Articles
- Page Path
- HOME > J Mov Disord > Volume 10(1); 2017 > Article
-
Original Article
Exosome-Based Delivery of miR-124 in a Huntington’s Disease Model - Soon-Tae Lee1,2*, Wooseok Im1,3*, Jae-Jun Ban1*, Mijung Lee1, Keun-Hwa Jung1,2, Sang Kun Lee1,2, Kon Chu1,2, Manho Kim1,4
-
Journal of Movement Disorders 2017;10(1):45-52.
DOI: https://doi.org/10.14802/jmd.16054
Published online: January 18, 2017
1Department of Neurology, Neuroscience Research Center, Seoul National University Hospital, Seoul, Korea
2Program in Neuroscience, Neuroscience Research Institute of SNUMRC, Seoul National University, Seoul, Korea
3Neuroscience Research Institute, Seoul National University College of Medicine, Seoul, Korea
4Protein Metabolism Medical Research Center, Seoul National University College of Medicine, Seoul, Korea
- Corresponding author: Manho Kim, MD, PhD, Department of Neurology, Seoul National University Hospital, Seoul National University College of Medicine, 101 Daehak-ro, Jongno-gu, Seoul 03080, Korea / Tel: +82-2-2072-2193 / Fax: +82-2-3672-7553 / E-mail: kimmanho@snu.ac.kr
- Corresponding author: Kon Chu, MD, PhD, Department of Neurology, Seoul National University Hospital, Seoul National University College of Medicine, 101 Daehak-ro, Jongno-gu, Seoul 03080, Korea / Tel: +82-2-2072-1878 / Fax: +82-2-3672-4949 / E-mail: stemcell.snu@gmail.com
- *Soon-Tae Lee, Wooseok Im, and Jae-Jun Ban contributed equally to this work.
Copyright © 2017 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Objective
- Huntington’s disease (HD) is a genetic neurodegenerative disease that is caused by abnormal CAG expansion. Altered microRNA (miRNA) expression also causes abnormal gene regulation in this neurodegenerative disease. The delivery of abnormally downregulated miRNAs might restore normal gene regulation and have a therapeutic effect.
-
Methods
- We developed an exosome-based delivery method to treat this neurodegenerative disease. miR-124, one of the key miRNAs that is repressed in HD, was stably overexpressed in a stable cell line. Exosomes were then harvested from these cells using an optimized protocol. The exosomes (Exo-124) exhibited a high level of miR-124 expression and were taken up by recipient cells.
-
Results
- When Exo-124 was injected into the striatum of R6/2 transgenic HD mice, expression of the target gene, RE1-Silencing Transcription Factor, was reduced. However, Exo-124 treatment did not produce significant behavioral improvement.
-
Conclusion
- This study serves as a proof of concept for exosome-based delivery of miRNA in neurodegenerative diseases.
- Generation of miR-124-expressing cell line
- Mature miR-124 was cloned into the pSUPER plasmid to overexpress the miRNA in cells. HEK 293 cells were transfected with the pSUPER-miR-124 vector using Lipofectamine 2000 (Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions. The pSUPER-miR-124-transfected cells were cultured in medium containing Zeocin (500 μg/mL) to select for cells expressing miR-124. The medium was changed every 2 or 3 days until single-cell colonies formed (2–3 weeks). Single colonies were isolated and regrown for 2 weeks with Zeocin media, and miR-124 expression was tested using quantitative reverse transcription polymerase chain reaction (qRT-PCR).
- Isolation of exosomes
- HEK 293 cells overexpressing miR-124 were cultured in exosome-free Dulbecco’s modified Eagle’s medium (DMEM; Gibco Invitrogen/Life Technologies, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco Invitrogen/Life Technologies) and 1% penicillin-streptomycin at 37°C in a 5% CO2 incubator. To deplete bovine exosomes, DMEM containing 10% FBS was centrifuged at 100,000 g for 15 h, and the supernatant was used for cell culture. Culture supernatants were harvested after 5 days. Cells and cellular debris were eliminated from the conditioned medium by centrifugation at 3,000 g for 10 min at room temperature. The conditioned medium was treated with citrate (3 mM, 6 mM) for 2 days at 37°C. Exosomes were isolated using the Exo-Quick exosome precipitation kit (System Biosciences, Mountain View, CA, USA) according to the manufacturer’s specifications. Briefly, 5 mL of conditioned medium was thoroughly mixed with 1 mL of Exo-Quick exosome precipitation solution, and the solution was incubated for 24 h at 4°C. The conditioned medium was centrifuged at 1,500 g for 30 min, and then the supernatant was removed and centrifuged at 1,500 g for 5 min. The exosome pellet was resuspended in buffer.
- Analysis of intercellular transfer of Exo-124
- Exosomes were labeled with PKH67 (Sigma-Aldrich, St. Louis, MO, USA) according to the manufacturer’s protocol. Briefly, the exosome pellet was suspended in 100 μL PBS and then diluted in 1 mL of Diluent C. The solution was then rapidly mixed with a freshly prepared solution of 4 μL of PKH67 dye in 1 mL of Diluent C. The mixture was incubated for 5 min. Labeling was stopped by the addition of 2 mL of 1% BSA in PBS for 1 min. The samples were washed 2 times with 25 mL of PBS and then centrifuged at 120,000 g for 100 min. After labeling, the PKH67-labeled exosomes were stored in PBS containing 2% paraformaldehyde until flow cytometric analysis (BD Biosciences Pharmingen, San Jose, CA, USA). PKH67 fluorescence was evaluated using the green channel, and the data were analyzed using WinMDI software (Windows Multiple Document Interface for Flow Cytometry, West Lafayette, IN, USA). Fluorescently stained sections of brains that were injected with PKH67-labeled exosomes were analyzed using an inverted microscope (BX61, Olympus Corporation, Tokyo, Japan).
- Real-time PCR for miRNA
- RNA was isolated using Trizol (Thermo Fisher Scientific, Asheville, NC, USA), and the level of miR-124 was quantified using the mirVana qRT-PCR miRNA Detection Kit and TaqMan miRNA assays (Ambion, Applied Biosystems, Foster City, CA, USA). All reactions were run in triplicate on an ABI PRISM 7,000 sequence detection system (Applied Biosystems). Relative expression levels were calculated using the comparative threshold cycle (Ct) method (relative expression levels = 2-∆Ct) and were normalized to the expression of the control, snoRNA202, which was measured in the same samples using an endogenous snoRNA detection kit (Ambion, Applied Biosystems).
- HD transgenic mice and injection of exosome
- The animal study was approved by the Institutional Animal Care and Use Committee of Seoul National University Hospital, which was accredited by the International Association for the Assessment and Accreditation of Laboratory Animal Care. We used the R6/2 line of transgenic HD mice [B6CBATg(HDexon1)62Gpb/3J, The Jackson Laboratory; 6 female and 6 male mice; 111 CAG repeats] and their wild type (WT) littermates (6 female and 6 male mice) at 6 weeks of age. These mice were obtained by crossing ovarian transplant hemizygous females with B6CBAF1/J males. Genotyping was performed using PCR [24]. An expanded CAG repeat in exon 1 of the HD gene is sufficient to cause a progressive neurological phenotype in these transgenic mice [24]. The mice were housed in groups with ad libitum access to food and water and a 12-h light/12-h dark cycle.
- To inject exosomes, 6-week-old (42 days) mice were anesthetized via intraperitoneal injection of 1% ketamine (30 mg/kg) and xylazine hydrochloride (4 mg/kg) and then positioned in a stereotaxic apparatus. All exosomes obtained from 5 mL of conditioned medium were suspended in 12 μL of PBS. In the exosome injection group (Exo-124), a 30-gauge Hamilton syringe was used to inject the exosome suspension into the bilateral striatum (2 μL of suspension per hemisphere) at the following coordinates: AP + 0.38 mm, ML ± 2.0 mm, and DV -3.5 mm from Bregma. The exosomes were slowly injected over a period of 5 min. The needle was left in position for another 5 min and then gently removed. All animal experiments and procedures complied with the NIH Guide for the Care and Use of Laboratory Animals.
