Articles
- Page Path
- HOME > J Mov Disord > Volume 13(3); 2020 > Article
-
Original Article
Parkinson Anxiety Scale: A Validation Study for the Brazilian Population -
Renilson Moraes-Ferreira1,2
 , Wilson Mateus Gomes da Costa Alves1,2
, Wilson Mateus Gomes da Costa Alves1,2 , Maysa Alves Rodrigues Brandao-Rangel1,2
, Maysa Alves Rodrigues Brandao-Rangel1,2 , Odilon Abrahin3
, Odilon Abrahin3 , Clebson Pantoja Pimentel3
, Clebson Pantoja Pimentel3 , Evitom Correa-Sousa3
, Evitom Correa-Sousa3 , Rodolfo Paula Vieira1,2,4,5
, Rodolfo Paula Vieira1,2,4,5 , Erik Artur Cortinhas-Alves3,6
, Erik Artur Cortinhas-Alves3,6
-
Journal of Movement Disorders 2020;13(3):199-204.
DOI: https://doi.org/10.14802/jmd.20031
Published online: July 28, 2020
1Department of Sciences of Human Movement and Rehabilitation, Federal University of São Paulo (UNIFESP), Sao Paulo, Brazil
2Brazilian Institute of Teaching and Research in Pulmonary and Exercise Immunology (IBEPIPE), Sao Jose dos Campos, Brazil
3Pará State University, Belém, Brazil
4Anhembi Morumbi University, School of Medicine, Sao Jose dos Campos, Brazil
5Post-graduate Program in Bioengineering and in Biomedical Engineering Brazil University, Sao Paulo, Brazil
6Exercise Biochemistry Laboratory, Center for Biological Sciences of Health III, State University of Pará, Belém, Brazil
- Corresponding author: Renilson Moraes-Ferreira, MSc Department of Sciences of Human Movement and Rehabilitation, Federal University of São Paulo (UNIFESP), Rua Eliza Costa Santos 160, Sao Jose dos Campos, Sao Paulo 12245-380, Brazil / Tel: +55-91-99294-1817 / Fax: +55-91-99294-1817 / E-mail: renilsonmoraesferreira@gmail.com
Copyright © 2020 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 6,707 Views
- 159 Download
- 1 Crossref
ABSTRACT
-
Objective
- The Parkinson Anxiety Scale (PAS) was developed to measure the severity of anxiety symptoms in patients with Parkinson’s disease (PD), and it has not yet been adapted and validated in Portuguese. Thus, this study evaluated the reliability and validity of a translated and adapted version of the PAS for the Brazilian population of PD patients.
-
Methods
- The Parkinson Anxiety Scale – Brazilian Version (PAS-BV) was completed by 55 patients with PD. The reliability (test-retest reliability, interrater reliability and internal consistency) and construct validity of the PAS-BV were assessed by comparing it with the Beck Anxiety Inventory (BAI), the Parkinson’s Disease Fatigue Scale (PFS) and the Unified Parkinson Disease Rating Scale (UPDRS) part III.
-
Results
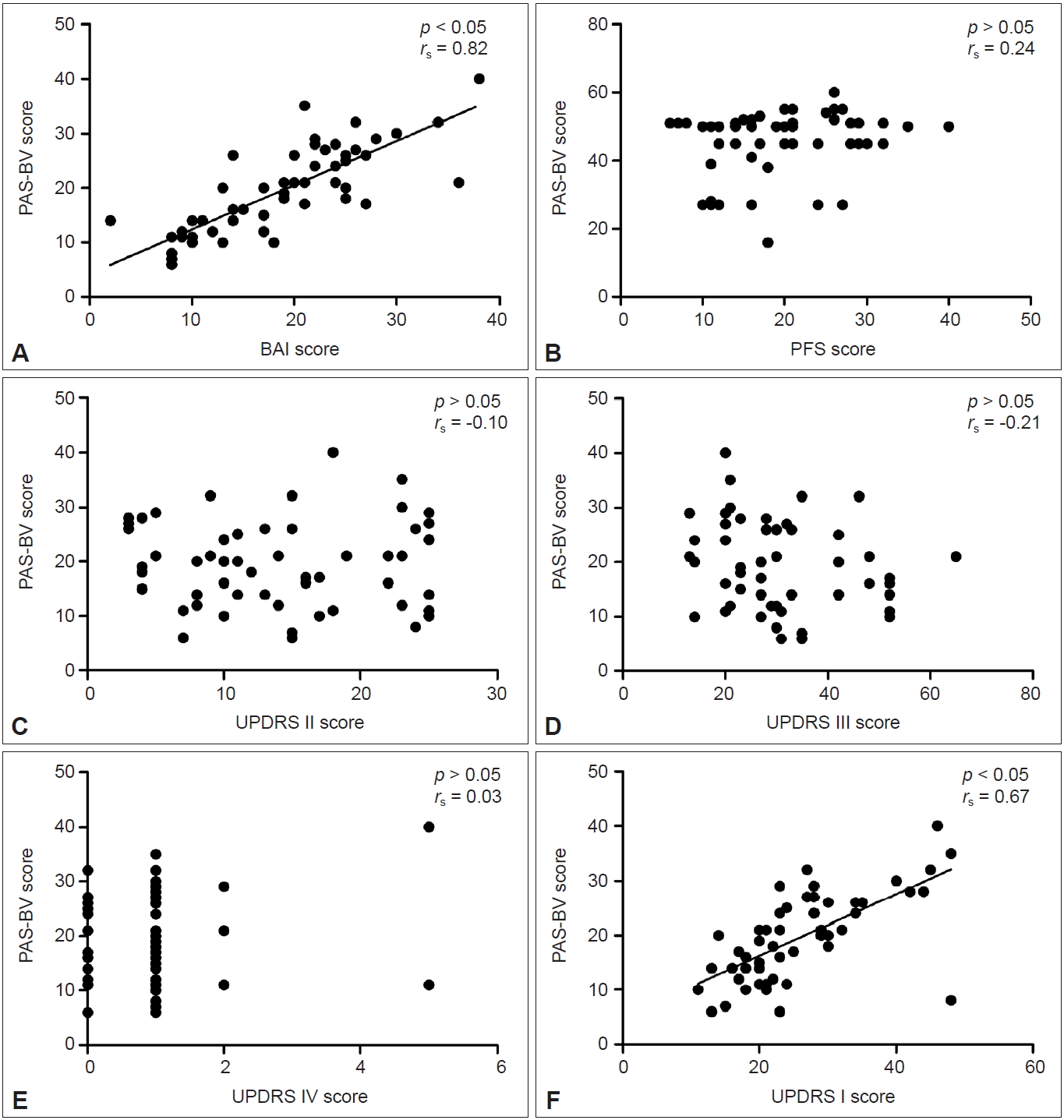
- Patients with PD had an average age of 64.51 ± 9.20 years and had PD for an average of 6.98 ± 5.02 years. The reliability of the PAS-BV was 0.83, and the intraclass correlation coefficient (ICC) (retest-test) was 0.88. The scale presented good convergent validity with the BAI (rs = 0.82, p < 0.05). It also presented good divergent validity with the PFS (rs = 0.24, p > 0.05) and the UPDRS part II (rs = -0.10, p > 0.05), part III (rs = -0.21, p > 0.05), and part IV (rs = 0.03, p > 0.05), as indicated by the absence of significant correlations. However, there was a significant correlation between the PAS-BV and part I of the UPDRS (rs = 0.67, p < 0.05).
-
Conclusion
- The PAS-BV presents substantial reliability and validity for patients with PD without dementia.
- Design and study participants
- The study was a cross-sectional and one-point-in-time evaluation with a retest study. The sample consisted of 55 patients with PD without dementia who were further invited and enrolled in the physical training project for patients with PD from the Pará State University. The patients had a mean age of 64.51 ± 9.20 years old and had been diagnosed with PD for an average of 6.98 ± 5.02 years. All evaluations were performed before patients participated in any intervention (e.g., physical exercise). All participants had previously been seen by a neurologist who specialized in the evaluation of PD; this neurologist performed the assessment of cognitive status and anxiety levels, and only patients who met the following inclusion criteria were enrolled in the study: (i) stage 1–3 on the Hoehn and Yahr scale (H&Y), (ii) stable use of medication, (iii) signed the free and informed consent form, (iv) was not performing any kind of regular (≥ 2x/week) physical training, and (v) no severe cognitive impairments (Mini-Mental State Examination score ≥ 24) [12]. This research was approved by the ethics and research committee of the Pará State University (82885818.6.0000.5167), according to the rules of resolution 466/2012 of the National Council for Research Involving Human Beings and according to the Helsinki Declaration.
- Translation and adaptation of the PAS
- The PAS was developed to specifically evaluate the severity of anxiety symptoms in PD patients [10]. It can be divided into three subscales (persistent anxiety, episodic anxiety, and avoidance behavior). The scale consists of 12 questions, each with five response options (0, never; 1, rarely; 2, sometimes; 3, frequent; and 4, always).
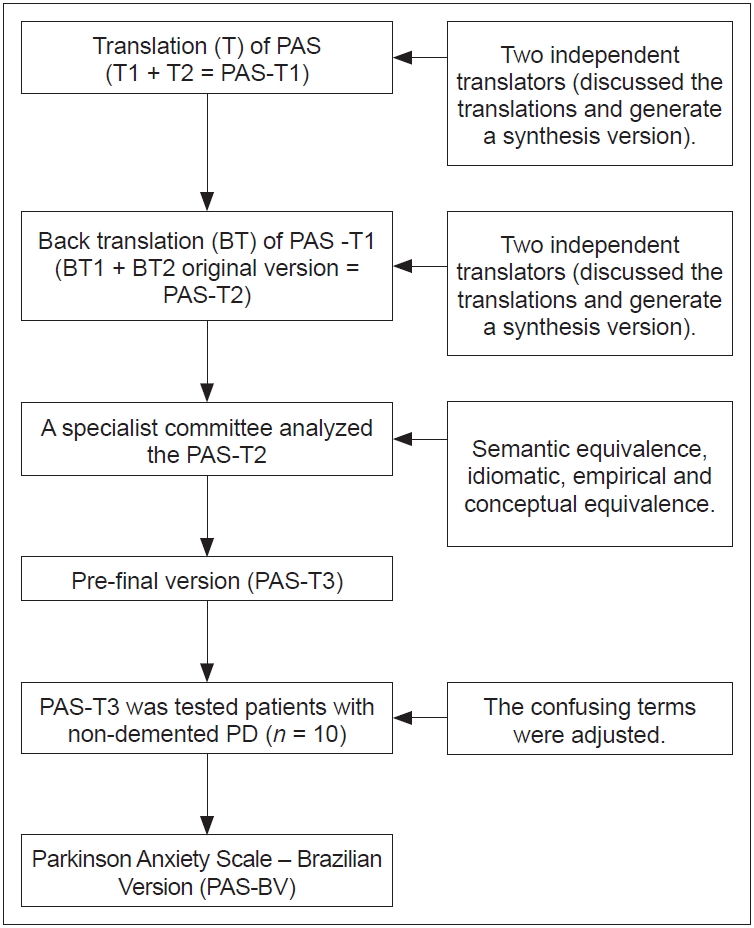
- The PAS was translated into Brazilian Portuguese by following the recommendations in the literature [13,14]. Four independent translators took part in the translation process. In addition, two translators who performed the back translation were unaware of the original version of the scale. The expert committee was composed of researchers who had experience in the application of interventions among patients with PD (Figure 1).
- Parkinson Anxiety Scale – Brazilian Version
- The observer-rated Parkinson Anxiety Scale – Brazilian Version (PAS-BV) preserved all the main characteristics of the original version. The PAS-BV is composed of 12 questions, each of which can receive scores from zero (0) to four (4) for a total maximum score of 48 points. The PAS-BV can be divided into the persistent anxiety subscale (PA; 1st to 5th questions), which measures generalized anxiety disorders; the episodic anxiety subscale (EA; 6th to 9th questions), which assesses panic disorder; and the avoidance behavior subscale (AB; 10th to 12th questions), which assesses anxiety symptoms of agoraphobia and social phobia.
- Assessments
- All patients were analyzed when they were in the “on” state of medication (1–1.5 hours after taking medication). Demographic data were collected, including gender, age, duration of the disease and treatments. We assessed the medication doses used by individuals with PD. Furthermore, the following evaluations were used: the H&Y scale [15], the Unified Parkinson’s Disease Rating Scale (UPDRS) [16], the Parkinson’s Disease Fatigue Scale (PFS) [17] and the Beck Anxiety Inventory (BAI) [18].
- The evaluations were made by two trained and experienced evaluators (Evaluator 1 and Evaluator 2) and at two time points (1st day and 2nd day) separated by an interval of 7 days. On the 1st day, all evaluations were performed (the PFS, UPDRS, BAI, and PAS-BV) by only one evaluator (EV1), and the second evaluator (EV2) only administered the PAS-BV. On the 2nd day, only one evaluator (EV1) administered the PAS-BV. There was a 15-minute interval between the PAS-BV evaluations made by the evaluators (EV1 and EV2) on the first day.
- Statistical analysis
- Data analysis was performed using SPSS version 21.0 software (IBM Corp., Armonk, NY, USA). Descriptive statistics were used as needed. The Shapiro-Wilk test was used to verify the normality of the data. Nonparametric tests were used for nonnormally distributed data. Internal consistency was evaluated by Cronbach’s alpha (a value ≥ 0.70 was considered acceptable), corrected item-total correlation (a value ≥ 0.30 was considered acceptable), and corrected item-total correlation (a total item correlation value ≥ 0.40 was considered acceptable). The test-retest analysis was performed calculating the intraclass correlation coefficients (ICCs). Interrater reliability was assessed by the Kappa coefficient. Convergent validity (PAS-BV vs. BAI) and divergent validity [PAS-BV vs. PFS and UPDRS (part I, II, III, and IV)] were evaluated by Spearman’s correlation test. The Bland–Altman plot between the 1st and 2nd day and the evaluators (EV1 and EV2) was used to verify the agreement. We performed post hoc analysis with the sample to assess the power of the analysis and found a power value of 0.74. Statistical significance was established at p < 0.05.
MATERIALS & METHODS
- The study consisted of 55 patients with PD. The clinical and demographic characteristics of the patients are presented in Table 1. No patients reporting using any antidepressants or anxiolytic medication. The PAS-BV score was not significantly different from the BAI score (Z = 1.38, p = 0.08). In the test-retest analysis and when examining the scores between the evaluators, the data were not different (Z = 1.20, p = 0.11) and (Z = 1.41, p = 0.07), respectively. The data from the subscales measured by the two evaluators were also not different [(PA: Z = 1.31, p = 0.09), (EA: Z = 1.33, p = 0.09), (AB: Z = 0.52, p = 0.29)].
- The Cronbach’s alpha of the PAS-BV was 0.83. We analyzed reliability for each subscale: persistent anxiety had a Cronbach’s alpha of 0.73, episodic anxiety had a Cronbach’s alpha of 0.71, and avoidance behavior had a Cronbach’s alpha of 0.70. In addition, we used the Cronbach’s alpha coefficients to determine how much each item impacts the total PAS-BV score (Table 2).
- The ICC between the first and second evaluation (test-retest reliability) was 0.88 (0.78–0.93; p < 0.05); the Kappa coefficient, representing the interrater reliability, was substantial (0.65); and the Bland-Altman plots presented good test-retest agreement and good agreement between the two evaluators for the PAS-BV (Figure 2).
- Acceptable convergent construct validity was indicated by the significant correlation between the BAI and the PAS-BV scores (Figure 3A: rs = 0.82, p < 0.05). Good divergent construct validity was indicated by the nonsignificant correlations between the PAS-BV and the PFS (Figure 3B: rs = 0.24, p > 0.05) and the UPDRS part II (Figure 3C: rs = -0.10, p > 0.05), part III (Figure 3D: rs = -0.21, p > 0.05), and part IV (Figure 3E: rs = 0.03, p > 0.05). However, there was a significant correlation between the PAS-BV and the UPDRS part I (Figure 3F: rs = 0.67, p < 0.05).
RESULTS
- The main finding of this study was that the observer-rated PAS-BV presented acceptable reliability and validity for patients with PD without dementia. Reliability refers to stable and consistent measurements for multiple attempts over time [14,19]. Validity refers to the ability to measure the variable (anxiety) with minimal error [14,19].
- When an instrument is developed to monitor the symptoms of a given disease, it is essential to translate and adapt the instrument to populations in other countries to account for cultural variations [14]. In the case of the PAS, the author highlighted the importance of translation and adaptation to other languages and populations with PD [11].
- The present study was the first to explore the psychometric properties of a translated version of the PAS-BV for patients with PD without dementia and with a disease staging between 1–3 on the H&Y scale. The reliability of the PAS-BV (0.83) and its subscales – persistent anxiety (0.73), episodic anxiety (0.71) and avoidance behaviors (0.70) – were acceptable in the present study. Additionally, we identified an acceptable correlation between the items within the instrument and their high reliability. In addition, acceptable test-retest reliability and interrater reliability were observed.
- Excellent and substantial reliability indicates an instrument’s ability to reproduce the results of a condition in space and time consistently, ensuring the accuracy, stability and coherence of the instrument [20]. PAS-BV retained the reliability aspects of the original version [20]. In the visual analysis of the Bland–Altman plot, the test-retest results indicate that the instrument has a risk of bias close to zero, with positive agreement [20].
- In addition to good reliability, it is essential to have validity, which is defined as the interpretation or specific purpose of the instrument [19,21,22]. The PAS-BV presented good convergent and divergent construct validity, as it was correlated with the BAI, which also evaluates anxiety symptoms. In addition, the PAS-BV was not associated with the PFS or the UPDRS. These data indicate that the instrument measures exactly what they propose to measure [15,22] and it not significantly influenced by other symptoms of PD [5,8,23].
- Our result shows a significant correlation between the scores of the PAS-BV and the UPDRS part I; previous studies have shown that anxiety symptoms occur in parallel with other non-motor symptoms of PD [6,7,24], which are measured by part I of the UPDRS [16].
- The study sought to nullify the observational and environmental systematic error that could underestimate or overestimate the reliability and validity of the instrument [25,26]. In this sense, the evaluators followed the same instructions as the instrument, with one not following the other performing the evaluations. In the test-retest procedure, an interval of 7 days was used so that the results were not influenced by the memory of the participants or by the changing symptoms of anxiety [14]. In all evaluations, only the volunteer and the evaluator were in the same neutral environment to avoid constraints and biases.
- Although the study presented significant results for clinical and research applications, some limitations need to be highlighted. The study included participants with PD in stages between 1–3 on the H&Y scale, making it impossible to extrapolate the findings to more severe stages of the disease and to the non-PD population. Therefore, our results cannot be generalized to patients without these characteristics. In addition, the power calculation of the sample size was slightly below 80%, but we believe that its value (79%) did not compromise the findings of the study. Therefore, we encourage future studies with larger sample sizes to further evaluate the applicability of the PAS-BV.
- In conclusion, the observer-rated PAS-BV presented acceptable reliability and validity for patients with PD without dementia with staging of 1–3 according to the H&Y scale.
DISCUSSION
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Author Contributions
Conceptualization: Renilson Moraes-Ferreira, Wilson Mateus Gomes da Costa Alves, Erik Artur Cortinhas-Alves. Data curation: Renilson Moraes-Ferreira, Erik Artur Cortinhas-Alves. Formal analysis: all authors. Investigation: Renilson Moraes-Ferreira, Wilson Mateus Gomes da Costa Alves, Clebson Pantoja Pimentel, Erik Artur Cortinhas-Alves. Methodology: all authors. Project administration: Renilson Moraes-Ferreira, Erik Artur Cortinhas-Alves. Resources: Renilson Moraes-Ferreira, Wilson Mateus Gomes da Costa Alves, Erik Artur Cortinhas-Alves. Software: Erik Artur Cortinhas-Alves. Supervision: Renilson Moraes-Ferreira, Wilson Mateus Gomes da Costa Alves, Erik Artur Cortinhas-Alves. Validation: Renilson Moraes-Ferreira, Wilson Mateus Gomes da Costa Alves, Erik Artur Cortinhas-Alves. Visualization: all authors. Writing—original draft: Renilson Moraes-Ferreira, Wilson Mateus Gomes da Costa Alves, Odilon Abrahin, Erik Artur Cortinhas-Alves. Writing—review & editing: all authors.
Notes
- We are grateful to Albert F.G. Leentjens for the permission to use the Parkinson Anxiety Scale. We also thank all the patients and translators who participated in this study.
Acknowledgments

| Variable | Values |
|---|---|
| Age (years) | 64.51 ± 9.20 |
| Duration of disease (years) | 6.98 ± 5.02 |
| Levodopa + carbidopa (mg) | 5 |
| Levodopa + decarboxylase inhibitor (mg) | 100 |
| Decarboxylase inhibitor (mg) | 25 |
| Dopamine agonist (mg) | 0.18 |
| Anticholinergic (mg) | 2 |
| Gender (F/M) | 20/35 |
| Hoehn and Yahr stage | 1–3 |
| BAI* | 19 (6–12) |
| PFS* | 20 (14–26) |
| UPDRS* | |
| UPDRS part I | 23 (20–30) |
| UPDRS part II | 14 (8–22) |
| UPDRS part III | 28 (21–35) |
| UPDRS part IV | 1 (1–1) |
| PAS-BV* | |
| PAS-BV day 1 | 20 (5–12) |
| PAS-BV day 2 | 17 (5–12) |
| PAS-BV interviewer 2 | 16 (12–26) |
| Education levels | |
| Primary school (%) | 49.09 |
| High school (%) | 40.00 |
| University (%) | 10.91 |
* median value of 25th and 75th.
Values are presented as mean ± standard deviation unless otherwise indicated. F: female, M: male, BAI: Beck Anxiety Inventory, PFS: Parkinson’s Disease Fatigue Scale, UPDRS: Unified Parkinson’s Disease Rating Scale, PAS-BV: Parkinson Anxiety Scale – Brazilian Version.
- 1. World Health Organization (WHO). Depression and other common mental disorders: global health estimates [Internet]; Geneva: WHO; c2017 [accessed on 2019 Jun 18]. Available at: https://www.who.int/mental_health/management/depression/prevalence_global_health_estimates/en/.
- 2. American Parkinson Disease Association. Anxiety in Parkinson’s Disease [Internet]; Staten Island (NY): American Parkinson Disease Association; c2019 [accessed on 2019 Apr 7]. Available at: https://www.apdaparkinson.org/article/anxiety-parkinsons-disease/.
- 3. Parkinson’s Foundation. Anxiety and Parkinson’s disease [Internet]; New York (NY): Parkinson’s Foundation; c2019 [accessed on 2019 Apr 7]. Available at: https://parkinson.org/pd-library/fact-sheets/Anxiety-and-Parkinsons?_ga=2.111763396.1753339149.1560943139-1430956530.1560943139.
- 4. Broen MP, Narayen NE, Kuijf ML, Dissanayaka NN, Leentjens AF. Prevalence of anxiety in Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 2016;31:1125–1133.ArticlePubMed
- 5. Kano O, Ikeda K, Cridebring D, Takazawa T, Yoshii Y, Iwasaki Y. Neurobiology of depression and anxiety in Parkinson’s disease. Parkinsons Dis 2011;2011:143547.ArticlePubMedPMCPDF
- 6. Pringsheim T, Jette N, Frolkis A, Steeves TD. The prevalence of Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 2014;29:1583–1590.ArticlePubMed
- 7. Nègre-Pagès L, Grandjean H, Lapeyre-Mestre M, Montastruc JL, Fourrier A, Lépine JP, et al. Anxious and depressive symptoms in Parkinson’s disease: the French cross-sectional DoPaMiP study. Mov Disord 2010;25:157–166.ArticlePubMed
- 8. Dissanayaka NN, Torbey E, Pachana NA. Anxiety rating scales in Parkinson’s disease: a critical review updating recent literature. Int Psychogeriatr 2015;27:1777–1784.ArticlePubMed
- 9. Leentjens AF, Dujardin K, Pontone GM, Starkstein SE, Weintraub D, Martinez-Martin P. The Parkinson Anxiety Scale (PAS): development and validation of a new anxiety scale. Mov Disord 2014;29:1035–1043.ArticlePubMed
- 10. Leentjens AF, Dujardin K, Marsh L, Richard IH, Starkstein SE, Martinez-Martin P. Anxiety rating scales in Parkinson’s disease: a validation study of the Hamilton anxiety rating scale, the Beck anxiety inventory and the hospital anxiety and depression scale. Mov Disord 2011;26:407–415.ArticlePubMed
- 11. Leentjens AF, Dujardin K, Marsh L, Martinez-Martin P, Richard IH, Starkstein SE, et al. Anxiety rating scales in Parkinson’s disease: critique and recommendations. Mov Disord 2008;23:2015–2025.ArticlePubMed
- 12. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189–198.ArticlePubMed
- 13. Sousa VD, Rojjanasrirat W. Translation, adaptation and validation of instruments or scales for use in cross-cultural health care research: a clear and user-friendly guideline. J Eval Clin Pract 2011;17:268–274.ArticlePubMed
- 14. World Health Organization (WHO). Process of translation and adaptation of instruments [Internet]; Geneva: WHO; c2018 [accessed on 2019 Apr 7]. Available at: http://www.who.int/substance_abuse/research_tools/translation/en/.
- 15. Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology 1967;17:427–442.ArticlePubMed
- 16. Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. Mov Disord 2003;18:738–750.ArticlePubMed
- 17. Kummer A, Scalzo P, Cardoso F, Teixeira AL. Evaluation of fatigue in Parkinson’s disease using the Brazilian version of Parkinson’s Fatigue Scale. Acta Neurol Scand 2011;123:130–136.ArticlePubMed
- 18. Cunha JA. [Manual of the Portuguese version of the Beck scales]. 1st ed. São Paulo: Casa do Psicólogo; 2001:171.
- 19. Roberts P, Priest H. Reliability and validity in research. Nurs Stand 2006;20:41–45.Article
- 20. Terwee CB, Bot SD, de Boer MR, van der Windt DA, Knol DL, Dekker J, et al. Quality criteria were proposed for measurement properties of health status questionnaires. J Clin Epidemiol 2007;60:34–42.ArticlePubMed
- 21. Kaur P, Stoltzfus JC. Bland–Altman plot: a brief overview. Int J Acad Med 2017;3:110–111.Article
- 22. Sullivan GM. A primer on the validity of assessment instruments. J Grad Med Educ 2011;3:119–120.ArticlePubMedPMC
- 23. Jankovic J. Parkinson’s disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry 2008;79:368–376.ArticlePubMed
- 24. Marinus J, Zhu K, Marras C, Aarsland D, van Hilten JJ. Risk factors for non-motor symptoms in parkinson’s disease. Lancet Neurol 2018;17:559–568.ArticlePubMed
- 25. Farrance I, Frenkel R. Uncertainty of measurement: a review of the rules for calculating uncertainty components through functional relationships. Clin Biochem Rev 2012;33:49–75.PubMedPMC
- 26. Glosup JG, Axelrod MC. Systematic error revisited [Internet]; California: Lawrence Livermore National Lab; c1996 [accessed on 2019 Aug 12]. Available at: https://digital.library.unt.edu/ark:/67531/metadc688304/.
REFERENCES
Figure & Data
References
Citations

- Should patients with Parkinson’s disease only visit a neurologist’s office? - a narrative review of neuropsychiatric disorders among people with Parkinson’s disease
Julita Szarpak, Paulina Drożak, Michał Piwoński, Hubert Wróblewski, Justyna Morylowska-Topolska
Current Problems of Psychiatry.2022; 23(4): 220. CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite