Articles
- Page Path
- HOME > J Mov Disord > Volume 15(3); 2022 > Article
-
Review Article
Pallidus Stimulation for Chorea-Acanthocytosis: A Systematic Review and Meta-Analysis of Individual Data -
Weibin He1
 , Chenhui Li1
, Chenhui Li1 , Hongjuan Dong2
, Hongjuan Dong2 , Lingmin Shao1
, Lingmin Shao1 , Bo Yin2
, Bo Yin2 , Dianyou Li3
, Dianyou Li3 , Liguo Ye1
, Liguo Ye1 , Ping Hu1
, Ping Hu1 , Chencheng Zhang3
, Chencheng Zhang3 , Wei Yi1
, Wei Yi1
-
Journal of Movement Disorders 2022;15(3):197-205.
DOI: https://doi.org/10.14802/jmd.22003
Published online: July 26, 2022
1Department of Neurosurgery, Renmin Hospital of Wuhan University, Wuhan, China
2Department of Neurology, Renmin Hospital of Wuhan University, Wuhan, China
3Department of Functional Neurosurgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Corresponding author: Wei Yi, MD, PhD Department of Neurosurgery, Renmin Hospital of Wuhan University, Wuhan 430060, China / Tel: +86-027-88041911-82079 / Fax: +86-27-88042292 / E-mail: weiyi.renmin@whu.edu.cn
- Corresponding author: Chencheng Zhang, MD, PhD Department of Functional Neurosurgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China / Tel: +86-021-64370045 / Fax: +86-021-53068810 / E-mail: i@cczhang.org
Copyright © 2022 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 3,590 Views
- 308 Download
- 2 Web of Science
ABSTRACT
- A significant proportion of patients with chorea-acanthocytosis (ChAc) fail to respond to standard therapies. Recent evidence suggests that globus pallidus internus (GPi) deep brain stimulation (DBS) is a promising treatment option; however, reports are few and limited by sample sizes. We conducted a systematic literature review to evaluate the clinical outcome of GPi-DBS for ChAc. PubMed, Embase, and Cochrane Library databases were searched for relevant articles published before August 2021. The improvement of multiple motor and nonmotor symptoms was qualitatively presented. Improvements in the Unified Huntington’s Disease Rating Scale motor score (UHDRS-MS) were also analyzed during different follow-up periods. A multivariate linear regression analysis was conducted to identify potential predictors of clinical outcomes. Twenty articles, including 27 patients, were eligible. Ninety-six percent of patients with oromandibular dystonia reported significant improvement. GPi-DBS significantly improved the UHDRS-motor score at < 6 months (p < 0.001) and ≥ 6 months (p < 0.001). The UHDRS-motor score improvement rate was over 25% in 75% (15/20 cases) of patients at long-term follow-up (≥ 6 months). The multiple linear regression analysis showed that sex, age at onset, course of disease, and preoperative movement score had no linear relationship with motor improvement at long-term follow-up (p > 0.05). GPi-DBS is an effective and safe treatment in most patients with ChAc, but no reliable predictor of efficacy has been found. Oromandibular dystonia-dominant patients might be the best candidates for GPi-DBS.
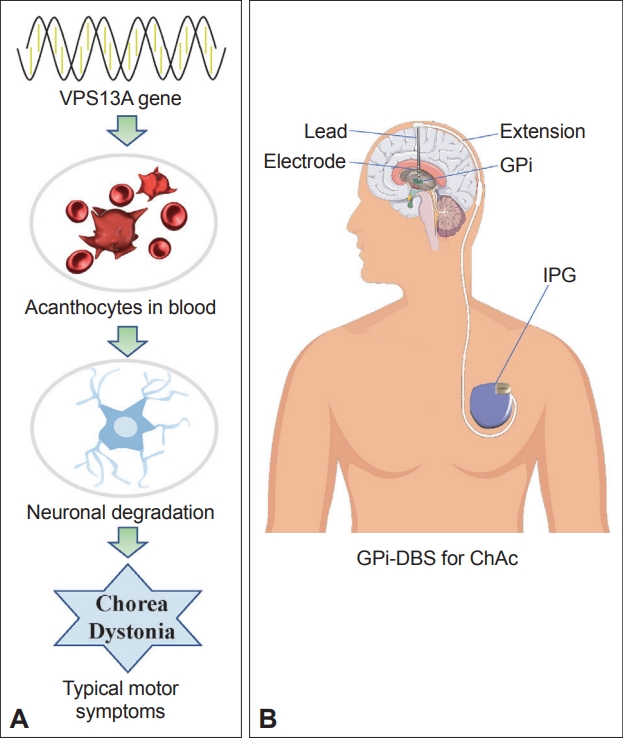
- Chorea-acanthocytosis (ChAc), an autosomal recessive neuroacanthocytosis associated with VSP13A mutations in the chorein gene, classically causes oromandibular chorea with lip and tongue biting, tongue protrusion or feeding dystonia, and paroxysmal head/neck and trunk movements [1-4]. ChAc is a chronic progressive disease. Currently, the management of ChAc is purely symptomatic and includes botulinum toxin for oral-oro-facio-lingual dystonia; phenytoin, lorazepam, and levetiracetam for seizure management; antidepressants or antipsychotics; and dopamine antagonists. However, medications and botulinum toxin injections have limited efficacy in treating various motor symptoms (such as limb chorea and dystonia) in patients with ChAc [5].
- Many studies have confirmed that deep brain stimulation (DBS) is an effective treatment modality for various movement disorders such as Parkinson’s disease (PD), essential tremor, dystonia, and Huntington’s chorea. Primary stimulation targets include the globus pallidus internus (GPi) and subthalamic nucleus (STN) [6-9]. ChAc has clinical features similar to Huntington’s disease. In 2013, Miquel et al. [10] reported that bilateral GPi-DBS could effectively reduce motor symptom severity and improve functional capacity in patients with ChAc (chorea-acanthocytosis mechanism and pallidus stimulation) (Figure 1). However, the study was limited by sample size (n = 15). In recent years, cases with good results after DBS in patients with ChAc have been reported. Therefore, we need to summarize recently reported cases of ChAc to more comprehensively and reliably evaluate the efficacy of DBS in treating ChAc.
- Current studies (mostly case reports or small case series) can only provide level III or IV evidence to confirm the efficacy of DBS in ChAc treatment. Three basic questions regarding the application of DBS to ChAc remain unanswered. First, what are the short-term and long-term effects of GPi-DBS on ChAc? Second, how should GPi-DBS be programmed to achieve the best clinical outcome, i.e., is there a difference in the efficacy of high- and low-frequency stimulations of each target? Third, what factors are correlated with postoperative motor improvements? As no large-scale randomized study has provided evidence to answer these questions, we collected available published patient data to perform an individual patient analysis and discuss the feasibility of DBS for ChAc treatment.
INTRODUCTION
- Search strategy
- This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We searched the PubMed, Embase, and Cochrane Library databases for studies published before August 1, 2021. We used the following keywords in the standard search: “chorea,” “acanthocytosis,” “chorea-acanthocytosis,” “pallidal stimulation,” “deep brain stimulation,” and “DBS”; 237 studies were retrieved.
- Inclusion and exclusion criteria
- The inclusion criteria were as follows: articles comprising patients 1) diagnosed with ChAc and 2) who underwent bilateral GPi-DBS surgery. The diagnosis was based on the clinical neurological manifestation, the presence of acanthocytes, and the molecular analysis of VPS13A mutations. The diagnosis of ChAc in some patients without Western blotting (absence of the chorein/VPS13A protein) and genetic testing is based on the patient’s medical history, clinical manifestations, the presence of acanthocytes, neurological examination, and cranial MRI findings. The exclusion criteria were as follows: 1) articles with cases reported elsewhere, 2) articles with unilateral surgical procedures or no reported laterality, 3) articles with no reported baseline features or postoperative symptom improvement, and 4) non-English articles, conference articles, and abstract-only articles. For a more comprehensive analysis of the effect of surgery, case series with missing individual patient data regarding nonmotor symptoms were included.
- Data extraction
- Two reviewers (H.W.B. and L.C.H.) independently extracted the data using custom data extraction tables to identify the baseline characteristics of patients, including age at the time of surgery and disease onset, sex, follow-up period, and disease course. In addition, the surgical process included the surgical target and stimulation parameters; postoperative improvement results, including the Unified Huntington’s Disease Rating Scale motor score (UHDRS-MS), UHDRS chorea score (UHDRS-CS), Abnormal Involuntary Movement Scale (AIMS), and Burke–Fahn–Marsden Dystonia Rating Scale (BFMDRS) score; and changes in non-motor symptoms (cognitive and psychiatric symptoms). The follow-up period was categorized as early follow-up (EFU, < 6 months) and long-term follow-up (LFU, ≥ 6 months).
- Analysis strategy
- The percent improvement in UHDRS-MS and UHDRS-CS was quantitatively analyzed. The changes in ChAc motor symptoms (chorea, bradykinesia, dystonia, and gait balance) and nonmotor symptoms (cognitive and psychiatric symptoms) were qualitatively analyzed. The stimulation parameters were placed in bins as follows: amplitude was binned by 0.5 mV/bin, frequency by 12.5 Hz/bin, and pulse width by 12.5 μs/bin. Finally, stimulation and hardware-related adverse events were analyzed qualitatively.
- Statistical analyses
- IBM SPSS Statistics for Windows version 22.0 (IBM Corp., Armonk, NY, USA) was used for data analysis. The changes in UHDRS-MS and UHDRS-CS scores were analyzed using a paired Student’s t-test or an independent sample t-test. A multivariate linear regression was performed to evaluate the association between clinical/demographic characteristics and percent change in UHDRS-MS at LFU. The latter was regarded as the dependent variable. Independent variables included sex (a binary variable, where male = 0 and female = 1), age at surgery (years), preoperative UHDRS-MS, time interval after surgery (months), stimulus frequency, and the UHDRS-MS improvement rate at EFU. All continuous data are presented as the mean ± standard deviation and range. We reported two-tailed p values and 95% confidence intervals (CIs). A significance threshold of p = 0.05 was selected, and the significance levels for multivariate linear regression were adjusted using the Benjamini–Hochberg procedure to account for multiple testing.
METHODS
- Search results and baseline features
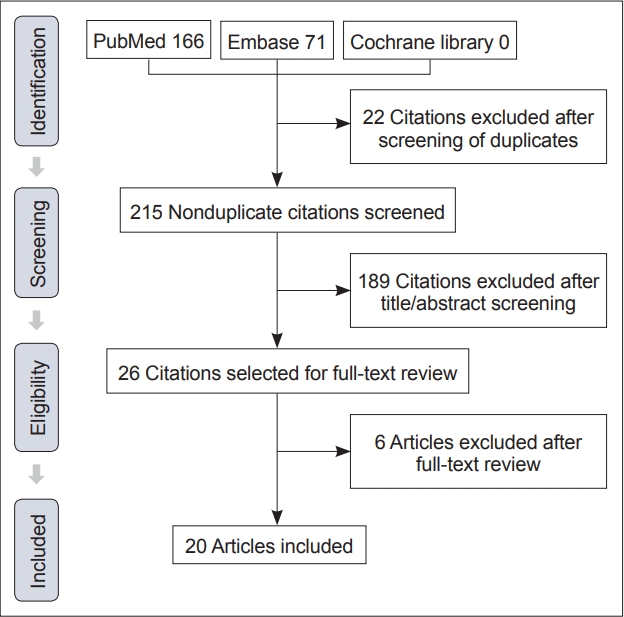
- Based on the inclusion and exclusion criteria, 20 articles, including 27 patients, were considered in the final analysis. The PRISMA flowchart is shown in Figure 2.
- Patient clinical features
- The characteristics of the patients receiving bilateral GPi-DBS are shown in Table 1. Twenty-seven patients (18 male and 9 female, mean age 37.37 years) underwent follow-up after an average duration of 22.77 months. Detailed information on the 27 patients is presented in Table 1.
- Stimulation parameters
- The program parameters of the 27 patients receiving bilateral GPi-DBS are shown in the Supplementary Table 1 (in the online-only Data Supplement). DBS treatment failed for one patient [11]; seven patients received low-frequency GPi stimulation, and the rest received high-frequency GPi stimulation. Symptoms in three low-frequency stimulation patients worsened with high-frequency stimulation. The other four patients reported optimal symptom improvement with low-frequency stimulation. The respective average stimulation parameters for patients with low- and high-frequency stimulations were as follows: amplitude, 3.1 ± 0.4 V; frequency, 55 ± 16 Hz; pulse width, 90 ± 36 μs and amplitude, 2.7 ± 0.8 V; frequency, 135 ± 18 Hz; and pulse width, 85 ± 35 μs. The average stimulation parameters of the 52 electrodes were amplitude, 3.1 ± 0.81 V; frequency, 128.1 ± 43.29 Hz; and pulse width, 89.5 ± 27.11 μs. Frequency histograms of the stimulation parameters are shown in Figure 3.
- Therapeutic effects
- The UHDRS-MS data are listed in Table 2. The UHDRS-MS scores of patients receiving GPi-DBS were significantly reduced at EFU and LFU (mean improvement rates, 51.75% ± 15.15% [n = 20] and 43.68% ± 35.42% [n = 21], respectively). The UHDRS-MS improvement rate showed a decreasing trend at LFU, but the difference in the improvement rate between the EFU and LFU periods was not statistically significant (p = 0.104). Additionally, the UHDRS-CS of patients receiving GPi-DBS was also reduced significantly at EFU and LFU (mean improvement rates, 65.32% ± 14.64% and 69.73% ± 17.55% [n = 8], respectively). The mean percent change of the UHDRS-MS was greater in the low- than in the high-frequency stimulation group (mean improvement rates at EFU, 62.21% ± 11.46% [n = 3] and 49.91% ± 15.70% [n = 17], respectively; mean improvement rates at LFU, 54.58% ± 16.70% [n = 4] and 41.12% ± 39.47% [n = 17], respectively).
- The total AIMS scores of the 17th patient improved by 40% (5 months after the surgery) and 50% (8 months after the surgery). The BFMDRS motor score (BFMDRS-MS) of the 13th patient improved by 75% at 1 year after the surgery.
- Outcome by symptoms
- Table 2 shows the motor effects of GPi-DBS. Among the 19 patients who received high-frequency stimulation, the chorea (trunk and limbs) of 16 (three did not report chorea) slightly improved, and the trunk spasm symptoms of five improved. The oromandibular dystonia of 18 (one patient did not report oromandibular dystonia) almost disappeared after bilateral high-frequency GPi stimulation. As their oromandibular dystonia was relieved, the patients’ symptoms of dysphagia also improved. The trunk and limb dystonia symptoms in eight patients improved after bilateral high-frequency GPi-DBS. The unstable standing posture and gait of four patients significantly improved under high-frequency GPi stimulation. One patient occasionally developed gait freezing after high-frequency GPi-DBS, which may have resulted from disease progression or stimulus-related side effects. There were 8 patients with dysarthria before surgery; 2 with complete remission, 1 with partial improvement, and the rest presented with no significant improvement.
- Seven patients received low-frequency GPi-DBS. The chorea of the remaining patients slightly improved except for one patient who did not report chorea. Trunk spasm symptoms completely improved in one patient and were partially relieved in two patients. After bilateral low-frequency GPi stimulation, the oromandibular dystonia almost disappeared in two patients, and the other five were partially relieved. Three of four patients had partly improved dystonia of the trunk and limbs, and one showed no such improvement. The unstable standing posture and gait of three patients significantly improved after the surgery, except for one without change. After low-frequency GPi stimulation, two of the six patients (four did not report dysarthria) showed a slight improvement in symptoms, whereas the remaining four showed no significant change in dysarthria. The burping symptoms in one patient significantly improved after low-frequency GPi-DBS.
- DBS can reduce disabling motor symptoms. In some patients with dystonia, the effect of microdamage can temporarily improve motor symptoms after DBS. Chronic stimulation significantly improves dyskinesia in patients with ChAc. At present, it is difficult to evaluate whether the initial symptoms reappear after the power of the pulse generator has been exhausted. However, the clinical symptoms reappear in some patients after the stimulator is turned off.
- Five patients had cognitive impairment before surgery; two had no change in cognitive function after GPi-DBS, and results regarding the postoperative cognition of the other patients were not reported. Eight patients had mental symptoms before surgery such as depression, anxiety, obsessive-compulsive disorder, and impulsive behavior. After the surgery, one patient’s anxiety and depression improved significantly, one patient’s mood and behavior did not change, and the results of the mental symptoms of the other patients were not reported. Therefore, there was insufficient evidence to draw conclusions about cognitive or neuropsychiatric changes in patients with ChAc after GPi-DBS.
- Adverse events
- There were two postoperative hardware-related side effects, three surgery-related side effects, and nine stimulation-induced side effects. The details of these adverse events are shown in Table 3.
- Factors associated with Unified Huntington’s Disease Rating Scale motor score change
- In the univariate linear regression analysis, sex, age at the time of surgery, DBS duration, baseline UHDRS-MS, and stimulus frequency were not associated with the percent change in UHDRS-MS at LFU in patients receiving bilateral GPi-DBS (p > 0.05) (Table 4).
RESULTS
Outcomes by scale evaluations
Motor symptoms
Nonmotor (cognitive and psychiatric) symptoms
- Our study shows that GPi-DBS effectively alleviates chorea and dystonia (including the limbs and trunk) in patients with ChAc. The UHDRS-MS improvement rate was over 25% in 75% (15/20 cases) of patients at LFU (≥ 6 months). The average improvement rate of UHDRS-MS was 43.68% at the final phase. Moreover, this study showed no significant difference between high- and low-frequency GPi-DBS in improving UHDRS-MS under the optimal parameter stimulation state. Therefore, our study is the first to explore the short- and long-term efficacy of DBS and the safety in treating ChAc in a relatively large cohort of patients with ChAc. Moreover, our study is the first to explore the UHDRS-MS improvement rates between high- and low-frequency GPi-DBS. In summary, our study has enriched the previous literature and has further validated the role of GPi-DBS in the treatment of ChAc, which may ultimately improve clinical procedures.
- Overall, these patients showed significant improvement in UHDRS-MS at EFU and LFU, indicating that GPi-DBS is a feasible surgical therapy in medically intractable ChAc treatment. Furthermore, it may alleviate motor symptoms and improve the functional status of patients. Contrary to other research [10], we could not find a correlation between surgical outcome and age at surgery, course of disease, or preoperative movement score. The heterogeneity of the diseases, wide age range of our population, and small sample size could explain these negative findings. Interestingly, it was shown that most patients benefited from stimulation of the ventral side of the GPi in the early phase of stimulation. In contrast, in the late stimulation phase, the electric field was larger and closer to the dorsal side of the GPi [10]. However, the pooled analysis could not be performed due to the unavailability of data.
- The DBS parameters of ChAc are inconsistent across various research centers. Interestingly, this study showed that the mean percent change of the UHDRS-MS improved more in the low- than high-frequency stimulation group under the optimal parameter stimulation state. Moreover, in some cases of ChAc, improvement was only achieved with low-frequency stimulation (40 Hz) with worsening of symptoms at high-frequency stimulation (130 Hz) [12,13]. According to previous studies, the therapeutic mechanism of DBS in movement disorders is unclear, and the mechanism by which different stimulation frequencies induce different therapeutic effects remains to be elucidated [14]. The cause of these antagonistic stimulatory effects remains unclear and requires further study.
- Given the heterogeneity and complexity of ChAc symptoms and the significant interindividual differences in the clinical response to DBS treatment, a single target stimulation is insufficient to manage all of the patient’s clinical symptoms (such as various involuntary movements). Using multiple targets rather than a single target may be more effective or produce long-term effects on some symptoms. A recent case report demonstrated the feasibility and effectiveness of combined complex stimulation. The trunk spasm and chorea symptoms in two patients improved significantly after combined stimulation of GPi and the ventralis oralis complex of the thalamus [15]. However, this strategy may result in a higher complication risk than using a single target. Therefore, the benefits and risks of using multiple targets must be clearly understood. Moreover, Wu et al. [16] reported that BFMDRS-MS and UHDRS-MS improved rapidly after STN-DBS in two siblings with ChAc. STN-DBS appears to provide effective, long-lasting, and more rapid-acting treatment for ChAc and shows potential economic advantages because it uses less electrical energy [17,18]. Although STN-DBS can quickly improve patients’ motor symptoms, its potential risks of causing or aggravating cognitive impairment, insufficient improvement of axial symptoms, and declines in verbal fluency tasks, in particular, should also be considered [16].
- In the present review, oromandibular dystonia significantly improved in 25 of the 26 patients postsurgery. In cases where oromandibular dystonia affects a patient’s voice, the voice becomes clearer as the condition improves. In this study, the dysarthria of five patients receiving GPi-DBS improved postoperatively. However, nine patients showed no improvement in postoperative dysarthria. The nonreactivity or deterioration of dysarthria symptoms may be related to disease progression or adverse reactions to DBS. Generally, DBS improves dysarthria, as seen in patients with PD after STN-DBS [19].
- Standard antiepileptic drugs can control epileptic symptoms before surgery, and DBS does not aggravate these symptoms. Moreover, there are limited reports on seizures in patients with other movement disorders caused by GPi or STN stimulation. Because most studies have not systematically reported the changes in nonmotor symptoms after DBS, there is insufficient evidence to make conclusions regarding the changes in nonmotor symptoms (cognitive and neuropsychic) after DBS in patients with ChAc.
- The adverse reactions reported in this study also deserve attention. Hardware-related complications can occur even 1 year after surgery. Implantable pulse generator protrusion or lead/wire fracture can occur, particularly in patients with ChAc presenting with chorea, which can be violent. Furthermore, in some patients, the main motor symptoms of chorea improved, whereas other symptoms such as dysarthria or gait problems often developed or worsened. Future studies should preferably involve a random division of patients into surgical and nonsurgical control groups to evaluate effects that may occur independently of DBS due to the natural progression of the disease. Fundamentally, DBS may be chosen when the patient has a relatively slow progression. For example, all the subjects who underwent DBS had an atypical presentation that indicated a milder disease course than the typical presentation from childhood. Therefore, DBS can be recommended in some but not all cases, although clinical symptoms are disabling even with many medications.
- In the context of these overall positive findings, some limitations must be highlighted in the published case reports. First, the data come from case reports or case series studies and articles involving operations performed by different surgeons and evaluations performed by different doctors (Table 1 [4,10-13,20-34]), which may affect surgical efficacy evaluation. The majority of the reported cases did not use blind methods to evaluate symptoms, which could bias results. Moreover, in some patients, neither western blotting nor VPS13A mutation tests were performed. Therefore, it is essential to establish a professional medical center to systematically evaluate and treat patients with ChAc. Second, publication bias (the publication of positive results) may exaggerate the real benefits of surgery in patients with ChAc. Moreover, the final follow-up of most reported cases was one year. Undoubtedly, a LFU study is needed to evaluate the effect of DBS on activities of daily living and quality of life. Third, there are no images to determine the electrode placement position for comparison between different studies. Due to the unavailability of data on the stimulation area, the correlation between symptoms and the stimulation area in patients with ChAc was not analyzed. Due to the limited data, the outcomes of GPi-DBS on cognition and behavior of patients with ChAc deserve further study. Currently, the GPi is the target of most DBS for ChAc; however, the best target is still unknown. Therefore, more extensive clinical and basic studies are needed to better understand the mechanism of ChAc and discover ideal targets and treatment modalities.
- In conclusion, this study demonstrated that GPi-DBS is an effective and safe treatment in most patients with ChAc, but no reliable predictor of efficacy has been found. Chorea and dystonia can be adequately and consistently controlled via GPi-DBS. Despite disease progression, oromandibular dystonia-dominant patients might be the best candidates for GPi-DBS surgery. Moreover, the optimal stimulation program remains unknown; stimulation frequency, pulse width, and amplitude should be adjusted according to the principle of individualization.
DISCUSSION
Supplementary Materials
Supplementary Table 1.
-
Conflicts of Interest
The authors have no financial conflicts of interest.
-
Funding Statement
Non
-
Author Contributions
Conceptualization: Wei Yi, Chencheng Zhang, Weibin He, Chenhui Li, Hongjuan Dong, Lingmin Shao, Bo Yin, Dianyou Li. Data curation: Weibin He, Chenhui Li, Chencheng Zhang, Wei Yi. Formal analysis: Weibin He, Chenhui Li, Liguo Ye, Ping Hu, Chencheng Zhang, Wei Yi. Investigation: Weibin He, Chenhui Li. Methodology: Weibin He, Chenhui Li, Chencheng Zhang, Wei Yi, Liguo Ye, Ping Hu. Project administration: Hongjuan Dong, Lingmin Shao, Bo Yin, Dianyou Li, Chencheng Zhang, Wei Yi. Software: Weibin He, Liguo Ye, Ping Hu. Validation: Weibin He, Chencheng Zhang, Wei Yi, Chenhui Li. Visualization: Weibin He. Writing—original draft: Weibin He. Writing—review & editing: all authors.
Notes
- We would like to express our deepest gratitude to Umberto Spampinato and David K. Simon for offering individual data, Yuhan Wang for assistance with statistical analysis, and Hongxia Li for her thoughtful suggestions and comments on the research conception and the revision of this manuscript.
Acknowledgments


| Patient number | Sex | Chorea in Western blot of red cells membranes | VPS13A mutations | CK (U/L) | MRI | Acanthocytes (%) | Age at disease onset (yr) | Age at the time of surgery (yr) | LFU (mo) |
|---|---|---|---|---|---|---|---|---|---|
| P1 [20] | F | AB | ND | Inc | Ca | 20 | 25 | 33 | 60 |
| P2 [21] | F | ND | Present | × 1.59 | NA | 20 | 35 | 39 | 13 |
| P3 [22] | M | AB | ND | NA | Ca | + | 30 | 54 | 5 |
| P4 [23] | M | ND | Present | Inc | NA | 21 | 33 | 36 | 24 |
| P5 [4] | M | ND | Present | 586 | NA | 20 | 32 | 35 | 12 |
| P6 [4] | M | ND | Present | 815 | Stri | Many | 30 | 37 | 12 |
| P7 [4] | M | ND | Present | 2,601 | NA | Many | 32 | 37 | 12 |
| P8 [4] | M | ND | Present | 688 | NA | Many | 29 | 35 | 12 |
| P9 [4] | F | ND | Present | 1,014 | NA | Many | 33 | 36 | 12 |
| P10 [4] | F | ND | Present | 233 | NA | 5 | 29 | 33 | 12 |
| P11 [24] | F | ND | ND | NA | Ca | 30 | 38 | 43 | 12 |
| P12 [25] | M | ND | Present | × 32.6 | Nor | + | 22 | 31 | 6 |
| P13 [26] | F | ND | ND | NA | NA | NA | 22 | 31 | 45 |
| P14 [13,27] | M | ND | Present | × 18.8 | Nor | 10 | 24 | 32 | 84 |
| P15 [12] | M | ND | ND | × 2.22 | Ca | 6 | 17 | 40 | 9 |
| P16 [12] | M | ND | ND | Normal | Ca | + | 18 | 30 | 21 |
| P17 [28] | M | ND | ND | NA | Ca; Pu | NA | 26 | 32 | 8 |
| P18 [29] | M | ND | Present | Inc | Nor | 7 | 30 | 43 | 12 |
| P19 [30] | F | AB | ND | × 4 | Ca | NA | 31 | 38 | 36 |
| P20 [10] | F | Present | Present | NA | Ca | NA | 35 | 41 | 36 |
| P21 [10] | F | Present | ND | NA | Nor | NA | 25 | 32 | 6 |
| P22 [11] | M | ND | Present | × 4.18 | Ca | 20–25 | 35 | 38 | 1 week microlesioning effect; stop DBS at 3 weeks, ineffective |
| P23 [22] | M | AB | ND | NA | Ca | + | 32 | 43 | 3 |
| P24 [31,32] | M | AB | Present | × 2.6 | Nor | NA | 48 | 49 | 41 |
| P25 [33] | M | AB | Present | NA | NA | NA | 29 | 48 | 24 |
| P26 [33] | M | AB | Present | NA | NA | NA | 30 | 32 | 36 |
| P27 [34] | M | ND | Present | NA | Ca; Pu | NA | 22 | 31 | 39 |
| Mean ± SD | 5 F (33%), 10 M (67%) | / | / | / | / | / | 29.3 ± 6.48 | 37.37 ± 6.17 | 22.77 ± 19.45 |
| Patient |
UHDRS-MS |
UHDRS-CS |
Symptom |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UHDRS-MS at baseline | UHDRS-MS at EFU (imp%) | UHDRS-MS at LFU (imp%) | UHDRS-CS at baseline | UHDRS-CS at EFU (imp%) | UHDRS-CS at LFU (imp%) | Chorea | Dystonia | Feeding difficulties | Dysarthria | Oromandibular dystonia | Trunk spasm | Gait balance | Involuntary movement | |
| P1 | 24 | 14 (41.7) | 21 (12.5) | 24 | 14 (41.67) | NA | ↑ | ↑ | NA | → | ↑ | ↑ | NA | NA |
| P2 | 44 | 13 (70.45) | 12 (72.73) | 13 | 2 (84.62) | 2 (84.62) | ↑ | ↑ | ↑ | ↑ | ↑ | ↑ | NA | NA |
| P3 | 40 | 23 (42.5) | NA | 8 | 1 (87.5) | NA | ↑ | NA | ↑ | NA | ↑ | ↑ | NA | ↑ |
| P4 | 59 | 36 (38.98) | 35 (40.68) | NA | NA | NA | ↑ | NA | NA | → | ↑ | ↑ | ↑ | NA |
| P5 | 62 | 20 (67.74) | 18 (70.97) | 20 | 7 (65) | 5 (75) | ↑ | ↑ | ↑ | → | ↑ | NA | ↑ | NA |
| P6 | 42 | 16 (61.9) | 12 (71.4) | 12 | 5 (58) | 3 (75) | ↑ | ↑ | ↑ | → | ↑ | NA | ↑ | NA |
| P7 | 24 | 11 (54.17) | 10 (58.33) | 8 | 4 (50) | 2 (75) | ↑ | ↑ | ↑ | → | ↑ | NA | ↑ | NA |
| P8 | 22 | 11 (50) | 10 (55) | 8 | 2 (75) | 2 (75) | ↑ | ↑ | ↑ | → | ↑ | NA | ↑ | NA |
| P9 | 48 | 26 (45.83) | 24 (50) | 14 | 4 (71.43) | 2 (85.71) | ↑ | ↑ | ↑ | → | ↑ | NA | ↑ | NA |
| P10 | 16 | 7 (56.25) | 7 (56.25) | 6 | 3 (50) | 4 (33) | ↑ | ↑ | ↑ | → | ↑ | NA | ↑ | NA |
| P11 | 61 | NA | 31 (49.18) | 24 | NA | 11 (54.17) | ↑ | NA | NA | ↑ | ↑ | NA | ↑ | NA |
| P12 | NA | NA | NA | 13 | NA | 4 (69.23) | ↑ | NA | ↑ | ↑ | ↑ | NA | ↑ | NA |
| P13 | NA | NA | NA | 14* | NA | 3.5 (75) | ↑ | NA | → | → | ↑ | NA | NA | NA |
| P14 | 34 | 26 (23.53) | 54 (-58.82) | NA | NA | NA | ↑ | → | NA | ↑ | ↑ | NA | → | NA |
| P15 | 36 | 13 (63.89) | 13 (63.89) | 14 | 3 (78.57) | 4 (71.43) | ↑ | ↑ | ↑ | → | ↑ | ↑ | ↑ | NA |
| P16 | 53 | 26.5 (50) | 37 (30) | 20 | 9 (55) | NA | ↑ | ↑ | ↑ | NA | ↑ | ↑ | NA | NA |
| P17 | NA | NA | NA | 20† | 12 (40) | 10 (50) | ↑ | ↑ | ↑ | ↑ | ↑ | NA | ↑ | ↑ |
| P18 | 39 | NA | 13 (66.67) | NA | NA | NA | ↑ | NA | ↑ | → | ↑ | ↑ | ↑ | NA |
| P19 | 32 | 12 (62.5) | 13 (59.4) | NA | NA | NA | NA | NA | NA | → | ↑ | NA | NA | NA |
| P20 | 35 | 27 (22.86) | 32 (8.57) | NA | NA | NA | NA | NA | NA | → | ↑ | NA | NA | NA |
| P21 | 55 | 33 (40) | 31 (44) | NA | NA | NA | NA | NA | NA | → | ↑ | NA | NA | NA |
| P22 | NA | NA | NA | NA | NA | NA | → | NA | → | NA | → | → | NA | NA |
| P23 | 74 | 50 (32.43) | NA | 24 | 8 (66.67) | NA | ↑ | NA | NA | → | NA | ↑ | ↑ | NA |
| P24 | 33 | 9 (72.73) | 14 (57.58) | NA | NA | NA | ↑ | NA | NA | → | ↑ | NA | NA | NA |
| P25 | 67 | 18 (73.13) | 10 (85.07) | NA | NA | NA | ↑ | NA | NA | → | ↑ | NA | ↑ | NA |
| P26 | 31 | 11 (64.52) | 11 (64.52) | NA | NA | NA | ↑ | NA | NA | → | ↑ | NA | ↑ | NA |
| P27 | 15 | NA | 21 (-40) | 14 | 9 (35.71) | 22 (-57.14) | ↑ | NA | NA | → | ↑ | NA | NA | NA |
| Number of patents with improvement/no change/worsening/NA | / | / | / | / | / | / | 23/1/0/3 | 12/1/0/15 | 13/2/0/12 | 5/19/0/3 | 25/1/0/1 | 8/1/0/18 | 15/1/0/11 | 2/0/0/25 |
* BFMDRS-MS;
† AIMS.
UHDRS-MS, Unified Huntington’s Disease Rating Scale motor score; EFU, early follow-up (< 6 months); LFU, long-term follow-up (≥ 6 months); UHDRS-CS, UHDRS chorea score; NA, not assessed/not reported; BFMDRS-MS, Burke–Fahn–Marsden Dystonia Rating Scale Score motor score; AIMS, Abnormal Involuntary Movement Scale; ↑, improvements; →, no change.
- 1. Jung HH, Danek A, Walker RH. Neuroacanthocytosis syndromes. Orphanet J Rare Dis 2011;6:68.ArticlePubMedPMC
- 2. Walker RH. Untangling the thorns: advances in the neuroacanthocytosis syndromes. J Mov Disord 2015;8:41–54.ArticlePubMedPMC
- 3. Bader B, Walker RH, Vogel M, Prosiegel M, McIntosh J, Danek A. Tongue protrusion and feeding dystonia: a hallmark of chorea-acanthocytosis. Mov Disord 2010;25:127–129.ArticlePubMed
- 4. Liu Z, Liu Y, Wan X, Yang Y, Wang L, Dou W, et al. Pallidal deep brain stimulation in patients with chorea-acanthocytosis. Neuromodulation 2018;21:741–747.ArticlePubMed
- 5. Liu Y, Liu ZY, Wan XH, Guo Y. Progress in the diagnosis and management of chorea-acanthocytosis. Chin Med Sci J 2018;33:53–59.ArticlePubMed
- 6. Deng ZD, Li DY, Zhang CC, Pan YX, Zhang J, Jin H, et al. Long-term follow-up of bilateral subthalamic deep brain stimulation for refractory tardive dystonia. Parkinsonism Relat Disord 2017;41:58–65.ArticlePubMed
- 7. Feinstein E, Walker R. An update on the treatment of chorea. Curr Treat Options Neurol 2018;20:44.ArticlePubMed
- 8. Tsuboi T, Wong JK, Almeida L, Hess CW, Wagle Shukla A, Foote KD, et al. A pooled meta-analysis of GPi and STN deep brain stimulation outcomes for cervical dystonia. J Neurol 2020;267:1278–1290.ArticlePubMed
- 9. Wojtecki L, Groiss SJ, Hartmann CJ, Elben S, Omlor S, Schnitzler A, et al. Deep brain stimulation in Huntington’s disease-preliminary evidence on pathophysiology, efficacy and safety. Brain Sci 2016;6:38.ArticlePubMedPMC
- 10. Miquel M, Spampinato U, Latxague C, Aviles-Olmos I, Bader B, Bertram K, et al. Short and long term outcome of bilateral pallidal stimulation in chorea-acanthocytosis. PLoS One 2013;8:e79241. ArticlePubMedPMC
- 11. Wihl G, Volkmann J, Allert N, Lehrke R, Sturm V, Freund HJ. Deep brain stimulation of the internal pallidum did not improve chorea in a patient with neuro-acanthocytosis. Mov Disord 2001;16:572–575.ArticlePubMed
- 12. Li P, Huang R, Song W, Ji J, Burgunder JM, Wang X, et al. Deep brain stimulation of the globus pallidus internal improves symptoms of choreaacanthocytosis. Neurol Sci 2012;33:269–274.ArticlePubMed
- 13. Guehl D, Cuny E, Tison F, Benazzouz A, Bardinet E, Sibon Y, et al. Deep brain pallidal stimulation for movement disorders in neuroacanthocytosis. Neurology 2007;68:160–161.ArticlePubMed
- 14. Montgomery EB Jr, Gale JT. Mechanisms of action of deep brain stimulation (DBS). Neurosci Biobehav Rev 2008;32:388–407.ArticlePubMed
- 15. Nakano N, Miyauchi M, Nakanishi K, Saigoh K, Mitsui Y, Kato A. Successful combination of pallidal and thalamic stimulation for intractable involuntary movements in patients with neuroacanthocytosis. World Neurosurg 2015;84:1177.e1–e7.ArticlePubMed
- 16. Wu Y, Li H, Zhang C, Sun B, Li D, Wu Y. Subthalamic nucleus deep brain stimulation in two siblings with chorea-acanthocytosis. Neurol Sci 2020;41:1623–1625.ArticlePubMedPMC
- 17. Lin S, Wu Y, Li H, Zhang C, Wang T, Pan Y, et al. Deep brain stimulation of the globus pallidus internus versus the subthalamic nucleus in isolated dystonia. J Neurosurg 2019;132:721–732.ArticlePubMed
- 18. Deng Z, Pan Y, Zhang C, Zhang J, Qiu X, Zhan S, et al. Subthalamic deep brain stimulation in patients with primary dystonia: a ten-year follow-up study. Parkinsonism Relat Disord 2018;55:103–110.ArticlePubMed
- 19. Koivu M, Huotarinen A, Scheperjans F, Laakso A, Kivisaari R, Pekkonen E. Motor outcome and electrode location in deep brain stimulation in Parkinson’s disease. Brain Behav 2018;8:e01003. ArticlePubMedPMC
- 20. Ruiz PJ, Ayerbe J, Bader B, Danek A, Sainz MJ, Cabo I, et al. Deep brain stimulation in chorea acanthocytosis. Mov Disord 2009;24:1546–1547.ArticlePubMed
- 21. Shin H, Ki CS, Cho AR, Lee JI, Ahn JY, Lee JH, et al. Globus pallidus interna deep brain stimulation improves chorea and functional status in a patient with chorea-acanthocytosis. Stereotact Funct Neurosurg 2012;90:273–277.ArticlePubMed
- 22. Kefalopoulou Z, Zrinzo L, Aviles-Olmos I, Bhatia K, Jarman P, Jahanshahi M, et al. Deep brain stimulation as a treatment for chorea-acanthocytosis. J Neurol 2013;260:303–305.ArticlePubMed
- 23. Lee JH, Cho WH, Cha SH, Kang DW. Globus pallidus interna deep brain stimulation for chorea-acanthocytosis. J Korean Neurosurg Soc 2015;57:143–146.ArticlePubMedPMC
- 24. Wang KL, Hess CW, Xu D, Zhang JG, Hu W, Meng FG. High frequency bilateral globus pallidus interna deep brain stimulation can improve both chorea and dysarthria in chorea-acanthocytosis. Parkinsonism Relat Disord 2019;62:248–250.ArticlePubMed
- 25. Richard A, Hsu J, Baum P, Alterman R, Simon DK. Efficacy of deep brain stimulation in a patient with genetically confirmed chorea-acanthocytosis. Case Rep Neurol 2019;11:199–204.ArticlePubMedPMC
- 26. Beaulieu-Boire I, Aquino CC, Fasano A, Poon YY, Fallis M, Lang AE, et al. Deep brain stimulation in rare inherited dystonias. Brain Stimul 2016;9:905–910.ArticlePubMed
- 27. Sibon I, Ghorayeb I, Arné P, Tison F. Distressing belching and neuroacanthocytosis. Mov Disord 2004;19:856–859.ArticlePubMed
- 28. Lim TT, Fernandez HH, Cooper S, Wilson KM, Machado AG. Successful deep brain stimulation surgery with intraoperative magnetic resonance imaging on a difficult neuroacanthocytosis case: case report. Neurosurgery 2013;73:E184–E187.discussion E188. PubMed
- 29. Fernández-Pajarín G, Sesar A, Ares B, Jiménez-Martín I, Blanco-Arias P, Corredera E, et al. Deep brain bilateral pallidal stimulation in choreaacanthocytosis caused by a homozygous VPS13A mutation. Eur J Neurol 2016;23:e4–e5.ArticlePubMed
- 30. Guridi J, Rodriguez-Oroz MC, Alegre M, Obeso JA. Hardware complications in deep brain stimulation: electrode impedance and loss of clinical benefit. Parkinsonism Relat Disord 2012;18:765–769.ArticlePubMed
- 31. Gan JJ, Gupta F, Cheung T, Alterman RL, Gora-Stahlberg G, Tagliati M. Long-term benefit of pallidal deep brain stimulation in a case of choreaacanthocytosis. Neurology 2011;76:A590.
- 32. Walker RH, Schulz VP, Tikhonova IR, Mahajan MC, Mane S, Arroyo Muniz M, et al. Genetic diagnosis of neuroacanthocytosis disorders using exome sequencing. Mov Disord 2012;27:539–543.ArticlePubMed
- 33. Yokochi F, Kimura K, Okiyama R, Taniguchi M, Yokochi M. Surgical treatment for neuroacanthocytosis. Mov Disord 2011;26(Suppl 2):S96–S97.
- 34. Schneider SA, Lang AE, Moro E, Bader B, Danek A, Bhatia KP. Characteristic head drops and axial extension in advanced chorea-acanthocytosis. Mov Disord 2010;25:1487–1491.ArticlePubMed
REFERENCES
Figure & Data
References
Citations

Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite