Articles
- Page Path
- HOME > J Mov Disord > Volume 13(2); 2020 > Article
-
Case Report
Treatment of Acute Delirium in a Patient with Parkinson’s Disease by Transfer to the Intensive Care Unit and Administration of Dexmedetomidine -
Morgan Lombardo1,2
 , Amanda DiPiazza1,2
, Amanda DiPiazza1,2 , Kelly Rippey3
, Kelly Rippey3 , Naomi Lubarr4
, Naomi Lubarr4 , Elana Clar2,4
, Elana Clar2,4 , Hooman Azmi1,2
, Hooman Azmi1,2
-
Journal of Movement Disorders 2020;13(2):159-162.
DOI: https://doi.org/10.14802/jmd.20005
Published online: May 29, 2020
1Department of Neurosurgery, Hackensack University Medical Center, Hackensack, NJ, USA
2North Jersey Brain and Spine Center, Hackensack, NJ, USA
3Division of Trauma and Critical Care, Department of Surgery, Hackensack University Medical Center, Hackensack, NJ, USA
4Department of Neurology, Hackensack University Medical Center, Hackensack, NJ, USA
- Corresponding author: Hooman Azmi, MD Department of Neurosurgery, Hackensack University Medical Center, 30 Prospect Ave, Hackensack, NJ 07601, USA / Tel: +1-201342-2550 / Fax: +1-2013427171 / E-mail: hoomanazmi@gmail.com
Copyright © 2020 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- The treatment of delirium or psychosis in patients with Parkinson’s disease (PD) can be complicated by the limited number of pharmacological agents that can be used in this population. Typical and atypical antipsychotics are contraindicated, as they can worsen motor symptoms. The treatment of acute delirium is even more complicated in the hospital setting, as many medications deemed safer in this population are only available in oral form. We present a case of acute delirium in a patient with PD, likely precipitated by a polypharmacy interaction of new medications, that was successfully managed by transferring the patient to the intensive care unit and administering dexmedetomidine for 72 hours.
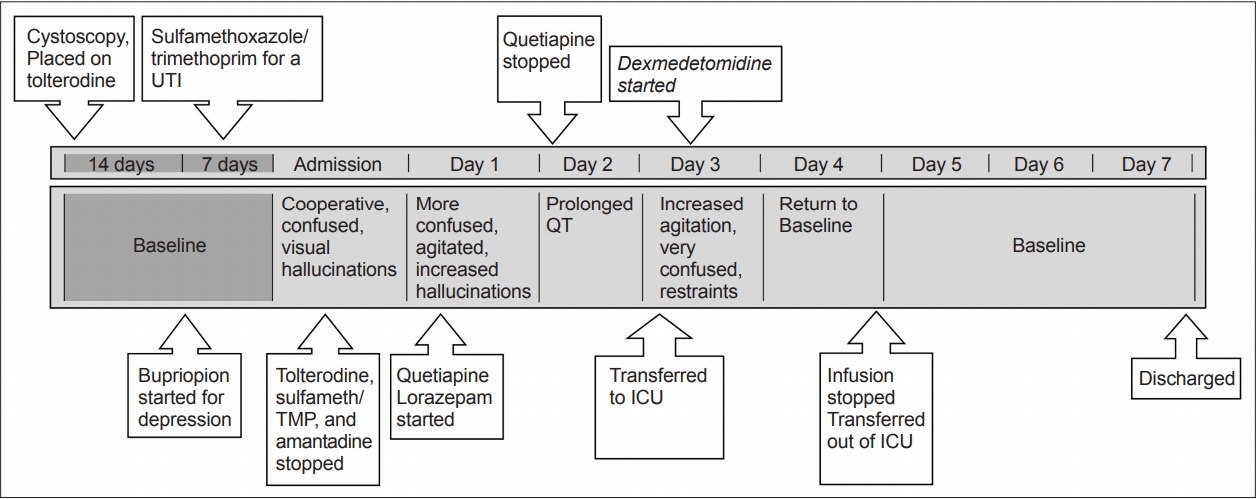
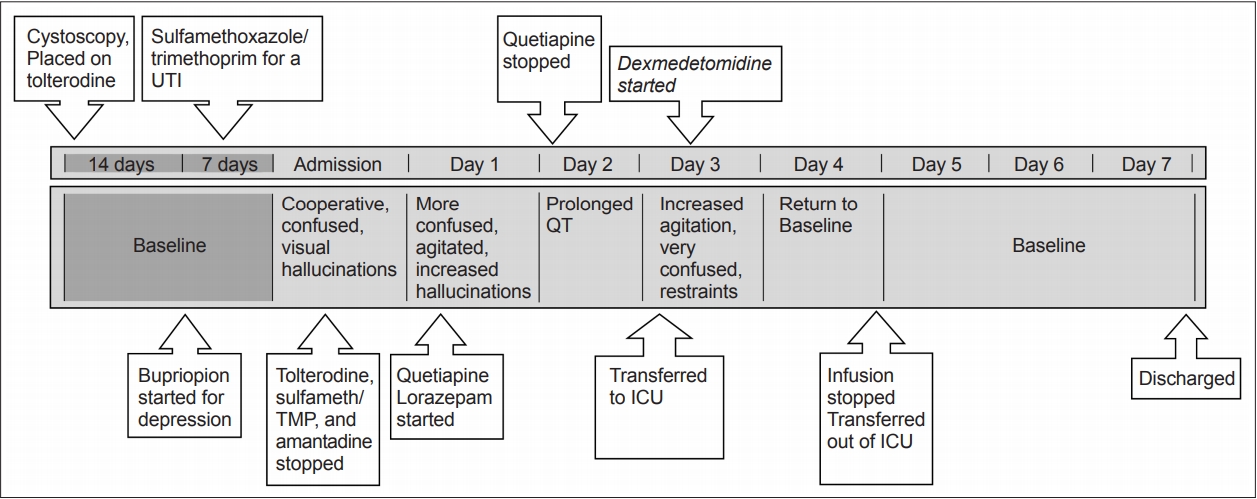
- A 71-year-old high-functioning male patient with a >15-year history of PD who had undergone deep brain stimulation (DBS) in the previous year presented with a one-week history of worsening urinary incontinence, unsteady gait and altered mental status progressively worsening in the last two days prior to his arrival at the emergency room. Prior to this decline, he had undergone a cystoscopy procedure for an overactive bladder and was placed on tolterodine and was also started on sulfamethoxazole/ trimethoprim for a urinary tract infection. Just before this episode, the patient’s psychiatrist had added bupropion for his long-standing depression. His other medications included carbidopta/ levodopa-entacapone 200 twice daily, rasagiline 1 mg daily, clonazepam 0.25 mg nightly, amantadine 100 mg twice daily, venlafaxine 75 mg daily, and midodrine 5 mg daily.
- On presentation to the emergency room, he was awake but confused and had visual hallucinations. Imaging of the brain and blood work were unremarkable. Tolterodine, sulfamethoxazole/trimethoprim, and amantadine were stopped, and the dose of bupropion was reduced. Increased agitation and worsening hallucinations over the next day prompted the initiation of quetiapine and lorazepam. Quetiapine was discontinued because of a prolonged QT. The patient’s agitation and confusion worsened, requiring restraints. He was transferred to the ICU for the initiation of a dexmedetomidine infusion. On arrival to the ICU, he was awake but disoriented to self, place and time. He was confused and impulsive and had poor safety awareness and poor attention and concentration. His right pupil was 3 mm and reactive and his left was 5 mm and sluggish (premorbid visual loss). His Glasgow coma score (GCS) was 13 (4E, 4V: able to speak but confused, 5M). His Riker score was 6 (very agitated). Shortly after arrival, dexmedetomidine infusion was started at a rate of 0.2 mcg/kg/hr and was titrated at a rate of 0.1 mcg/kg/hr every 20–30 minutes with a goal of a Riker score of 4 (calm and cooperative). Within three hours after the infusion was started, he was still confused but following some commands, still with impulsivity, poor safety awareness, and poor attention and concentration. His GCS had improved to 14 (E4, V4, M6), and his Riker score was 5 (agitated). The next morning, his exam improved to full orientation with no more concern for impulsivity, safety or poor attention or concentration. His GCS was 14 (E4, V4, M6), and his Riker Score was 4 (calm and cooperative). After twenty-four hours of dexmedetomidine infusion, the patient’s mentation returned to baseline. He was observed outside of the ICU, and quetiapine was restarted. He was subsequently discharged to a rehabilitation facility. Figure 1 illustrates this timeline.
CASE REPORT
- Management of delirium for patients with PD in the hospital can be challenging, as recognition of delirium may be delayed by presumptions regarding patients’ baseline cognitive status, and the treatment can be hampered by limitations in availability of medications [1]. Overall management of delirium in the PD population is aimed at addressing causative issues, stopping or reducing medications that can exacerbate delirium, and administering medications to reduce the symptoms [1]. In hospitals with acutely deteriorating patients, there may not be the opportunity to systematically address these concerns, and rapid pharmacological intervention may be necessary. While delirium in the hospital can be significantly more prevalent among patients with PD than those without [1], there is a paucity in the literature regarding pharmacological treatment options for delirium in the PD population.
- The formulary for acute delirium in the hospital is not expansive. Neuroleptics, anxiolytics such as benzodiazepines, and sedatives such as propofol all may play some role traditionally; however, there are some potential drawbacks with each. Typical, and some atypical, antipsychotics can worsen PD motor symptoms, and their use in the PD population has been shown to increase length of stay (LOS) [2]. Additionally, there are no studies that have assessed the use of neuroleptics for delirium in patients with PD [1]. Nevertheless, because of the lower risks of motor side effects, quetiapine, clozapine, and pimavanserin, all serotonin 2A receptor antagonists, have been deemed safe with varied efficacy in PD patients with psychosis [3-5]. Their role in the treatment of acute delirium in the hospital has limitations, mainly that these medications are only orally available, and their latency to effect could be long.
- Quetiapine can be a reasonable first-line option for the treatment of delirium in the hospital for patients who are able to take oral medication. Side effects include potential hypotension and some sedation. In addition, it is not recommended for use in patients with QT interval increases, as it may increase the likelihood of developing torsade de points [6].
- Clozapine is the only atypical antipsychotic recommended for patients with PD [4]. It is not, however, commonly used in the acute setting, as it is not available in parenteral forms. While there is evidence for its use for psychosis in the PD population, evidence of its efficacy for delirium does not exist [1]. Additionally, because of the small but real myelosuppressive risk, this medication is heavily regulated and presents transition in care barriers as often the hospital care team is not the outpatient care team and there is often a reluctance to start a medication that may have difficulties in continuation outside the hospital.
- Pimavanserin is the most recent serotonin 2A receptor antagonist to reach the market. It is the only medication approved for psychosis in the PD population [5]. It does carry a black box warning of increased mortality for elderly individuals with dementia; however, the more common side effects are nausea and other gastrointestinal issues and extremity swelling. Nevertheless, it is generally well tolerated; however, its use in acutely delirious patients is limited by parenteral unavailability and its latency to effect duration.
- Parenterally delivered medication is important for treating an acutely deteriorating patient. Propofol, a GABA receptor agonist, is fast acting and has sedative and hypnotic properties. While its quick action makes it an option for prompt treatment, its respiratory depressive properties will require intubation for use. More importantly, it does not directly address delirium. Side effects include potential hypotension. As such, the guidelines for the treatment of delirium in the ICU do not recommend its use for this purpose [6].
- Benzodiazepines, which act by increasing the inhibitory effects of GABA receptors on neuronal activity, are available in oral and parenteral forms. They can be used in severely agitated and psychotic patients in the hospital setting because of their anxiolytic properties. However, there is no evidence of their efficacy in the treatment of delirium, especially in the PD population. In the elderly population, benzodiazepines can increase the risk of gait instability, resulting in falls and fractures, outcomes that would further debilitate a PD patient with delirium [7]. This class of medications can also cause respiratory depression and hypotension, and the guidelines for the treatment of delirium in the ICU recommend against their first-line use [1,6].
- Dexmedetomidine is a highly selective alpha-2 receptor agonist that has analgesic, sedative and anxiolytic effects without respiratory depression and is administered intravenously. In the PD population, dexmedetomidine has been used during DBS surgeries, demonstrating sedation without respiratory depression in this patient group in the perioperative context [8].
- Dexmedetomidine, unlike other sedatives, does not adversely affect respiratory efforts and is therefore crucial for mechanically ventilated patients [8,9]. In addition, it is also approved for sedation for non-intubated patients. While not specific to PD, it has been used in the ICU setting for the treatment of delirium, and a systematic review and meta-analysis of the use of dexmedetomidine for the treatment of agitation in the ICU concluded that it reduced LOS in the unit and resulted in earlier extubations [9].
- There are no studies that have examined the use of dexmedetomidine specifically for patients with delirium and PD. However, two randomized trials compared benzodiazepines to dexmedetomidine and found a significant difference in the ability of dexmedetomidine to treat delirium. As such, the guidelines for the treatment of delirium in ICU patients recommend the use of dexmedetomidine [6]. Dexmedetomidine is not associated with respiratory depression, and as such, it is the only sedative approved for use in patients who are not intubated. The side effects of this drug are potential hypotension and bradycardia, as well as reduction in oropharyngeal muscle tone, so continuous monitoring in the ICU setting is important [6]. Another detractor from this medication is lack of availability in an oral form that can be continued once the patient has been stabilized.
- Currently, there are no definitive or widely accepted guidelines for the transfer of a patient to the ICU. The taskforce for the development of guidelines for the Society of Critical Care’s recommendation is that each hospital develops policies based on their needs, capacities and expertise. To optimize resources while improving outcomes, ICU admissions should be based on patient needs, expertise, capacity, and prognosis, among other things. Our decision to transfer the patient to the ICU was based on judgment, taking into consideration each of these issues and focusing on the goal of providing the best and safest outcome for the patient [10].
- Our treatment algorithm for this patient was dictated by his clinical course. On arrival at the hospital where he had periods of lucidity without agitation, we began by discontinuing potential offending agents. When he did not improve and worsened to a more hyperactive delirious state, an oral benzodiazepine and the atypical antipsychotic quetiapine were initiated. Due to a lack of improvement, progression to a very agitated state and demonstration of QT prolongation, quetiapine was discontinued. The state of severe agitation and florid delirium required prompt action in an attempt to stabilize the patient with the use of intravenous medications. Without any meaningful improvement on oral benzodiazepines, dexmedetomidine was chosen as the first agent. We chose this medication based on its rapidity of action, lack of respiratory depression effects, and our experience in its use intraoperatively in the PD population for DBS procedures. The patient was transferred to the ICU for the purpose of starting the infusion, as dexmedetomidine cannot be administered outside of an intensively monitored setting. Our patient’s course corrected fairly quickly, and we were able to stop the infusion and transfer him out of the ICU within a few days. While there is evidence for the use of dexmedetomidine for the treatment of delirium, the literature does not exist for the PD population, and there are no guidelines regarding transfer to the ICU. While other medication regimens could have reasonably been initiated, including intravenous benzodiazepines, in this case, our choice was informed by our experience and the need for urgent intervention in a rapidly declining patient.
