8.57 ± 6.04 | 0.0010 | 0.0004 |
| UPDRS III | 11.07 ± 7.63 | 16.12 ± 9.39 | 0.0001 | 14.81 ± 8.05 | 0.0014 | 14.90 ± 9.34 | 0.0002 | <0.0001 |
| UPDRS IV | 8.71 ± 3.27 | 4.08 ± 2.89 | <0.0001 | 2.35 ± 2.02 | <0.0001 | 5.33 ± 3.38 | <0.0001 | <0.0001 |
| UPDRS IV-dyskinesia | 3.37 ± 2.56 | 1.63 ± 1.76 | <0.0001 | 1.84 ± 1.83 | 0.0018 | 2.52 ± 2.02 | 0.1771 | <0.0001 |
| LEDD | 1181.77 ± 420.82 | 931.77 ± 418.00 | 0.0002 | 897.06 ± 459.88 | 0.0001 | 851.79 ± 357.53 | 0.0009 | <0.0001 |
All p-values were calculated by a linear mixed model using a random intercept model for repeated measures.
* p-value for the mean equality of the score across the time points. p-value for the mean difference in the score from baseline to each of the time points using Dunnett’s test for multiple comparisons.
UPDRS: Unified Parkinson’s Disease Rating Scale, LEDD: levodopa equivalent daily dosage, SD: standard deviation.
- 1. Okun MS. Deep-brain stimulation for Parkinson’s disease. N Engl J Med 2012;367:1529–1538.ArticlePubMed
- 2. Benabid AL, Chabardes S, Mitrofanis J, Pollak P. Deep brain stimulation of the subthalamic nucleus for the treatment of Parkinson’s disease. Lancet Neurol 2009;8:67–81.ArticlePubMed
- 3. Bronstein JM, Tagliati M, Alterman RL, Lozano AM, Volkmann J, Stefani A, et al. Deep brain stimulation for Parkinson disease: an expert consensus and review of key issues. Arch Neurol 2011;68:165.ArticlePubMed
- 4. Cossu G, Pau M. Subthalamic nucleus stimulation and gait in Parkinson’s Disease: a not always fruitful relationship. Gait Posture 2017;52:205–210.ArticlePubMed
- 5. Fasano A, Aquino CC, Krauss JK, Honey CR, Bloem BR. Axial disability and deep brain stimulation in patients with Parkinson disease. Nat Rev Neurol 2015;11:98–110.ArticlePubMedPDF
- 6. Daneault JF, Duval C, Barbat-Artigas S, Aubertin-Leheudre M, Jodoin N, Panisset M, et al. Subthalamic stimulation improves motor function but not home and neighborhood mobility. Mov Disord 2014;29:1816–1819.ArticlePubMed
- 7. St George RJ, Nutt JG, Burchiel KJ, Horak FB. A meta-regression of the long-term effects of deep brain stimulation on balance and gait in PD. Neurology 2010;75:1292–1299.ArticlePubMedPMC
- 8. Van Nuenen BF, Esselink RA, Munneke M, Speelman JD, van Laar T, Bloem BR. Postoperative gait deterioration after bilateral subthalamic nucleus stimulation in Parkinson’s disease. Mov Disord 2008;23:2404–2406.ArticlePubMed
- 9. McNeely ME, Earhart GM. Medication and subthalamic nucleus deep brain stimulation similarly improve balance and complex gait in Parkinson disease. Parkinsonism Relat Disord 2013;19:86–91.ArticlePubMed
- 10. Castrioto A, Lozano AM, Poon YY, Lang AE, Fallis M, Moro E. Ten-year outcome of subthalamic stimulation in Parkinson disease: a blinded evaluation. Arch Neurol 2011;68:1550–1556.ArticlePubMed
- 11. Fasano A, Romito LM, Daniele A, Piano C, Zinno M, Bentivoglio AR, et al. Motor and cognitive outcome in patients with Parkinson’s disease 8 years after subthalamic implants. Brain 2010;133:2664–2676.ArticlePubMedPDF
- 12. Schüpbach WM, Chastan N, Welter ML, Houeto JL, Mesnage V, Bonnet AM, et al. Stimulation of the subthalamic nucleus in Parkinson’s disease: a 5 year follow up. J Neurol Neurosurg Psychiatry 2005;76:1640–1644.ArticlePubMedPMC
- 13. Ryu HS, Kim MS, You S, Kim MJ, Kim YJ, Kim J, et al. Comparison of pallidal and subthalamic deep brain stimulation in Parkinson’s disease: therapeutic and adverse effects. J Mov Disord 2017;10:80–86.ArticlePubMedPMCPDF
- 14. Chung SJ, Jeon SR, Kim SR, Sung YH, Lee MC. Bilateral effects of unilateral subthalamic nucleus deep brain stimulation in advanced Parkinson’s disease. Eur Neurol 2006;56:127–132.ArticlePubMed
- 15. Schlenstedt C, Shalash A, Muthuraman M, Falk D, Witt K, Deuschl G. Effect of high-frequency subthalamic neurostimulation on gait and freezing of gait in Parkinson’s disease: a systematic review and metaanalysis. Eur J Neurol 2017;24:18–26.ArticlePubMed
- 16. Roper JA, Kang N, Ben J, Cauraugh JH, Okun MS, Hass CJ. Deep brain stimulation improves gait velocity in Parkinson’s disease: a systematic review and meta-analysis. J Neurol 2016;263:1195–1203.ArticlePubMedPDF
- 17. Collomb-Clerc A, Welter ML. Effects of deep brain stimulation on balance and gait in patients with Parkinson’s disease: a systematic neurophysiological review. Neurophysiol Clin 2015;45:371–388.ArticlePubMed
- 18. Mera TO, Filipkowski DE, Riley DE, Whitney CM, Walter BL, Gunzler SA, et al. Quantitative analysis of gait and balance response to deep brain stimulation in Parkinson’s disease. Gait Posture 2013;38:109–114.ArticlePubMed
- 19. Weaver FM, Follett K, Stern M, Hur K, Harris C, Marks WJ Jr, et al. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. JAMA 2009;301:63–73.ArticlePubMedPMC
- 20. Johnsen EL, Mogensen PH, Sunde NA, Østergaard K. Improved asymmetry of gait in Parkinson’s disease with DBS: gait and postural instability in Parkinson’s disease treated with bilateral deep brain stimulation in the subthalamic nucleus. Mov Disord 2009;24:590–597.ArticlePubMed
- 21. Ferraye MU, Debû B, Fraix V, Xie-Brustolin J, Chabardès S, Krack P, et al. Effects of subthalamic nucleus stimulation and levodopa on freezing of gait in Parkinson disease. Neurology 2008;70(16 Pt 2):1431–1437.ArticlePubMed
- 22. Maurer C, Mergner T, Xie J, Faist M, Pollak P, Lücking CH. Effect of chronic bilateral subthalamic nucleus (STN) stimulation on postural control in Parkinson’s disease. Brain 2003;126(Pt 5):1146–1163.ArticlePubMedPDF
- 23. Bloem BR, Marinus J, Almeida Q, Dibble L, Nieuwboer A, Post B, et al. Measurement instruments to assess posture, gait, and balance in Parkinson’s disease: Critique and recommendations. Mov Disord 2016;31:1342–1355.ArticlePubMed
- 24. Hubble RP, Naughton GA, Silburn PA, Cole MH. Wearable sensor use for assessing standing balance and walking stability in people with Parkinson’s disease: a systematic review. PLoS One 2015;10:e0123705.ArticlePubMedPMC
- 25. Pötter-Nerger M, Volkmann J. Deep brain stimulation for gait and postural symptoms in Parkinson’s disease. Mov Disord 2013;28:1609–1615.ArticlePubMed
- 26. Schuepbach WM, Rau J, Knudsen K, Volkmann J, Krack P, Timmermann L, et al. Neurostimulation for Parkinson’s disease with early motor complications. N Engl J Med 2013;368:610–622.ArticlePubMed
- 27. Krack P, Batir A, Van Blercom N, Chabardes S, Fraix V, Ardouin C, et al. Five-year follow-up of bilateral stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N Engl J Med 2003;349:1925–1934.ArticlePubMed
- 28. Marsden CD. Problems with long-term levodopa therapy for Parkinson’s disease. Clin Neuropharmacol 1994;17 Suppl 2:S32–S44.PubMed
- 29. Tsang EW, Hamani C, Moro E, Mazzella F, Poon YY, Lozano AM, et al. Involvement of the human pedunculopontine nucleus region in voluntary movements. Neurology 2010;75:950–959.ArticlePubMedPMC
- 30. Bonnet AM, Loria Y, Saint-Hilaire MH, Lhermitte F, Agid Y. Does longterm aggravation of Parkinson’s disease result from nondopaminergic lesions? Neurology 1987;37:1539–1542.ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Effects of subthalamic nucleus deep brain stimulation using different frequency programming paradigms on axial symptoms in advanced Parkinson’s disease
Yifeng Cheng, Guangrui Zhao, Lei Chen, Deqiu Cui, Chunjuan Wang, Keke Feng, Shaoya Yin
Acta Neurochirurgica.2024;[Epub] CrossRef - Long-term motor outcomes of deep brain stimulation of the globus pallidus interna in Parkinson's disease patients: Five-year follow-up
Yun Su Hwang, Sungyang Jo, Seung Hyun Lee, Nayoung Kim, Mi-Sun Kim, Sang Ryong Jeon, Sun Ju Chung
Journal of the Neurological Sciences.2023; 444: 120484. CrossRef - Smartwatch gait coordination index: New measure for human gait utilizing smartwatch sensor
Sumin Han, Rob Paul
Medicine.2023; 102(12): e33267. CrossRef - WITHDRAWN: Laterality and frequency settings of subthalamic nucleus DBS for Parkinson's disease: A systematic review and network meta-analysis
Rajiv Dharnipragada, Lalitha S. Denduluri, Anant Naik, Mario Bertogliat, Matthew Awad, Salman Ikramuddin, Michael C. Park
Parkinsonism & Related Disorders.2023; : 105455. CrossRef - Frequency settings of subthalamic nucleus DBS for Parkinson's disease: A systematic review and network meta-analysis
Rajiv Dharnipragada, Lalitha S. Denduluri, Anant Naik, Mario Bertogliat, Matthew Awad, Salman Ikramuddin, Michael C. Park
Parkinsonism & Related Disorders.2023; 116: 105809. CrossRef - Unlocking potential: low frequency subthalamic nucleus stimulation enhances executive function in Parkinson’s disease patients with postural instability/gait disturbance
Guofan Qin, Hutao Xie, Lin Shi, Baotian Zhao, Yifei Gan, Zixiao Yin, Yichen Xu, Xin Zhang, Yaojing Chen, Yin Jiang, Quan Zhang, Jianguo Zhang
Frontiers in Neuroscience.2023;[Epub] CrossRef - The Role of Microelectrode Recording in Deep Brain Stimulation Surgery for Parkinson’s Disease: A Systematic Review and Meta-Analysis
R. Saman Vinke, Martin Geerlings, Ashok K. Selvaraj, Dejan Georgiev, Bastiaan R. Bloem, Rianne A.J. Esselink, Ronald H.M.A. Bartels
Journal of Parkinson's Disease.2022; 12(7): 2059. CrossRef - Axial impairment and falls in Parkinson’s disease: 15 years of subthalamic deep brain stimulation
Alessandro Zampogna, Francesco Cavallieri, Francesco Bove, Antonio Suppa, Anna Castrioto, Sara Meoni, Pierre Pélissier, Emmanuelle Schmitt, Amélie Bichon, Eugénie Lhommée, Andrea Kistner, Stephan Chabardès, Eric Seigneuret, Valerie Fraix, Elena Moro
npj Parkinson's Disease.2022;[Epub] CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite