Asymptomatic Hearing Impairment Frequently Occurs in Early-Onset Parkinson’s Disease
Article information
Abstract
Objective
Recent reports of hearing impairment in Parkinson’s disease (PD) have suggested that auditory dysfunction could be a non-motor manifestation of PD. These reports were based on observations of elderly patients for whom presbycusis may, to some extent, have contributed to hearing dysfunction. Therefore, we aimed to explore the auditory functions in younger patients with PD.
Methods
We conducted a case-control study in a relatively younger (< 55 years of age at study time) population of PD patients and healthy volunteers to test whether auditory dysfunction is a significant non-motor dysfunction in PD. Pure tone audiometry (PTA) and brainstem evoked response audiometry (BERA) were performed in all participants.
Results
None of the patients or controls reported hearing deficits. Fifty-one patients with PD and 50 healthy volunteers who were age- and gender-matched to the patients participated. PTA-detected hearing impairment was found in 64.7% of patients and 28% of controls (p < 0.001) for both low-mid and/or high frequencies. Hearing impairment was more frequent in the younger subgroups of patients than age-matched controls, while the frequency of hearing impairment was similar in older groups of subjects. BERA was not different between patients and controls.
Conclusion
Asymptomatic auditory dysfunction is a common non-motor manifestation of early-onset PD and more frequent in younger patients, indicating that it may be independent of aging. The mechanism underlying this dysfunction appears to be peripheral, although a central dysfunction cannot be ruled out based on the findings of this study.
Parkinson’s disease (PD) is currently recognized as a multisystem neurodegenerative disorder with protean non-motor manifestations [1-3]. The non-motor dysfunctions in PD encompass multiple domains, including cognitive, neuropsychiatric, autonomic and sensory [4-7]. Though non-motor symptoms (NMS) contribute to morbidity and reduced quality of life for patients with PD, they are often underreported by patients and underrecognized by clinicians compared with motor symptoms [8]. A proper assessment of the burden of NMS requires focused clinical evaluations. Therefore, instruments such as the NMS questionnaire (NMS Quest) and non-motor symptoms scale (NMSS) were developed for this purpose [9,10]. The evaluation and management of NMS is currently part of the standard of care for PD patients.
Sensory dysfunction is one of the important domains of nonmotor PD [6]. The instruments used for the evaluation of NMS in PD focus primarily on somatic sensations such as pain and paraesthesias [9,10]. PD is known to affect the nervous system at all levels, from the enteric nervous system to the cortex. Auditory dysfunction as a manifestation of PD has received only scanty attention from researchers, probably because hearing impairment is not a common complaint of PD patients [11]. Auditory dysfunction has been shown to be present in other neurodegenerative diseases including Alzheimer’s disease, multiple system atrophy and Huntington’s disease [12-15] as well as essential tremor (ET) [16]. Demonstration of alpha synuclein in the auditory pathway (outer hair cells of the cochlea) in animal studies indicate that hearing may not be spared in PD [17,18]. This hypothesis was supported by a few recent studies in which PD patients were assessed using pure tone audiometry (PTA) and brainstem evoked response audiometry (BERA) [11,19-21]. These studies reported auditory dysfunction in PD patients using PTA, although the results of BERA were not consistent across the studies [11,15,20,22-24]. The NMS of PD differ from symptoms reported in normal aging [7]. As PD is a disease of aging, age-related changes in sensorineural functions (presbycusis) could be a confounding factor when addressing auditory dysfunction in PD [25]. In this study, we investigated auditory functions in a subset of relatively young (less than 55 years at assessment) patients and healthy, age-matched controls to explore whether hearing impairment is a non-motor manifestation of PD in younger patients for whom the effects of presbycusis are minimal.
MATERIALS & METHODS
We recruited consecutive patients with PD who were diagnosed using the United Kingdom Parkinson Disease Society Brain Bank criteria [26]. All patients had early-onset PD (age of onset ≤ 50 years) [27] and were aged ≤ 55 years at the time of recruitment. The healthy volunteers were age- and gender-matched subjects. Patients and healthy controls with known otological diseases or a conductive pattern of hearing loss in audiometry, a family history of deafness or concomitant neurological or other medical illnesses known to negatively affect hearing were excluded. Patients with significant cognitive impairment [diagnosed by clinical history and the Montreal Cognitive Assessment (MoCA) scores] or neuropsychiatric symptoms were also excluded. The study protocol was reviewed and approved by the Institutional Ethics Committee (IEC approval number: SCT/IEC/719) and all subjects gave written informed consent.
All participating patients were assessed using the Unified Parkinson’s Disease Rating Scale (UPDRS) Parts I–IV, the Hoehn & Yahr (H&Y) staging scale, the NMS Quest [9] and the NMSS [10]. A basic otological examination was performed on all subjects to rule out abnormalities in the ear canal. Healthy volunteers were interviewed and examined to ensure that they had no signs of neurological disorders and had normal cognition.
PTA and BERA
All subjects underwent PTA and BERA. For PTA, acoustic stimulation was done according to the standard protocol at frequencies of 0.25, 0.5, 1, 2, 4, and 8 kHz. Both air and bone conductions were assessed. Hearing loss was classified according to the WHO classification scheme and grouped into the following five grades: hearing threshold 0–25 dB, normal; 26–40 dB, mild hearing loss; 41–60 dB, moderate hearing loss; 61–80 dB, severe hearing loss; and ≥ 81 dB, profound hearing loss [28,29]. Hearing loss was also divided into either low-mid frequency hearing loss (0.25–2 kHz) and high frequency hearing loss (≥ 4 kHz) based on the testing frequency at which it was evident [11]. BERA was obtained by stimulating each ear at 60 dB above the hearing threshold while masking the contralateral ear with noise 40 dB lower than that used for the stimulation. The latencies of waves I, II, III, IV, and V and the interpeak latencies I–III, III–V, and I–V were recorded in milliseconds from both ears. Otological investigations were performed in an audiometry lab by an experienced audiologist and in the ON stage in patients experiencing motor fluctuations.
Statistical analysis
Fisher’s exact tests/chi-square tests were used to compare the prevalence of hearing impairment between patients and controls and to investigate the association of hearing impairment with other categorical variables in the patient and control groups. Continuous variables were compared using Student’s t-test. Odds ratios (ORs) and 95% confidence intervals were calculated using binary logistical regression. The statistical analysis was performed using IBM SPSS Statistics 21 (IBM Corp., Armonk, NY, USA).
RESULTS
Fifty-five patients with PD and an equal number of healthy controls were recruited for the study. Four patients and five healthy volunteers had a conductive pattern of hearing loss and were excluded from further analysis. The data from the remaining subjects (51 patients and 50 healthy volunteers) were included for final analysis.
The baseline demographic and clinical characteristics of the participating subjects are shown in Table 1. The patients and controls did not differ with regard to age and gender. The frequencies of vascular risk factors were also similar in the two groups [PD vs. controls: diabetes, 2% vs. 4% (p = 0.62); hypertension, 6% vs. 4% (p = 1.00)]. Two patients (3.9%) were in H&Y stage 1, one patient (1.9%) was in stage 1.5, 17 (33.3%) were in stage 2, 23 (45%) were in stage 2.5 and eight (15.6%) were in stage 3. Symptom onset was right sided in 70.5% of the patients. Motor fluctuations were present in 31 patients, while 22 experienced dyskinesia. At least one NMS (detected by NMS Quest/NMSS) was present in 43 (84.3%) of the patients. The most common NMS domains affected included mood in 24 (47%) of patients, sleep disorders/fatigue in 23 (45%) of patients, “miscellaneous” (which includes hyposmia/paresthesia) in 10 (19.6%) patients, gastrointestinal disturbance in nine (17.6%) patients, the domains of urinary disturbances, sexual dysfunction and memory dysfunction/inattention with three patients in each (5.9%) and hallucinations in one (2%) patient. None of the patients reported NMS in the cardiovascular domain of the questionnaires. Positive family history (having at least one first degree relative diagnosed with PD) was present in eight of the patients. The levodopa equivalent daily dosage [30] is shown in Table 1. All of the patients were being treated at the time of the assessments. There were 38 patients taking dopamine agonists (pramipexole or ropinirole; 28 of these patients were on levodopa as well) and 41 were on levodopa. Other medications taken by the patients include amantadine in 17, anticholinergics in nine, rasagiline in 11 and entacapone in 12 patients.
Audiometry data
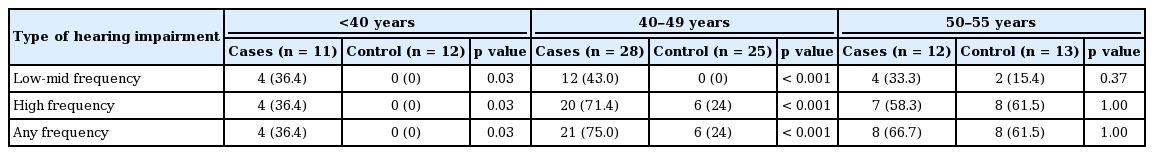
The frequency of hearing impairment among cases and controls is presented in Table 2. Hearing was considered impaired if the subject had an abnormal hearing threshold in at least one ear. Overall, hearing was abnormal (either at low-mid or at high frequencies or both) in 33 (64.7%) patients and 14 (28%) controls (p < 0.001). Hearing impairment was significantly more common in patients compared to controls at low-mid frequencies as well as at high frequencies (p < 0.001) (Table 2). The OR was higher for low-mid frequency hearing loss when compared to high frequency hearing loss. None of the affected subjects were aware of their hearing impairment. There were 6 patients and 2 controls with low-mid frequency hearing loss with bilateral involvement. The remaining patients experienced unilateral involvement. Among the subjects with hearing loss at high frequency stimulation, 15 patients and 9 controls had bilateral involvement. For any type of hearing loss, the involvement was bilateral in 16 patients (31.3%) and 7 controls (14%). For cases with low-mid frequency hearing loss, the deficit was mild in 19 and moderate in 1 patient. Both of the controls affected with low-mid frequency hearing loss had mild deficits only. The high frequency hearing impairment detected was mild in 21 patients, moderate in 9, and severe in 1. For controls, the high frequency hearing impairment was mild in 9 patients, moderate in 5 and severe in none.
Hearing impairment across different age groups
Table 3 shows a comparison of the frequency of hearing impairment when the subjects were stratified into three age groups. Hearing impairment was more frequent in younger PD patients when compared to their age-matched healthy controls but similar to controls in older PD patients.
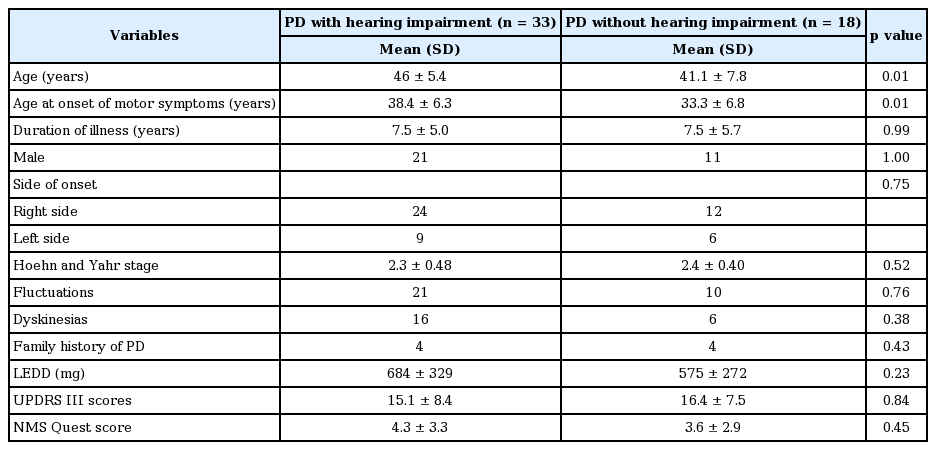
Characteristics of patients with hearing impairment in PTA
The demographic and clinical characteristics of patients with and without hearing impairment in PTA were compared (Table 4). Though the hearing-impaired group was older at the time of the study and had a later age of PD onset, no significant difference was noted in the prevalence of hearing impairment when the patients were divided into two groups based on the age of onset after taking 40 years-of-age as the cut-off value. Seventeen (58.6%) of the 29 patients with an age of onset of PD of less than 40 years had hearing impairment while 16 (72.7%) of the 22 with a higher age of onset were affected (p = 0.30). There was no significant difference among the groups when the analysis was done separately for low-mid frequency hearing loss (44.8% vs. 31.8%; p = 0.35) and high frequency hearing loss (55.2% vs. 72.7%; p = 0.20). There was no relation between hearing impairment and severity of motor or non-motor dysfunction, as measured by UPDRS III scores in the OFF state, H&Y stage or NMSS scores.
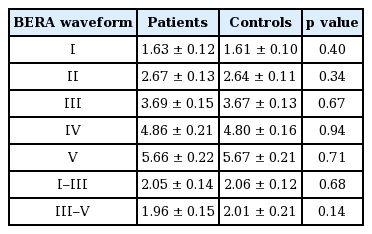
BERA results
BERA was recorded in all study participants. The mean peak latencies and interpeak latencies are shown in Table 5. There was no difference between cases and controls in any of the latencies.
DISCUSSION
In this case-control study of auditory functions in a relatively younger population of patients with PD, PTA-identified hearing impairment was more frequent in patients than in age- and gender-matched healthy controls. This dynamic was observed for both low-mid frequency and high frequency ranges and the difference with controls was more striking in patients who were younger at the time of assessment. Patients with hearing loss were older at the time of the study and had reached an older age at disease onset when compared to those without hearing loss. Although we found abnormalities in PTA, none of the subjects reported any hearing impairment. BERA did not differ between patients and healthy controls.
In spite of having abnormalities in PTA, none of our subjects reported having any hearing impairment as a symptom. Previous questionnaire-based studies have found that PD patients do not report increased occurrences of hearing impairment as a symptom compared to healthy controls. Ondo et al. [19] used a 10-point questionnaire to compare patients with ET and PD with healthy controls for hearing impairment; they failed to find any differences in the PD group. A study on 106 PD patients and healthy controls found abnormal PTA in 86.8% of patients and 56.6% controls, but none reported hearing impairment [11]. The reasons for the lack of reporting of hearing deficits in PD might be due to the chronic nature of the problem along with possible inattention and cognitive changes associated with PD, although detailed cognitive testing apart from screening using MoCA was not done in our patients. The hearing deficits may have been underreported because hearing loss was mild or moderate in the majority of patients. Most of our patients were in the early/mid stages of PD, therefore longitudinal studies are needed to explore whether hearing impairment worsens to functionally significant levels as PD progresses to later stages.
We used the WHO classification system for grading hearing impairment [28,29] and found that hearing impairment, when compared to healthy controls, was more frequent in PD patients younger than 50 years of age. It is possible that in older individuals, presbycusis and other otological pathologies overshadow the effects of PD and we were unable to pick up the smaller differences between patients and controls due to our small sample size for the subgroup analysis. Most of the previous studies that have reported audiological abnormalities in PD included patients who were elderly. The mean age of patients with PD in a study by Yýlmaz et al. [20] was approximately 70 years and, in a Vitale et al. [11] study, it was approximately 64 years. To the best of our knowledge ours is the first study reporting hearing involvement in a relatively younger cohort of PD patients (all of our patients were less than 55 years of age at the time of assessment, with a mean age of 44.2 years) for whom the effects of presbycusis was minimal and would not mask the effects of PD on hearing ability. It is known that the prevalence of presbycusis starts to rise after middle age, with a steep rise after the 6th decade [31]. Our findings also support this as we found a sharp increase in the occurrence of hearing loss in controls aged above 50 years. This increase could have masked any difference in hearing due to PD in the older groups. We found that hearing impairment in our patients involved both low-mid and high frequencies while a previous study in older patients found a predominance of high frequency hearing involvement [11]. In spite of having impairment for both low-mid and high frequencies, the OR was higher for low-mid frequencies in our patients. We believe this finding is interesting, as presbycusis tends to preferentially affect high frequencies [31,32]. Alpha-synuclein has been demonstrated to be present in the inner ear and synucleinopathy could facilitate noise-induced presbycusis in older patients with PD [11,17], while younger patients may have a mechanism independent of presbycusis exaggeration. However, we admit the limitations due to our relatively small sample size and suggest studies be conducted with larger sample sizes to explore this hypothesis. The hearing impairment was also mild or moderate for the majority of patients and only a single patient had severe hearing loss. This is similar to patterns seen in earlier studies [11,20].
Even though PD patients with hearing loss in our study population were older with an older age at onset when compared to those without hearing loss (Table 4), there was no significant difference in the prevalence of hearing impairment between the two groups when patients were dichotomized into youngonset PD and those with an older age of PD onset. This indicates that it is probably a true NMS related to PD pathology, irrespective of age of onset or genetic status. Young-onset PD is known to have a more frequent and stronger genetic contribution in its pathogenesis [27].
There was no relationship with the duration or severity of motor symptoms, which was not surprising considering that auditory involvement could be peripheral and asymptomatic and could, therefore, have predated the motor symptoms, as hyposmia does [33]. The study sample size was insufficient to examine this association with different H&Y stages of PD to explore whether auditory dysfunction correlates with severity of motor dysfunction. All of our audiological evaluations were done in the ON state for ensuring patient comfort during the examination and achieve the maximum possible level of attention and cooperation. We did not explore the effects of levodopa on the audiological abnormalities in PD; however, other investigators have shown that levodopa does not correct the PTA abnormalities in PD [23,34].
We found that BERA was not abnormal in PD patients when compared to age-matched healthy controls. This is similar to earlier reports by others, which suggest sparing of central pathways [11,15]. These results are in contrast to observations from other reports of prolonged interpeak latencies of waves III and V, and prolonged latencies of wave V and I–V interpeak latencies [20,23]. We acknowledge the possibility that the relatively high stimulus intensity (60 dB) above the hearing threshold used for BERA might have overcome abnormalities in the central pathways. This is known to be a cause for normal BERA results in subjects with hearing impairment [35]. Alpha-synuclein has been demonstrated in the outer hair cells of the inner ear in animal studies, but whether misfolded alpha-synuclein deposition in the outer hair cells leads to dysfunction of the auditory receptor apparatus is still unknown [17,18]. A central mechanism cannot be ruled out based on the BERA due to its limitations as discussed above. Some recent studies suggest impairment of auditory processing in PD [25]. Further studies using techniques such as otoacoustic emission to assess cochlear function, and fMRI to assess central auditory processing may provide more clarity on this topic.
PD, particularly young-onset and genetic forms such as those with Parkin and PINK-1 mutations, are known to be associated with a significant degree of mitochondrial dysfunction. Interestingly, disorders of mitochondrial function are also known to have hearing impairment as a manifestation [36]. Pending studies showing neuropathological changes in relevant areas of the nervous system (unlike the other major, non-motor manifestations of PD including REM-sleep behavior disorder, constipation and anosmia, which are supported by neuropathological changes in relevant areas of the nervous system in those with PD) and larger clinical studies characterizing the nature of hearing impairment, we admit that at this stage, we can only suggest the possibility that hearing impairment could be another nonmotor manifestation of PD. Studies in subgroups such as early PD and prodromal PD, and longitudinal follow-up studies are needed to answer questions on whether hearing impairment precedes motor manifestations of PD, has any role in predicting the future occurrence of PD in those without motor symptoms, or a role in predicting the progression of motor signs [21].
We acknowledge the limitations of our study posed by the relatively small sample size, which precluded the analysis of hearing functions in subgroups of patients in different stages of the disease, or different motor phenotypes such as tremor-dominant and postural instability and gait-dominant subtypes. The study was primarily aimed at exploring whether hearing impairment, as a non-motor manifestation, is present in younger PD patients for whom the effects of presbycusis are minimal. Thus, the study design permitted the inclusion of only early-onset PD patients, who constitute a heterogenous group with regard to genetic etiological factors, pathogenesis and progression [27]. This is another limitation. We did not conduct a longitudinal followup assessment to examine whether hearing loss becomes more prominent and clinically significant with disease progression. Studies with larger sample sizes and longitudinal follow-ups are needed to confirm our findings and to establish hearing loss as a non-motor manifestation of PD. Despite these limitations, to the best of our knowledge, this is the first study to report PTA-confirmed hearing impairment in early onset PD, adding strength to earlier, similar observations in older PD patients which could have been confounded to some extent by presbycusis. We conclude that asymptomatic auditory dysfunction is a relatively common non-motor dysfunction seen in PD patients who are relatively younger and is similar to hearing impairments demonstrated earlier in unselected groups of patients with PD by other investigators. Larger clinical and neuropathological studies may establish hearing impairment as a true nonmotor manifestation of PD.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.
Acknowledgements
In-house project of the Comprehensive Care Centre for Movement Disorders, Sree Chitra Tirunal Institute for Medical Sciences and Technology, Thiruvananthapuram, Kerala, India.