- There clearly needs to be more studies on the efficacy of dexmedetomidine for the treatment of delirium in the PD population, and no conclusion can be made from a single patient. Nevertheless, this report offers a potential option for practitioners faced with a PD patient with delirium in the hospital.
DISCUSSION
-
Ethics
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Author Contributions
Conceptualization: Hooman Azmi. Data curation: Morgan Lombardo. Supervision: Hooman Azmi. Writing—original draft: Morgan Lombardo. Writing—review & editing: all authors.
Notes
- None.
Acknowledgments
- 1. Ebersbach G, Ip CW, Klebe S, Koschel J, Lorenzl S, Schrader C, et al. Management of delirium in Parkinson’s disease. J Neural Transm (Vienna) 2019;126:905–912.ArticlePubMedPDF
- 2. Martinez-Ramirez D, Giugni JC, Little CS, Chapman JP, Ahmed B, Monari E, et al. Missing dosages and neuroleptic usage may prolong length of stay in hospitalized Parkinson’s disease patients. PLoS One 2015;10:e0124356.ArticlePubMedPMC
- 3. Shotbolt P, Samuel M, David A. Quetiapine in the treatment of psychosis in Parkinson’s disease. Ther Adv Neurol Disord 2010;3:339–350.ArticlePubMedPMC
- 4. Parkinson Study Group. Low-dose clozapine for the treatment of druginduced psychosis in Parkinson’s disease. N Engl J Med 1999;340:757–763.ArticlePubMed
- 5. Cummings J, Isaacson S, Mills R, Williams H, Chi-Burris K, Corbett A, et al. Pimavanserin for patients with Parkinson’s disease psychosis: a randomised, placebo-controlled phase 3 trial. Lancet 2014;383:533–540.ArticlePubMed
- 6. Barr J, Fraser GL, Puntillo K, Ely EW, Gélinas C, Dasta JF, et al. Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med 2013;41:263–306.ArticlePubMed
- 7. Tannenbaum C. Inappropriate benzodiazepine use in elderly patients and its reduction. J Psychiatry Neurosci 2015;40:E27–E28.ArticlePubMedPMCPDF
- 8. Sassi M, Zekaj E, Grotta A, Pollini A, Pellanda A, Borroni M, et al. Safety in the use of dexmedetomidine (precedex) for deep brain stimulation surgery: our experience in 23 randomized patients. Neuromodulation 2013;16:401–406.ArticlePubMed
- 9. Ng KT, Shubash CJ, Chong JS. The effect of dexmedetomidine on delirium and agitation in patients in intensive care: systematic review and meta-analysis with trial sequential analysis. Anaesthesia 2019;74:380–392.ArticlePubMed
- 10. Nates JL, Nunnally M, Kleinpell R, Blosser S, Goldner J, Birriel B, et al. ICU admission, discharge, and triage guidelines: a framework to enhance clinical operations, development of institutional policies, and further research. Crit Care Med 2016;44:1553–1602.ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Toxin Induced Parkinsonism and Hospitalization Related Adverse Outcome Mitigation for Parkinson’s Disease: A Comprehensive Review
Kenneth R. Dalton, Charles J. Kidd, Nawaz Hack
Journal of Clinical Medicine.2023; 12(3): 1074. CrossRef - Fountain of youth—Targeting autophagy in aging
Lea Danics, Anna Anoir Abbas, Balázs Kis, Karolina Pircs
Frontiers in Aging Neuroscience.2023;[Epub] CrossRef - Effect of dexmedetomidine on postoperative delirium in patients undergoing brain tumour resections: study protocol of a randomised controlled trial
Dexiang Wang, Ruowen Li, Shu Li, Juan Wang, Min Zeng, Jia Dong, Xiaoyuan Liu, Nan Lin, Yuming Peng
BMJ Open.2021; 11(11): e051584. CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite