Pseudobulbar Affect in Parkinsonian Disorders: A Review
Article information
Abstract
Pseudobulbar affect (PBA) is a neurological symptom of inappropriate and uncontrollable laughter or crying that occurs secondary to a variety of neurological conditions, including parkinsonian disorders. PBA is a socially and emotionally debilitating symptom that has been estimated to affect 3.6% to 42.5% of the population with Parkinson’s disease. While indexing measures and treatment options for PBA have been extensively studied in neurological conditions such as amyotrophic lateral sclerosis and multiple sclerosis, there has been considerably less attention given in the literature to PBA in parkinsonian disorders. The purpose of this review is to discuss the pathophysiology of PBA, its prevalence and impact on quality of life in parkinsonian disorders, and the treatment options currently available. Areas requiring further study, including the development of standardized, cross-culturally validated methods of symptom assessment, and evidence-based studies exploring the efficacy of current treatment options in parkinsonian disorders, are also highlighted.
In 1911, Hermann Oppenheim used the phrase pseudobulbar affect (PBA) to describe “explosive bursts of laughter or weeping.” [1] Today, PBA describes a neurological symptom involving involuntary and sudden laughter or crying that is disproportionate to or incongruent with an individual’s emotional state [1-6]. Unlike a mood disorder, which pertains to an individual’s emotional state over an extended period of time, PBA involves a deficit in regulation of emotional expression resulting in explosive and irregular emotional responses [2,5]. Various labels have been used to describe this symptom, including emotional lability, emotional incontinence, involuntary emotional expression disorder, emotional dysregulation, emotionalism, or pathological laughter or crying. PBA can occur in a variety of neurological conditions, including amyotrophic lateral sclerosis (ALS), multiple sclerosis (MS), Alzheimer’s disease (AD), stroke, traumatic brain injury (TBI) and parkinsonian disorders [1,2,7-9]. This article focuses on PBA specifically in parkinsonian disorders, examining its pathophysiology, prevalence, impact on quality of life (QoL), commonly used screening tools, and treatment options.
METHODS
References for this review were found by searching PubMed, focusing on papers published between January 1993 and March 2018. A literature search using the search terms “Pseudobulbar affect” or “PBA” or “emotional lability” or “emotional incontinence” AND “Parkinson’s” or “Parkinson’s Disease” or “Parkinson” yielded a total of 48 references. An additional PubMed search performed using the search terms “Pseudobulbar affect” or “PBA” or “emotional lability” or “emotional incontinence” AND “atypical parkinsonian disorder” or “Lewy body dementia” or “progressive supranuclear palsy” or “multiple system atrophy” or “corticobasal syndrome” yielded 4 references. A literature search using the search terms “Treatment” or “SNRI” or “SSRI” or “SSRIs” or “tricyclic antidepressant” or “dextromethorphan/quinidine” or “DMQ” AND “Parkinson’s Disease” or “Parkinson” AND “Pseudobulbar affect” or “PBA” or “emotional lability” or “emotional incontinence” yielded 27 references, many of which overlapped with the prior search results. A total of 19 articles were deemed to be relevant to this review given its focus on PBA in patients with parkinsonian disorders specifically. Additional references were identified after reading through the 19 chosen articles and were included based on their discussion of PBA pathophysiology, prevalence, and treatment. Only articles published in English were included.
PATHOPHYSIOLOGY OF PBA
The pathophysiology of PBA is complex and incompletely understood. It is believed that PBA occurs as a result of injury to the cortico-ponto-cerebellar circuitry involved in regulating the motor aspects of emotional expression [2,4,7,9]. Given that PBA commonly occurs in a diverse range of neurological conditions, it appears that the location of the pathology is in fact more important than the underlying pathophysiology of the primary disease process.
The original hypothesis for the pathophysiology underlying PBA as theorized by Wilson [10] in 1923 was based on postmortem studies, and purported that PBA stems from lesions to the motor cortex resulting in loss of voluntary inhibition to the brainstem. This disinhibition was hypothesized to subsequently contribute to the release of lower bulbar tracts regulating motor expression of laughter and crying and thus disinhibition of the involuntary emotional pathway [2,4]. More recently, it has been proposed that PBA occurs secondary to dysfunction of a broader cortico-ponto-cerebellar circuitry, including motor, limbic, and association cortices with descending pathways to the brainstem, basis pontis, and cerebellum [2,4,11,12]. In the normal state, the cerebellum modulates emotional expression to produce a response that is congruent with the emotional context, information that is received from the cortex. Disruption of the cortico-ponto-cerebellar circuitry results in emotional expression that is exaggerated or inconsistent with a given context [4,5,11,12].
A number of lines of evidence, including neuroimaging and lesion studies, support the involvement of this circuitry in the pathophysiology of PBA. A recent neuroimaging study found that compared to patients without PBA, patients with ALS or primary lateral sclerosis and PBA exhibited increased mean diffusivity of white matter tracts underlying the frontotemporal cortex, the transverse pontine fibers, and the middle cerebellar peduncle, corresponding to a loss of integrity of these tracts in patients with PBA [13]. Furthermore, the study found reduced fractional anisotropy in a region underlying the left motor cortex in patients with PBA. Taken together, these imaging findings corroborate the presence of disrupted fronto-cerebellar circuitry in PBA, and point to the involvement of the motor cortex in this disorder. Reports of patients with lesions restricted to the cerebellum, its relay center in the basis pontis, or the thalamus also demonstrate that lesions to these regions are sufficient to produce PBA symptomatology [14-19], confirming the involvement of this circuitry in the pathophysiology of PBA.
The suggested pathophysiology is also in accordance with the increased prevalence of PBA reported in atypical parkinsonian disorders, such as progressive supranuclear palsy (PSP) and the cerebellar type of multiple system atrophy (MSA-C). MSA-C is characterized by significant pathological changes in the cerebellum and associated brainstem structures in the basis pontis, which may result in impaired cerebellar modulation of unconscious expression of emotion. The higher prevalence of PBA in MSA-C patients with more severe structural derangements of the cerebellum and basis pontis has been provided as evidence in support of this potential mechanism [5]. The higher prevalence of PBA in PSP may similarly stem from greater involvement of brainstem structures known to play a role in regulating the motor expression of human affect [20].
While numerous neurotransmitters are known to regulate signaling of this cortico-ponto-cerebellar circuitry, the exact role of these neurotransmitters and how they are disrupted in PBA has not been thoroughly elucidated to date. Serotonin and glutamate are thought to be key players in PBA, given the efficacy of drugs known to target these neurotransmitters in treating PBA symptomatology. Specifically, the efficacy of selective serotonin reuptake inhibitors (SSRIs) in the treatment of PBA supports the role of serotonin in the underlying pathophysiology of the disorder [4,5,21,22]. The role of serotonin as an important emotional modulator and the presence of serotonergic projections throughout the cortico-limbic areas, including the cerebellum, is also consistent with the importance of serotonergic neurotransmission in PBA [4]. While the precise mechanism of dextromethorphan/quinidine (DMQ), currently the only FDA-approved treatment for PBA, remains unknown, it is thought to act as an σ-1 agonist and noncompetitive NMDA antagonist. The efficacy of DMQ is speculated to stem from anti-glutamatergic effects that serve to restore inhibitory neurotransmission from the cortex to the brainstem, thereby compensating for the gate-control disinhibition of emotional expression thought to underlie PBA [4]. DMQ has also been demonstrated to bind to the serotonin transporter and serotonin 5-HT1B/D receptor, suggesting that its efficacy may also stem in part from modulating serotonergic neurotransmission [23,24].
EVALUATION OF PBA
Several scales assessing PBA symptomatology currently exist, although unfortunately none of these scales have been specifically validated in the parkinsonian population. Of note, the results from these scales have been demonstrated to poorly correlate with the results of more formal diagnostic interviews [25], suggesting the scales may not accurately capture PBA in the parkinsonian population. Furthermore, the reliance of these scales on patient self-reports may limit their utility in patients with Parkinson’s dementia who cannot provide an accurate history. In addition to their lack of validation in parkinsonian disorders, these scales have not been validated cross-culturally outside of North America, another serious shortcoming of these indexing methods. Nevertheless, we feel it is important to discuss these scales below given their common use in studies exploring PBA prevalence in the parkinsonian population.
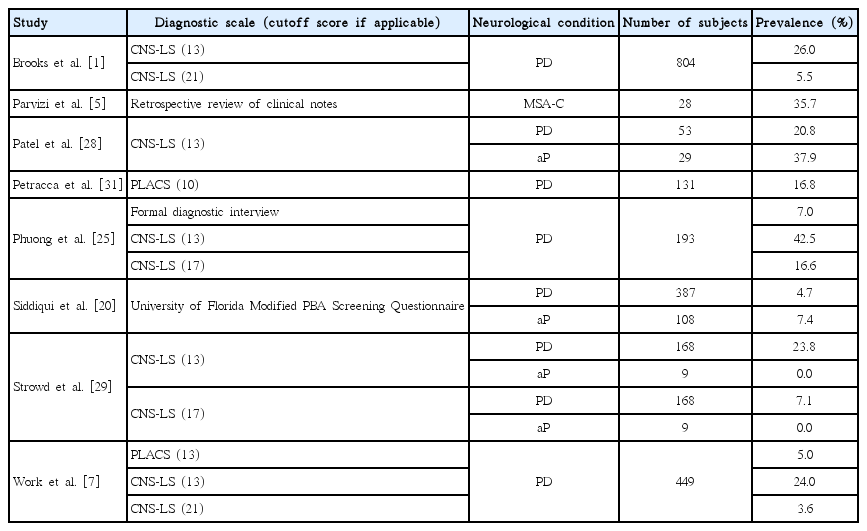
The Center for Neurological-Study Lability Scale (CNS-LS) is a self-reported measure of PBA that was first validated in the United States and Canada in ALS patients [26] and subsequently validated in patients with MS [27]. The questionnaire items were initially identified through interviews with patients and their families, and expert review. A 7-item questionnaire was developed composed of two subscales: one measuring labile laughter, comprised of 4 questions, and the other measuring labile tearfulness, comprised of 3 questions. Given that the CNS-LS has only been validated in PBA patients with ALS or MS, it may not be a viable measure for gauging PBA symptoms in patients with parkinsonian disorders. In fact, one study found that using a CNS-LS cutoff score of greater than or equal to 11 for detecting the presence of PBA symptoms in PD patients only provided a specificity of 51% for diagnosing PBA [25]. In addition to uncertainty regarding the validity of the CNS-LS in parkinsonian disorders, there is also a lack of agreement regarding the appropriate cutoff score to define PBA symptoms, with different studies using cutoff scores ranging from 11 to 21 [1,25,28,29].
The Pathological Laughing and Crying Scale (PLACS) is an 18-item questionnaire that has been validated for the assessment of PBA symptom severity in stroke patients in the United States [30]. Two items are designed to screen for the presence of sudden episodes of crying or laughing, eight items pertain to laughing, and the remaining eight pertain to crying. When investigators administered the questionnaire in stroke patients to diagnose PBA, they determined that a cutoff score of 13 provided a sensitivity of 0.88 and specificity of 0.96; however, the PLACS has not been validated in other patient populations. Despite its lack of validation, the PLACS has been used in several studies investigating PBA prevalence in neurological conditions other than stroke, including parkinsonian disorders [7,31].
PREVALENCE OF PBA
Estimates of the prevalence of PBA symptoms vary considerably across different neurological conditions and are dependent upon scales used to assess symptoms and the cutoff thresholds selected. In one study, the prevalence of PBA in a group of patients with AD, ALS, MS, stroke, TBI and PD ranged from 9.4% to 37.5% depending upon the scale and the threshold used [7]. Across different neurological conditions, prevalence estimates ranged from 9% to 40% in AD, 12% to 70% in ALS, 10% to 50% in MS, 5% to 60% in stroke, 5% to 80% in TBI, and 3.6% to 43% in patients with PD [1,4,31]. The Pseudobulbar Affect Registry Investigating Symptom Management (PRISM) study, whose goal was to estimate the prevalence of PBA symptoms across various neurological conditions, found that the proportion of patients with a CNS-LS score of at least 13 was lowest in the PD group (26%) and highest in patients with TBI (52.4%). Using a CNS-LS threshold of 21, PBA symptom prevalence was still lowest in the PD group (5.5%) and highest in the TBI group (16.4%) [1,2].
While PBA tends to be less prevalent in movement disorders as compared to conditions such as ALS and TBI, it remains a disabling condition in many patients with parkinsonian disorders. PBA prevalence rates in patients with PD have been shown to range from 3.6% to 43%, and this high variability is in part secondary to differences in indexing measures and thresholds used (Table 1) [1,7,20,25,28,29,31]. Furthermore, some studies have found that PBA prevalence increases with disease progression [20,31]; between-study differences in disease severity may therefore contribute to between-study variability in PBA prevalence. One study of clinic-based movement disorders patients using a pilot PBA screening tool found the prevalence of PBA to be 4.7% in idiopathic PD and 7.4% in patients with atypical parkinsonism (aP). Among atypical parkinsonian disorders, the prevalence of PBA was 5.7% in MSA, 7.7% in Lewy body dementia (DLB), 11.1% in vascular parkinsonism, and 21.0% in PSP; the investigators failed to detect PBA in patients with corticobasal degeneration (CBD) [20]. The relatively high prevalence of PBA in PSP in this study is notable, and merits replication in further studies. Another study that used the CNS-LS to assess PBA prevalence in PD found prevalence rates of 7.1% vs. 23.8% depending upon whether a cutoff score of 13 or 17 was used [29]. None of the 9 patients in this study with aP, which included patients with PSP, CBD, MSA, DLB, and vascular parkinsonism, were found to have PBA. In contrast, another study found the prevalence of PBA in their cohort of patients with aP to be nearly double that seen in their PD population (37.9% vs. 20.8%) [28]. A retrospective chart review of patients with MSA-C (n = 28) found that 35.7% experienced PBA, with 50% exhibiting pathological crying and 50% exhibiting both laughter and crying [5]. The variability in PBA prevalence among atypical parkinsonian disorders in these studies may in part stem from the small sample sizes and sample heterogeneity; studies with larger sample sizes are required to further address symptom prevalence in these patients.
Some investigators have tried to address the shortcomings of the clinical scales for PBA by comparing prevalence rates obtained using scales to those obtained using more formal diagnostic interviews. When these two methods were compared, investigators found that 7% of PD patients met criteria for PBA when assessed with formal interviews, whereas 42.5% met criteria for PBA when using the CNS-LS at a threshold of 13 [25]. This variability speaks to the need for a standardized and crossculturally validated method of assessing PBA in patients with parkinsonian disorders. This study also found that depressive symptoms were highly correlated with higher CNS-LS scores, but not with a formal diagnosis of PBA, suggesting that the CNS-LS may not be able to effectively discriminate depression from PBA in patients with parkinsonian disorders.
A number of demographic and clinical characteristics have been correlated with the increased prevalence and/or severity of PBA symptoms in patients with parkinsonian disorders. PD patients with more advanced disease have been demonstrated to exhibit an increased prevalence of PBA [20,31], while younger PD patients have been shown to exhibit greater PBA symptom severity [28]. These factors need to be accounted for when examining PBA prevalence given that they may contribute to between-study variability.
Interestingly, patients with movement disorders and PBA seem to suffer more frequently from pseudobulbar crying than pseudobulbar laughter or both laughter and crying [20,29,31]. One study reported that among 37 movement disorders patients with PBA, 75.7% had pathological crying, 13.5% had pathological laughter, and 10.8% had both [20]. Another study found that among the 22 PD patients identified with PBA, all had pathological crying and none exhibited pathological laughter [31]. An increased frequency of pseudobulbar crying relative to pseudobulbar laughter has also been reported in ALS [32,33] and MS [27].
PBA IN POSTSURGICAL PD PATIENTS
Several case reports have described the onset or worsening of PBA symptoms in patients with PD after deep brain stimulation (DBS) or gamma knife thalamotomy [8,34].
Pseudobulbar laughter has been described as an “off” levodopa/“on” stimulation state after DBS of the subthalamic nucleus (STN-DBS) [8]. In one case, pseudobulbar laughter present in the “off” medication state was exacerbated during STN monopolar review, regardless of which contact was stimulated. Pseudobulbar crying has also been seen following STN-DBS [22,35]. This symptom was described in a 46-year-old woman with a prior history of left pallidotomy who subsequently underwent unilateral left STN-DBS. Monopolar stimulation at all contacts resulted in uncontrollable crying in the absence of changes in mood [22]. Another case report described pseudobulbar crying in a 48-year-old woman with PD who underwent bilateral STN-DBS. Pseudobulbar crying was observed in the “off” levodopa state during monopolar review of the distal-most contacts on the left, located in the region of the caudal internal capsule. Again, this phenomenon was not associated with changes in mood [35]. Pseudobulbar laughter has been reported six months after gamma knife thalamotomy targeting the right ventral intermedius nucleus in a patient with a history of tremor predominant PD; this symptom resolved within 48 hours of treatment with sertraline [21].
It is important to not overly interpret this literature, given the lack of large-scale studies confirming the findings of these case studies. Nevertheless, the mechanism by which neurosurgical procedures such as DBS can trigger PBA remains intriguing. The medial STN is known to interface with limbic networks, and it is possible that stimulation of these networks may play a role in triggering PBA. Stimulation of fiber tracts surrounding the STN, including the caudal zona incerta, caudal internal capsule and prelemniscal fibers, may also potentially contribute to PBA via their modulation of brainstem bulbar tracts regulating motor expression of laughter and crying. Tractography studies may be able to better elucidate the circuitry by which STN-DBS contributes to symptoms of PBA.
IMPACT OF PBA ON QUALITY OF LIFE IN PD
While the symptoms of PBA may appear physically harmless, they can be emotionally taxing and have a significant negative impact on the patient and caregiver QoL and on social and occupational functioning [1,9]. The PRISM study estimated both the prevalence of PBA symptoms and the impact of PBA on QoL in a sample of patients with various neurological conditions, including PD [1]. Using the CNS-LS to define PBA, the investigators found that patients with PBA reported significantly worse QoL compared with patients without PBA. Furthermore, they found that patients with more prominent PBA symptoms reported worse QoL and greater use of antipsychotics and antidepressants.
A study including 269 movement disorders patients, including 168 patients with PD, found that patients with PBA, as defined by the CNS-LS, exhibited more impaired social well-being and greater depression, as indexed by the Parkinson’s Disease Questionnaire social well-being subscale and the Beck Depression Inventory, respectively [29]. Another study including 719 patients with movement disorders, including 387 PD patients, similarly found significantly higher depression scores in patients with PBA, and a trend towards impaired social well-being in PD patients with PBA [20]. Another study found that greater PBA symptom severity correlated with greater anxiety, greater depression, and reduced overall QoL in PD patients [28]. PBA has also been associated with changes in living status, i.e., being moved to supervised living and to reduced caregiver work productivity [9]. These studies highlight the significant negative impact of PBA on patients’ and caregivers’ daily life experiences.
TREATMENT OF PBA IN PARKINSONIAN DISORDERS
Despite its relatively high prevalence in patients with neurological diseases, the recognition and diagnosis of PBA in the community remains subpar. A large epidemiological survey including over 8,500 patients assessed for PBA using current diagnostic tools found that none of the patients who screened positive for PBA and discussed their symptoms of involuntary crying and/or laughing with a physician were diagnosed with PBA. Only 41% of patients ultimately received a diagnosis, which was most often depression [7]. This failure to appropriately diagnose patients unfortunately can translate into a failure to properly treat these patients. The treatment agents currently available for PBA are described below.
SSRIs, SNRIs, and TCAs
Historically, off-label treatment for PBA has included antidepressants including SSRIs, selective serotonin and norepinephrine reuptake inhibitors (SNRIs), and tricyclic antidepressants (TCAs) [4]. Support for the use of such therapies has come from numerous case studies, open label trials, and case control trials demonstrating rapid resolution of PBA symptoms with drug administration [4,11,19,30,36-39]. The large majority of these trials, however, have been performed in patients with MS, ALS, and stroke. The only support for the use of such medications in patients with parkinsonian disorders comes from case reports in PD patients with PBA secondary to STN-DBS or thalamotomy and in patients with MSA-C [4,5,21,22]. Experimental evidence in the form of clinical trials supporting the efficacy of these medications in treating PBA in patients with parkinsonian disorders is lacking.
Given that many patients with parkinsonian disorders suffer from depression, the use of antidepressants may be beneficial as monotherapy in treating PBA and comorbid depression in patients with parkinsonian disorders [28]. Intriguingly, however, several studies have found that movement disorders patients taking antidepressants exhibit a higher prevalence of PBA compared to patients not on antidepressants [20,29]. This association between PBA and antidepressant use can be interpreted in several different ways. It has been suggested that this correlation serves as evidence that antidepressants may in fact paradoxically promote PBA symptomatology. Alternatively, it is possible that patients with PBA are more likely to have a comorbid depression, for which they are being treated with antidepressant medication. Given that the indication for antidepressant use was not specified in these studies, it is also possible that the patients were on antidepressant medication for treatment of PBA. Additional work is needed to distinguish between these possibilities.
Dextromethorphan/quinidine
DMQ, currently the only FDA-approved treatment for PBA, was specifically designed for treatment of PBA [23,24]. While its exact mechanism is unknown, dextromethorphan is thought to function by exerting anti-glutamatergic effects at NMDA and σ-1 receptors [40,41]. Dextromethorphan binds most avidly in the brainstem and cerebellum, and its therapeutic effects may stem from its modulation of neurotransmission within cortico-pontocerebellar circuits thought to be integral to the pathophysiology of PBA [4]. While the main therapeutic component of DMQ is dextromethorphan, quinidine acts to inhibit the CYP450 enzyme CYP2D6, which rapidly metabolizes dextromethorphan, thereby increasing the plasma concentration of dextromethorphan and allowing for lower dosages of the drug to be used [4,23,24,41-43].
Similar to evidence supporting the use of SSRIs and TCAs in the treatment of PBA, clinical trials on DMQ have primarily focused on the drug’s efficacy and safety in patients with ALS, MS, AD, stroke and TBI [40-42,44]. In the Safety, Tolerability, and Efficacy Results of AVP-923 in the PBA (STAR) randomized, placebo-controlled trial, investigators found that MS and ALS patients with PBA who were given DMQ at doses of 20/10 mg or 30/10 mg twice daily showed significant improvement in PBA symptoms both in terms of number of PBA episodes and symptom severity over a 12-week period [42]. The Pseudobulbar Affect Registry Investigating Symptom Management II (PRISM II) trial, an open-label trial testing DMQ in patients with dementia, stroke, and TBI, found that patients receiving DMQ at 20/10 mg twice daily experienced significant improvements in their PBA symptoms [44]. Both studies found that DMQ is well-tolerated in patients with MS, ALS, dementia, stroke, and TBI, with most adverse events being transient and mild or moderate in severity, including dizziness, nausea, headache, fatigue, and diarrhea [23,24,42,44]. One open-label multicenter trial investigating the safety of DMQ in treating PBA in patients with a number of neurological conditions, including a small sample of patients with PD (n = 11) and atypical parkinsonian disorders (n = 7), found that taking 30/30 mg of DMQ twice daily over a 52-week period was safe, with the most common treatment-related side effects in parkinsonian patients being nausea (4.3%), dizziness (21.7%), headache (21.7%), somnolence (13.0%), fatigue (4.3%), and dry mouth (8.7%) [45]. In accordance with previous studies, they found that the adverse events were mild to moderate in severity. At the present time, there are no studies exploring the efficacy of DMQ in treating PBA in parkinsonian disorders. Table 2 provides a summary of the limited studies investigating treatments for PBA in patients with parkinsonian disorders.
CONCLUSIONS
PBA is a distressing neurological condition that is relatively common but underappreciated in the parkinsonian population. While various studies have attempted to quantify the prevalence of PBA in parkinsonian disorders, there is great variability in the estimates between studies, emphasizing the need for standardized and cross-culturally validated methods of assessing PBA in patients with parkinsonian disorders, not only for research purposes but for clinical purposes, as well. Furthermore, while the gold-standard treatments for PBA, which include SSRIs, TCAs, and DMQ, have been investigated in neurological conditions, including ALS, MS and stroke, evidence-based medicine for these treatments in parkinsonian disorders is lacking. Greater attention to and exploration of PBA in patients with parkinsonian disorders is urgently needed.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.