Recent Advances in the Development of Experimental Therapeutics for Levodopa-Induced Dyskinesia
Article information
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disease worldwide, with an estimated prevalence of approximately 1% in people over 60, and it represents an increasingly important medical problem in our aging population [1]. For decades, the standard of care for PD has involved treatment with levodopa (L-DOPA), which elevates dopamine levels in the nigrostriatal pathway, enhancing movement and coordinated motor functions. Chronic L-DOPA results in motor complications, including levodopa-induced dyskinesia (LID), which occur in at least 50% of patients after 5 to 10 years of treatment. LID is a significant limitation to the viability of long-term L-DOPA use because patient function and quality of life are compromised and individual and societal costs are increased [2].
Although treating LID in PD patients can be challenging, significant progress has been made in the past few years. Experimental treatments are making their way through preclinical and clinical stages. This review will focus on data that demonstrate the rationale, safety, efficacy, and long-term benefits of these medical treatments in both animal models of PD and PD patients suffering from LID.
SEROTONIN RECEPTORS
Serotonergic neurons are a significant source of dopamine release in striatal synapses and possess the enzymes necessary to convert L-DOPA into dopamine [3]. When L-DOPA is administered to PD patients, synaptic dopamine levels oscillate as serotonergic neurons metabolize L-DOPA to dopamine but fail to autoregulate in response to elevated dopamine levels (Figure 1) [4]. PET studies have confirmed that L-DOPA produces higher synaptic dopamine levels in dyskinetic PD patients than in non-dyskinetic patients [5]. This loss of the ability to buffer excess synaptic dopamine is a proposed mechanism for LID development and may explain why LID is more prevalent when L-DOPA is administered orally than with continuous duodenal infusion.
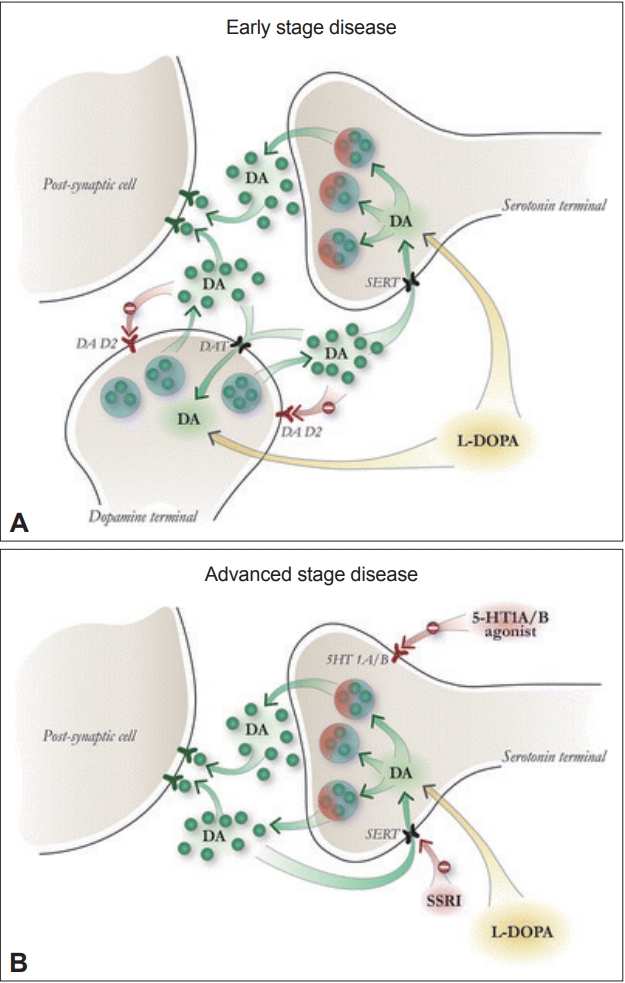
(A) At the early disease stage, spared dopaminergic terminals retain the ability to modulate and fine-tune dopamine release and storage with the help of dopamine D2 autoreceptors and dopamine transporters. Serotonergic neurons also contribute to dopamine release; however, the retained buffering capacity of spared dopaminergic neurons prevents excessive levels of synaptic dopamine. (B) In more advanced stages of disease, more significant destruction of dopaminergic terminals results in a loss of buffering capacity and a lack of D2 autoreceptor-mediated feedback. Because serotonergic neurons lack feedback mechanisms for dopamine levels, they play a much more significant role in converting levodopa (L-DOPA) to dopamine, prompting greater dysregulated release of dopamine when L-DOPA is administered to the patient. Reprinted with permission from Carta M and Björklund A [4]. DA: dopamine, DAT: dopamine transporter, D2: dopamine D2 receptor, 5-HT1A/B : serotonin 1A and 1B receptor, SERT: serotonin transporter, SSRI: selective serotonin reuptake inhibitor.
Pharmacological validation of this framework has been provided by several preclinical experiments. The administration of a selective 5-HT1 agonist to dampen serotonin neuron-derived dopamine release by autoreceptor stimulation significantly reduced LID in animals [6]. A synergistic effect on LID reduction was observed when 5-HT1A and 5-HT1B agonists were coadministered in rodent and primate [6] PD models. Selective serotonin reuptake inhibitors (SSRIs), including fluoxetine and citalopram, completely attenuated LID when they were administered to rodent models of PD, and this effect was blocked by the coadministration of a 5-HT1 antagonist [7]. Unlike 5-HT1 agonists, SSRIs do not reduce L-DOPA efficacy, suggesting that the temporarily reduced serotonin synaptic output from agonist-stimulated 5-HT1 autoreceptors may be responsible. This finding has additional clinical relevance when one considers that PD patients often experience depressive symptoms alongside motor fluctuations [8]. SSRIs may therefore be especially useful in these situations.
Until recently, there was a dearth of clinical evidence supporting the idea that serotonergic neurons represent a viable therapeutic target for LID. Previously, a double-blinded, multicenter, open-label study found no improvement in LID in PD patients treated with the partial 5-HT1A agonist sarizotan [9,10]. This lack of efficacy was speculated to be due to the fact that sarizotan only selectively agonizes the 5-HT1A receptor and that the synergistic effect of 5-HT1B on LID reduction was not utilized. Promising results have emerged from a recent phase I/IIa randomized controlled trial for the mixed 5-HT1A/B agonist eltoprazine. Eltoprazine (5 and 7.5 mg) significantly reduced dyskinetic symptoms, as measured by both the Clinical Dyskinesia Rating Scale and the Rush Dyskinesia Rating Scale [11]. Importantly, Unified Parkinson’s Disease Rating Scale III scores did not differ significantly between the treatment and control groups, and patients treated with eltoprazine did not experience any decline in the therapeutic efficacy of subsequently administered L-DOPA. While the effects of long-term treatment with eltoprazine have not yet been established, early studies have shown very promising results, and further studies are underway to establish optimal dosing regimens in which the drug is delivered over extended periods of time.
Together, preclinical and clinical data have demonstrated exciting advancements in understanding how perturbations in neurotransmitter systems can manifest as LID. These findings justify further studies with additional pharmacological agents that modulate the serotonergic system for the purpose of reducing LID.
OPIOID RECEPTOR MODULATORS
In 2017, AT-390 and AT-403, two chemically distinct agonists of the nociceptin/orphanin FQ opioid peptide (NOP) receptor, were discovered [12]. These compounds were examined based on previous findings that NOP receptor agonists have the potential to decrease LID symptoms in animal models of PD [13]. Both compounds improved akinetic and dyskinetic symptoms in rodents, likely through an extracellular signal-regulated kinase (ERK)-related signaling mechanism and have the potential to be useful tools for further probing the role of NOP receptors in LID pathophysiology.
Another recent pharmacological approach for attenuating LID involves targeting delta and mu opioid receptors. The stimulation of the delta opioid receptor improves PD symptoms in animals [14], while antagonizing mu opioid receptors diminishes LID symptoms [15]. A novel compound, termed DPI-289, was designed as a combined delta opioid receptor agonist/mu opioid receptor antagonist [16] and possesses antiparkinsonian and antidyskinetic properties in rodent and primate PD models. As a monotherapy in PD animal models, this compound exhibited significant, though limited, antiparkinsonian effects without any associated dyskinesia. In contrast, when DPI-289 was coadministered with L-DOPA, which is known to produce LID in these animals, a significant increase in anti-parkinsonian effects was observed, but it still did not produce any significant dyskinetic symptoms. These preclinical findings suggest that a drug with this polypharmacological profile may serve as a useful adjunctive therapy to traditional dopaminergic therapy for PD to alleviate motor deficits without eliciting associated LID symptoms.
LIPOIC ACID
Previous studies have shown that there is enhanced oxidative stress in the brain tissue of patients treated with L-DOPA and that this may be the result of decreased levels of antioxidants, the excessive oxidation of dopamine, and disruptions in the mitochondrial transport chain [17]. Additional studies have found that PD patients treated with L-DOPA have increased plasma levels of neuroinflammation markers, including oxidized-low density lipoproteins and soluble intracellular adhesion molecule [18]. Collectively, the literature suggests that monitoring oxidative stress markers and inflammatory factors may be useful for PD patients undergoing treatment with L-DOPA [19].
Previous work has indicated that α-lipoic acid (ALA) can scavenge reactive oxygen species (ROS) and inhibit free radical generation via metal chelation [20]. These antioxidative processes suggest that ALA treatment may be useful for various disorders in which oxidative stress is proposed to play a role. Furthermore, ALA may also possess anti-inflammatory properties and may promote increases in intracellular glutathione formation, which has been postulated to be beneficial in neurodegenerative diseases [21]. In fact, previous studies have shown that ALA treatment attenuates ROS formation, resulting in neuroprotective effects on dopaminergic neurons against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced apoptosis [22].
Based on these purported pathophysiological processes and the proposed mechanism of action of ALA, a recent study supported the hypothesis that ALA may serve as a useful diseasemodifying therapy and attenuate LID when coadministered with L-DOPA in an animal model of PD [19]. This study by Zhang et al. [19] found that cotreatment had dose-dependent antidyskinetic effects. Molecular biomarkers and metabolites have been profiled in the substantia nigra, revealing that the antidegenerative effects of ALA likely stem from its ability to reduce oxidative stress and apoptosis in this neuronal cell population [19]. ALA may be a promising disease-modifying therapy that slows the loss of dopaminergic neurons in patients with early-stage PD and could allow for earlier incorporation of L-DOPA into medical regimens to significantly slow symptomatic disease progression while also delaying LID onset.
GLUTAMATERGIC RECEPTOR MODULATORS
It is hypothesized that the inhibition of glutamatergic N-methyl-D-aspartate (NMDA) receptors may be beneficial in PD since central dopamine depletion in the nigrostriatal pathway results in excessive glutamatergic activity in the basal ganglia [23]. Animal studies have indicated that selectively inhibiting a specific subset of NMDA receptors containing the NMDA Receptor 2B subunit (NR2B) subunit elicits a therapeutic effect and may potentiate the positive effects observed with dopaminergic therapy [24].
Promising results have been observed in preclinical studies, but human studies have shown mixed results. A recent phase Ib randomized, double-blind, controlled crossover study was conducted to investigate the efficacy of a single dose of MK-0657, an NMDA receptor antagonist, against the motor symptoms of LID and PD [25]. The study found that MK-0657 did not significantly improve dyskinesia or motor function over a 5-hour period. Earlier studies conducted to assess the efficacy of CP-101,606, another NR2B-selective NMDA antagonist, however, did find significant beneficial effects on the motor symptoms of both LID [26] and PD [24,26].
There may be a number of pharmacodynamic reasons for the inconsistencies among clinical trial results regarding different NR2B-selective NMDA antagonists. The research community still considers the NR2B subunit and other glutamatergic receptors to be valuable targets for LID drug discovery, given the robust findings implicating their pathological functions in key motor areas in PD patients. Several preclinical studies other modulators of metabotropic glutamate receptors for LID and PD treatment are still underway [27,28].
β-ARRESTINS
Dopamine receptors are classified as G protein-coupled receptors (GPCRs), a superfamily of proteins that are targeted by approximately 25% of currently approved drugs [29]. A recent breakthrough in the GPCR pharmacology field is the ability to design drugs with functional selectivity, or signaling bias, whereby the GPCR ligand can selectively activate either G protein- or β-arrestin-mediated downstream signaling pathways (Figure 2) [30]. Functional selectivity offers the possibility to create drugs that can maintain the therapeutic benefits of their targets while dramatically decreasing the adverse side effect profiles that often come with targeting certain GPCRs [31]. One particularly promising application of this principle involves the possibility of treating the motor symptoms of PD without inducing LID.
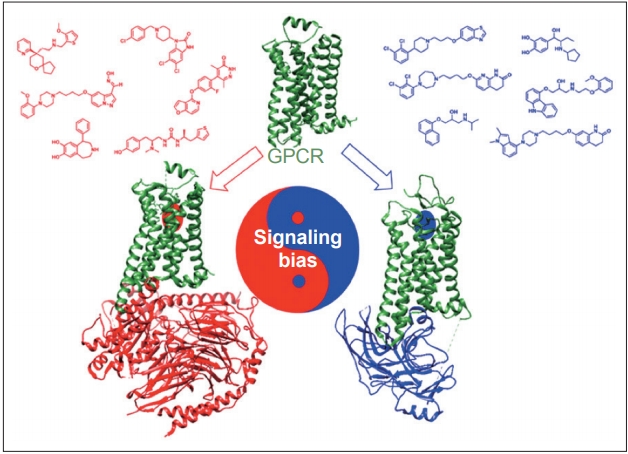
Functional selectivity, or signaling bias, is a recent concept in G protein-coupled receptor (GPCR) pharmacology whereby the ligand can selectively activate either G protein- (red) or β-arrestin-(blue) mediated downstream signaling pathways. It is hypothesized that selectively activating β-arrestin at dopamine receptors reduces levodopa-induced dyskinesia by desensitizing supersensitive GPCRs while still maintaining intracellular signaling that allows for improved motor functioning. Reprinted with permission from Tan et al [31].
One of the prevailing hypotheses of LID involves GPCRs on dopaminergic neurons entering a supersensitive state after being deprived of postsynaptic dopamine due to PD. This supersensitive state results in enhanced G protein-mediated signaling [32], which is believed to contribute to uncontrolled downstream signaling and neuronal hyperactivity [33]. Several attempts, including attempts to decrease dopamine receptor surface expression on neurons [34], reduce abnormal signaling [35], and inhibit A2A [36], mGluR5 [27], or NMDA receptors [37], have been made to limit either uncontrolled signaling or neuronal hyperactivity. While none of these have proven successful, one promising strategy to address this problem involves the selective activation of β-arrestins, which act as signal transducers downstream of GPCRs [38] in motor-related signaling pathways. It has been hypothesized that selectively activating β-arrestin2 on dopamine receptors reduces LID by desensitizing supersensitive GPCRs while still maintaining the intracellular signaling that allows for improved motor function [33].
Animal studies have supported the notion that LID is associated with increased G protein signaling and have also validated β-arrestins as novel targets for treating PD without causing LID. For example, knocking out β-arrestin2 in nonhuman primate models of PD resulted in worsened LID following L-DOPA administration, while β-arrestin2 overexpression reduced LID and increased locomotion via β-arrestin-mediated signaling [33].
Although further preclinical and clinical testing must be done, these results have generated significant interest in this area. Recently, Gray et al. [39] reported a novel noncatechol-containing agonist scaffold with excellent blood-brain barrier permeability, potency, and selectivity for the D1 dopamine receptor (D1R). Although the reported scaffold is highly biased for the stimulatory G protein (GS) pathway, displaying almost no activation of β-arrestin2 recruitment to D1R, the scaffold has been shown to have a sustained effect during a 3-day period on eye-blink rate in a nonhuman primate model of PD, which is considered a functional marker of central dopaminergic activity mediated through D1R [39]. The sustained response observed in this short-term study was attributed to reduced desensitization and tachyphylaxis associated with diminished β-arrestin2 signaling. Although a fully β-arrestin2-biased D1R ligand has not yet been reported, recent studies have reported promising findings regarding adapting the novel noncatechol D1R scaffold to increase its β-arrestin2 bias [40]. Advancements in GPCR drug discovery and biological validation have ensured that progress towards developing a functionally selective clinical drug candidate for the treatment of PD is underway.
CONCLUSION
L-DOPA is a staple of therapeutic regimens for PD patients because it effectively improves motor function and quality of life. LID emerges after chronic L-DOPA use and often complicates therapeutic management. Recent innovations have spawned new pharmacological strategies for the treatment of LID, and these innovations have progressed significantly in recent years and have shown great promise in preclinical and clinical studies. It is possible that some of these strategies may eventually be a viable option for patients who are ineligible for neurosurgical procedures or unable to tolerate the adverse side effects of other pharmacological interventions.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.
Acknowledgements
This work was supported in part by the F30AG062054 from the U.S. National Institutes of Health, awarded to M.L.M.
