Deep Brain Stimulation for Cockayne Syndrome-Associated Movement Disorder
Article information
Abstract
Cockayne syndrome (CS) is a rare progeroid disorder characterized by multisystem degeneration, including neurological dysfunction, for which deep brain stimulation (DBS) is a proposed treatment. This study represents only the third case of DBS for CS-associated movement disorder and the first in which both proposed targets had devices implanted, allowing for direct comparison. A case of DBS for CS-associated movement disorder is presented. Previous literature documents two cases with one targeting the ventral intermediate nucleus of the thalamus (VIM) and the other targeting the globus pallidus interna (GPi). Our patient underwent stimulation of GPi nuclei followed by repositioning to VIM nuclei with improved symptom control using VIM stimulation. In all cases, there was a significant clinical benefit without off-target effects. CS-associated movement disorder exhibits phenotypic variability for which DBS is a viable treatment. Target selection should be driven by clinical phenotype.
As the use of deep brain stimulation (DBS) in the treatment of movement disorders continues to expand, there is increasing interest in its application for rare disorders. Cockayne syndrome (CS) is a rare progeroid disorder that causes premature aging of all tissues and is characterized by multisystem degeneration, including neurological dysfunction [1,2]. CS is a disorder of nucleotide excision repair mechanisms, of which there are several other known disorders with overlapping phenotypes, such as xeroderma pigmentosum and trichothiodystrophy [3]. CS has many identified causative genetic mutations that manifest a spectrum of phenotypic variation, including neurological symptoms [3]. Neurological symptoms include medically refractory dystonia and tremor, which make the application of DBS a promising treatment. We provide only the third documented case utilizing DBS for the treatment of hyperkinetic movement disorder associated with CS and review the details of the previous cases for a comparative series.
CASE REPORT
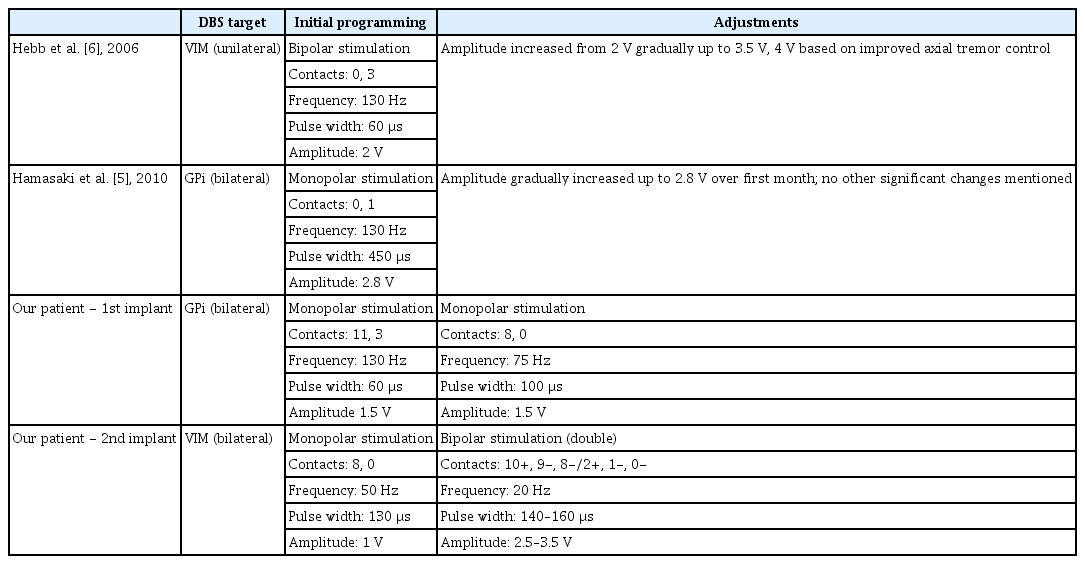
Our patient is a 10-year-old male with CS who presented to the pediatric neurology clinic for consideration of treatment options, including DBS, for CS-associated movement disorder. The patient began to show initial symptoms at approximately 12–15 months of age, as he did not begin to walk until 15 months of age. His parents report frequent stumbling and clumsy movements with poor coordination, which has been pervasive through his development. He began to exhibit a tremor at 5 years of age, which prompted further investigation, including magnetic resonance imaging of his brain, which revealed imaging characteristics suggestive of CS (Supplementary Figure 1 in the online-only Data Supplement) [1]. A physical exam revealed a bilateral high amplitude, low to moderate frequency jerky postural and intention tremor. Additional clinical criteria consistent with CS were progressive growth failure, microcephaly, cutaneous photosensitivity, and speech delay [1]. Genetic testing was pursued, and DNA sequencing confirmed mutations in both copies of ERCC6, a gene involved in nucleotide excision repair, which is diagnostic of CS [3]. Two separate variant mutations (p.Gln156*, p.Tyr1179Leufs*22) were identified in our patient, both of which matched previously identified pathogenic mutations [4]. There was no family history of CS and no other known affected family members. Medical therapy for tremor, including gabapentin and primidone, was ineffective; therefore, DBS treatment was discussed with the family, who elected to proceed with the surgery. The patient subsequently underwent placement of DBS electrodes to the globus pallidus interna (GPi) nuclei bilaterally in a standard fashion utilizing the Medtronic Activa DBS platform (Medtronic, Minneapolis, MN, USA). There are only two prior publications regarding DBS therapy for CS, with one targeting the GPi and the other targeting the ventral intermediate nucleus of the thalamus (VIM) [5,6]. GPi was chosen as the initial target, with the idea that it would address both dystonic and hyperkinetic symptoms [7]. Shortly after implantation, the patient experienced an episode of noninfectious perielectrode edema resulting in encephalopathy, a seizure and a 6-day hospitalization. He was treated with high-dose steroids followed by steroid taper without residual deficits. Once the patient recovered, programming was initiated with monopolar stimulation of contacts 11 and 3 at a frequency of 130 Hz, a pulse width (PW) of 60 μs, and an amplitude of 1.5 V. The patient noted decreased tripping and a 5-pound weight gain with these settings but minimal improvement in his tremor, suggesting the treatment had a greater impact on dystonia than tremor (Table 1). Due to a tremor phenomenology similar to Holmes tremor [8], programming was adjusted to monopolar stimulation of posteroventral contacts 8 and 0 at a frequency of 75 Hz, a PW of 100 μs, and an amplitude of 1.5 V, with some improvement in tremor control (Table 2).
Five months after the initial implantation, high impedance values were noted bilaterally, prompting a surgical exploration of the system. During surgery, wire disconnects were noted in both leads requiring replacement of the intracranial leads. Given the need to replace the intracranial leads, the decision was made to reposition the electrodes to the bilateral VIM nuclei in hopes of gaining improved tremor control. Surgery was uneventful, and the patient was discharged home the following day. Programming of the bilateral VIM was initiated with monopolar stimulation of contacts 8 and 0 at a frequency of 50 Hz, a PW of 130 μs, and an amplitude of 1 V (Table 2). This programming was eventually transitioned to double bipolar stimulation with an increased amplitude and a lower frequency (10+, 9-, 8-/2+, 1-, 0-, 2.5–3.5 V, PW 140–160 μs, Freq 20 Hz), which led to improved tremor control. With the VIM target and these programming settings, the patient had resolution of his resting tremor and significant improvement in his intention tremor, leading to an improvement in functionality to the point that he was now able to hold his lunch tray at school and eat meals using utensils without spilling his food. Video from the initial programming visit is shown with the patient performing the finger-to-nose task prior to the activation of the system and then with the initial settings prior to fine-tuning at subsequent visits (Supplementary Video 1 in the online-only Data Supplement). Informed consent was obtained from the patient’s parents for the use of the video in publication.
DISCUSSION
CS-associated movement disorder exhibits phenotypic variability ranging from dystonia-predominant to tremor-predominant subtypes. CS has been described to have several types, with Type 1 being the classical form, Type 2 being a more severe subtype, and “mild CS” including patients with some clinical features but lacking at least some cardinal features [2]. CS 2 is more severe, with death often occurring by age 6 or 7, while patients with CS 1 have an average lifespan of 12.3 years, with a proportion surviving into young adulthood [1,2]. Given the rarity of CS-associated movement disorder, there is limited experience in its treatment. The underlying pathology is thought to be secondary to pathologic changes in the basal ganglia thalamocortical motor loop. Based on the experiences in this series of patients, DBS is a viable treatment for CS. DBS can be considered for patients with CS who display a more slowly progressing phenotype, as they may have a sufficient lifespan to benefit from the treatment. In the two previous cases, a patient with a mixed hyperkinetic movement disorder responded to VIM stimulation, while a patient with dystonia predominance responded to GPi stimulation.
The first case was described by Hebb et al. [6], in which a 17-year-old male with CS underwent implantation of a DBS electrode in the VIM nucleus. The patient exhibited a mixed hyperkinetic movement disorder with severe chorea, myoclonus, bilateral intention tremor, and dystonic features, which continued to progress and were refractory to multiple medications. A unilateral system was implanted targeting the VIM nucleus, and programming data are described in Table 2. The chorea and myoclonus improved in the first few days to weeks on the contralateral side; eventually, with increased stimulation amplitude, axial and ipsilateral improvements were noted (Table 1).
The second case was described by Hamasaki et al. [5], in which a 52-year-old male with CS underwent implantation of DBS electrodes to the bilateral GPi nuclei. The patient had progressive symptoms primarily manifesting cervical dystonia that later generalized, greatly impairing function. He underwent placement of bilateral GPi electrodes, and programming data are described in Table 2. The patient’s cervical dystonia and dystonic tremor improved over the first several weeks with continued improvement to his last follow-up five months postoperatively. His axial symptoms did not improve; however, he was able to feed himself one week after surgery, which he could not do preoperatively (Table 1).
In our patient, we had the unique opportunity to evaluate clinical responses to each of the previously utilized targets in CS. He experienced greater symptom reduction and functional improvement with VIM stimulation as opposed to GPi stimulation. This may not be completely unexpected given that the patient had tremor-predominant symptomatology as opposed to dystoniapredominant symptomatology, which may respond better to GPi stimulation. Additionally, the patient’s physical exam revealed a bilateral high amplitude, low to moderate frequency jerky postural and intention tremor, consistent with a cerebellar tremor. On histopathologic analysis, there was notable Purkinje cell degeneration, which is likely a contributing factor in this phenotype in CS [9]. In regard to our patient’s GPi stimulation, several potential limiting factors are present that deserve comment. The first is that there was an episode of noninfectious perielectrode edema, which is a rare complication of DBS; however, the clinical outcomes of patients who experience this are equivalent to those who do not [10]. The second is that both leads ended up with defects requiring replacement. Since we had several months of GPi system stimulation with normal functioning prior to noting the increased impedance values, we believe there was adequate time to evaluate its effectiveness, or lack thereof.
In making strong recommendations, we are limited by the rarity of the condition, leaving us to draw conclusions from the experience of a case series. However, it seems evident that DBS is safe and efficacious in treating CS-associated movement disorders with multiple viable targets. Target selection should be driven by the clinical phenotype, with tremor predominance responding better to VIM stimulation, while dystonia predominance responds better to GPi stimulation.
Supplementary Materials
The online-only Data Supplement is available with this article at https://doi.org/10.14802/jmd.21005.
Video 1.
Patient performing the finger-to-nose task 4 weeks after bilateral VIM implant prior to the initial activation of the deep brain stimulation (DBS) system (segment 1) and then after initial system activation (segment 2), demonstrating improved tremor control with DBS stimulation.
Supplementary Figure 1.
Neuroimaging characteristics of Cockayne syndrome. Neuroimaging characteristics of Cockayne syndrome, including basal ganglia calcifications shown on the CT of the head (A), white matter abnormalities on T2 fluid-attenuated inversion recovery (B), basal ganglia susceptibility on susceptibility weighted imaging (C), and cerebral atrophy with predominant white matter loss on T2 (D) were noted in our patient.
Notes
Ethics Statement
The authors confirm that the approval of an Institutional Review Board was not required for this work. Informed consent was obtained from the patient’s parents for the use of the video in publication. The authors confirm that additional informed consent was not required for this work. We confirm that we have read the journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Conflicts of Interest
The authors have no financial conflicts of interest.
Funding
None.
Author Contributions
Conceptualization: Joseph S. Domino, Christian Kaufman. Data curation: Joseph S. Domino. Formal analysis: Joseph S. Domino, Rose Gelineau-Morel. Investigation: Joseph S. Domino. Methodology: all authors. Resources: Rose Gelineau-Morel. Supervision: Christian Kaufman. Visualization: Joseph S. Domino. Writing—original draft: Joseph S. Domino. Writing—review & editing: Rose Gelineau-Morel, Christian Kaufman.