- Behavioral test
- Rota-Rod performance was assessed using an accelerating Rota-Rod (San Diego Instruments, San Diego, CA, USA) as previously described [25]. The speed of the Rota-Rod was set to linearly increase from 4 to 40 rpm over 3 min. The mice were initially trained on the Rota-Rod for three consecutive days. Three trials were conducted per day, with a 30-min rest period between trials. The trials and tests were terminated if the animal fell off the rungs or gripped the device and completed two consecutive revolutions. At each age, mice underwent three trials, and the mean latency to fall was calculated.
- Histological analysis
- When the mice were 8 weeks old (1 week after the exosome injection, n = 3), histological evaluations were performed as previously described [25]. Briefly, the animals were deeply anesthetized and then perfused through the heart with 10 mL of cold saline and 10 mL of 4% paraformaldehyde in 0.1 M PBS. Sections (20 μm thick) were counter-stained with DAPI (Sigma-Aldrich, St. Louis, MO, USA), and PKH67 fluorescence was analyzed using a microscopic imaging system (Carl Zeiss, Thornwood, NY, USA).
- Western blotting and real-time PCR in R6/2 mice
- At the age of 8 weeks (1 week after the exosome injection), the mice were sacrificed by decapitation, and the brains were immediately removed. Homogenates of each hemisphere were separately and serially processed for Western blotting and RNA work. Western blotting was performed using antibodies against REST (Abcam, Cambridge, UK), doublecortin (DCX; Santa Cruz Biotechnology, Santa Cruz, CA, USA), or β-actin (Santa Cruz Biotechnology). The blots were developed using enhanced chemiluminescence reagents (Pierce, Rockford, IL, USA) and digitally scanned (GS-700; Bio-Rad, Hercules, CA, USA). The optical density of each band relative to that of the β-actin band was determined using Molecular Analyst® software (Bio-Rad). For real-time PCR, total RNA was isolated from each hemisphere using QIAshredder and RNeasy kits (Qiagen, Valencia, CA, USA). miR-124 levels were measured using the mirVana qRT-PCR miRNA Detection Kit and TaqMan miRNA assays (Ambion, Applied Biosystems).
- Data analysis and statistics
- All data in this study are presented as the mean ± standard deviation. The Mann-Whitney U test was used for nonparametric, inter-group comparisons. SPSS 17.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analyses. A two-tailed p-value < 0.05 was considered significant.
MATERIALS & METHODS
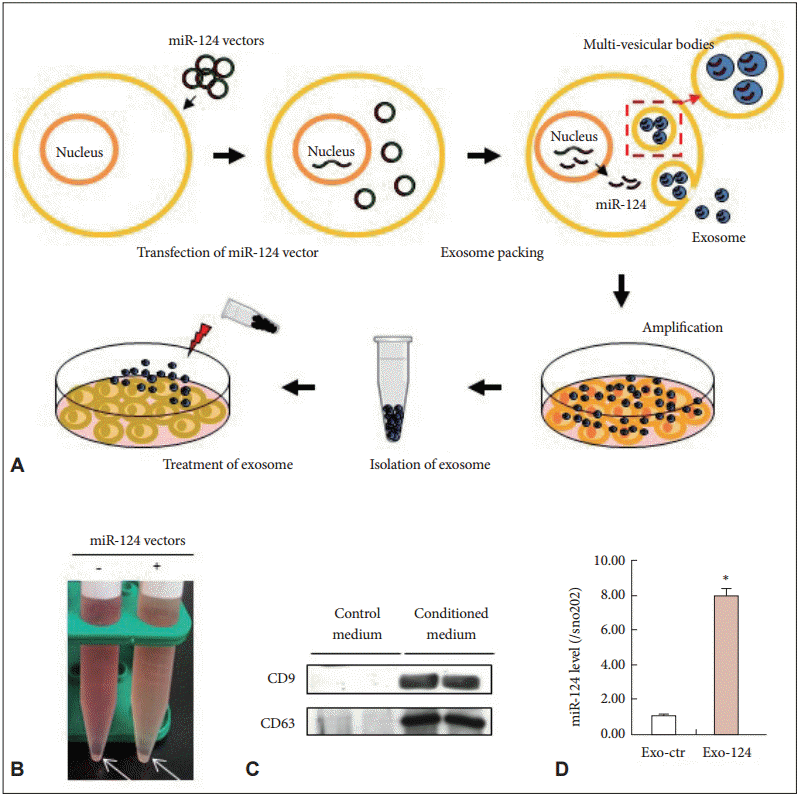
- We first generated a miR-124-expressing cell line by transfecting the miR-124 expression vector into HEK 293 cells. miR-124 expression was confirmed by real-time PCR (data not shown). We harvested exosomes from the medium used to culture these cells, as presented in Figure 1A. Based on previous reports [26], we added citrate and Mg2+ during the procedure to protect the RNA contained in the exosomes. Figure 1B shows the harvested exosome pellets.
- We evaluated the pellets by blotting for CD9 and CD63, which are tetraspanin proteins that are expressed at the cell surface and thus serve as exosome markers [14,27]. The exosomes expressed both CD9 and CD63 (Figure 1C), whereas centrifugation of unused culture medium did not produce any CD9- and CD63-bearing materials. We thus confirmed the presence of exosomes.
- We then measured the expression of miR-124 in the exosomes. Control exosomes (Exo-ctr) harvested from naïve HEK 293 cells that were not transfected with miR-124 expressed very low levels of miR-124. On the other hand, miR-124-enhanced exosomes (Exo-124) harvested from HEK 293 cells that were transfected with the miR-124 expression vector expressed much higher levels of miR-124 (Figure 1D). Thus, we moved forward to in vitro and in vivo applications of Exo-124.
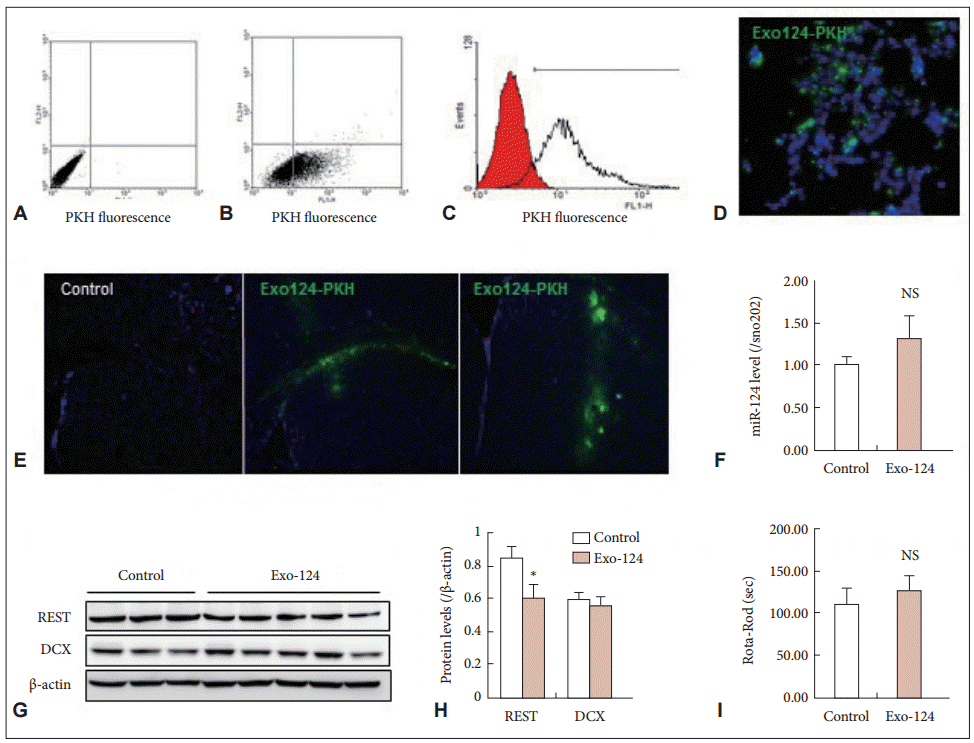
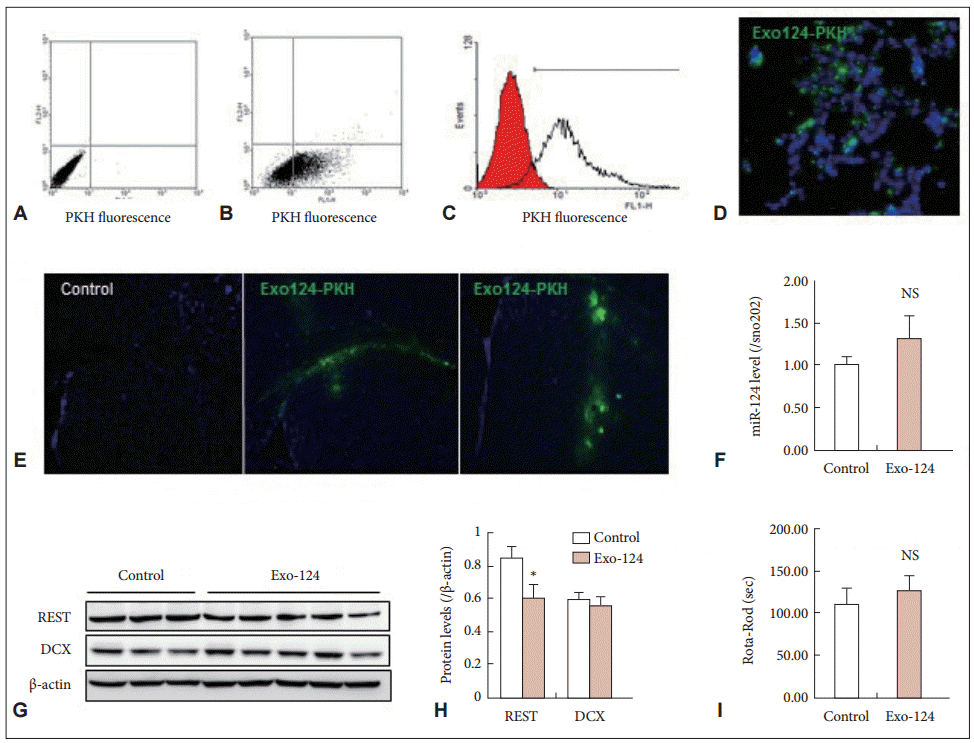
- To confirm intercellular transfer of the exosomes, we labeled the exosomes with PKH67 and added Exo-124 to the culture medium of HEK 293 cells for 24 h. After Exo-124 treatment, the PKH67 fluorescence of the recipient HEK 293 cells was confirmed by flow cytometry (Figure 2A, B, and C) and immunocytochemistry (Figure 2D). These results demonstrated that Exo-124 produced by our isolation and treatment protocol was efficiently taken up by recipient cells.
- Because previous studies have demonstrated the efficacy of exosome-mediated delivery of miRNA [14], we proceeded directly to the therapeutic application of Exo-124 in R6/2 HD transgenic mice. We injected Exo-124 into the striatum of 6-week-old R6/2 mice. Upon analysis 1 week later, PKH67-labeled Exo-124 had been taken up by the striatum (Figure 2E). We measured miR-124 expression in the mice and found that the Exo-124-treated R6/2 mice exhibited slightly higher levels of miR-124 expression compared to the control (non-treated) R6/2 mice, but this difference was not significant (Figure 2F). We hypothesized that the endogenous abundance of miR-124 in the brain masked the effect of delivery. Thus, we measured brain levels of REST protein, the key target protein of miR-124, and we found that REST expression was lower in the Exo-124-treated R6/2 mice than in the control R6/2 mice (Figure 2G and H). Next, because the miR-124-REST system regulates neurogenesis, we measured Dcx (doublecortin) expression in the brain and found that Exo-124 treatment had no effect on Dcx protein levels (Figure 2G and H). Moreover, at 7 weeks of age, no differences in Rota-Rod performance were observed between the Exo-124-treated R6/2 mice and the control R6/2 mice (Figure 2I).
RESULTS
- In this study, we demonstrated the feasibility of exosome-based therapeutic delivery of miR-124 in a HD model. We could generate Exo-124 from a miR-124-overexpressing cell line, enabling stable and efficient generation of miRNA-bearing exosomes. In addition, when Exo-124 was delivered to the striatum, it reduced the expression of REST. However, Exo-124 had little effect on Rota-Rod performance.
- The delivery method is one of the critical issues in the development of miR-based therapeutics for neurological diseases. When using antagomirs, the antisense inhibitor oligonucleotides of miRNAs, it is possible to use naked formulations and to deliver them via stereotaxic injections into the brain [28]. However, when miRNAs themselves are delivered, it is necessary to use additional methods to protect the miRNAs from degradation, such as lipid-based delivery systems, poly-(lactic-co-glycolic acid) particles, or viral vectors [29]. The exosome method is more physiological and less immunogenic, and if the exosomes are engineered to express specific surface ligands such as integrins, the exosome method can be used to selectively reach specific target cells [23].
- Exosomes or extracellular vesicles can deliver not only miRNAs but also mRNAs, non-coding RNAs, cellular proteins, or even prions [30]. Under natural conditions, exosomes mediate cross-talk between neurons and other cells, such as microglia and immune cells [31]. Exosomes regulate development and normal brain function and also contribute to synaptic plasticity and regeneration [23,32,33]. In disease conditions, exosomes can be used to deliver RNAi, antigens for cancer immunotherapy, and therapeutic chemicals. In the present study, we showed that naïve miRNAs can be packed in exosomes and delivered as a therapeutic treatment for neurodegenerative diseases.
- Although we showed that exosome-encapsulated miRNAs could be transferred to recipient cells in vitro, the current method must be further developed for in vivo applications. Because the therapeutic efficacy of Exo-124 was modest, it may be necessary to increase the dose of miRNAs packed in the exosomes. In addition, because the therapeutic effect of miR-124 might be limited, it is necessary to test whether the delivery of other miRNAs via the exosome method might have greater therapeutic effects. Candidate miRNAs might include miR-9, miR-22, miR-125b, miR-146a, miR-150, and miR-214 [7,34-36]. Moreover, differences in the tissue distribution and efficiency of target gene repression should be compared between systemic injections, such as intravenous or subcutaneous methods, and local injection for future in vivo applications. In this case, for targeted delivery to the brain, we can add integrin ligands or peptides that recognize transferrin receptors [37,38]. Overall, the current study provides a proof of concept for exosome-based delivery of miRNAs to the brain, warranting further studies of this method.
DISCUSSION
- This work was supported by the National Research Foundation of Korea (NRF) (2014R1A2A1A11051520), Korea Health 21 R & D Project (HI14C2348) by the Ministry of Health & Welfare, Republic of Korea, the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (2016M3C7A1914002).
Acknowledgments
- 1. Landles C, Bates GP. Huntingtin and the molecular pathogenesis of Huntington’s disease. Fourth in molecular medicine review series. EMBO Rep 2004;5:958–963.ArticlePubMedPMC
- 2. Sugars KL, Rubinsztein DC. Transcriptional abnormalities in Huntington disease. Trends Genet 2003;19:233–238.ArticlePubMed
- 3. Zuccato C, Tartari M, Crotti A, Goffredo D, Valenza M, Conti L, et al. Huntingtin interacts with REST/NRSF to modulate the transcription of NRSE-controlled neuronal genes. Nat Genet 2003;35:76–83.ArticlePubMedPDF
- 4. Rigamonti D, Mutti C, Zuccato C, Cattaneo E, Contini A. Turning REST/NRSF dysfunction in Huntington’s disease into a pharmaceutical target. Curr Pharm Des 2009;15:3958–3967.ArticlePubMed
- 5. Lee ST, Chu K, Im WS, Yoon HJ, Im JY, Park JE, et al. Altered microRNA regulation in Huntington’s disease models. Exp Neurol 2011;227:172–179.ArticlePubMed
- 6. Johnson R, Zuccato C, Belyaev ND, Guest DJ, Cattaneo E, Buckley NJ. A microRNA-based gene dysregulation pathway in Huntington’s disease. Neurobiol Dis 2008;29:438–445.ArticlePubMed
- 7. Packer AN, Xing Y, Harper SQ, Jones L, Davidson BL. The bifunctional microRNA miR-9/miR-9* regulates REST and CoREST and is downregulated in Huntington’s disease. J Neurosci 2008;28:14341–14346.ArticlePubMedPMC
- 8. Buckley NJ, Johnson R, Zuccato C, Bithell A, Cattaneo E. The role of REST in transcriptional and epigenetic dysregulation in Huntington’s disease. Neurobiol Dis 2010;39:28–39.ArticlePubMed
- 9. Cheng LC, Pastrana E, Tavazoie M, Doetsch F. miR-124 regulates adult neurogenesis in the subventricular zone stem cell niche. Nat Neurosci 2009;12:399–408.ArticlePubMedPMCPDF
- 10. Das E, Jana NR, Bhattacharyya NP. MicroRNA-124 targets CCNA2 and regulates cell cycle in STHdh(Q111)/Hdh(Q111) cells. Biochem Biophys Res Commun 2013;437:217–224.ArticlePubMed
- 11. Gil-Mohapel J, Simpson JM, Ghilan M, Christie BR. Neurogenesis in Huntington’s disease: can studying adult neurogenesis lead to the development of new therapeutic strategies? Brain Res 2011;1406:84–105.ArticlePubMed
- 12. Phillips W, Morton AJ, Barker RA. Abnormalities of neurogenesis in the R6/2 mouse model of Huntington’s disease are attributable to the in vivo microenvironment. J Neurosci 2005;25:11564–11576.ArticlePubMedPMC
- 13. Liu T, Im W, Mook-Jung I, Kim M. MicroRNA-124 slows down the progression of Huntington’s disease by promoting neurogenesis in the striatum. Neural Regen Res 2015;10:786–791.ArticlePubMedPMC
- 14. Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 2007;9:654–659.ArticlePubMedPDF
- 15. Deregibus MC, Cantaluppi V, Calogero R, Lo Iacono M, Tetta C, Biancone L, et al. Endothelial progenitor cell derived microvesicles activate an angiogenic program in endothelial cells by a horizontal transfer of mRNA. Blood 2007;110:2440–2448.ArticlePubMed
- 16. Katsuda T, Tsuchiya R, Kosaka N, Yoshioka Y, Takagaki K, Oki K, et al. Human adipose tissue-derived mesenchymal stem cells secrete functional neprilysin-bound exosomes. Sci Rep 2013;3:1197.ArticlePubMedPMCPDF
- 17. Sahoo S, Klychko E, Thorne T, Misener S, Schultz KM, Millay M, et al. Exosomes from human CD34(+) stem cells mediate their proangiogenic paracrine activity. Circ Res 2011;109:724–728.ArticlePubMedPMC
- 18. Ohno S, Takanashi M, Sudo K, Ueda S, Ishikawa A, Matsuyama N, et al. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther 2013;21:185–191.ArticlePubMedPMC
- 19. Michael A, Bajracharya SD, Yuen PS, Zhou H, Star RA, Illei GG, et al. Exosomes from human saliva as a source of microRNA biomarkers. Oral Dis 2010;16:34–38.ArticlePubMedPMC
- 20. Zhou H, Yuen PS, Pisitkun T, Gonzales PA, Yasuda H, Dear JW, et al. Collection, storage, preservation, and normalization of human urinary exosomes for biomarker discovery. Kidney Int 2006;69:1471–1476.ArticlePubMedPMC
- 21. Gupta SK, Bang C, Thum T. Circulating microRNAs as biomarkers and potential paracrine mediators of cardiovascular disease. Circ Cardiovasc Genet 2010;3:484–488.ArticlePubMed
- 22. Tsujiura M, Ichikawa D, Komatsu S, Shiozaki A, Takeshita H, Kosuga T, et al. Circulating microRNAs in plasma of patients with gastric cancers. Br J Cancer 2010;102:1174–1179.ArticlePubMedPMCPDF
- 23. Lai CP, Breakefield XO. Role of exosomes/microvesicles in the nervous system and use in emerging therapies. Front Physiol 2012;3:228.ArticlePubMedPMC
- 24. Mangiarini L, Sathasivam K, Seller M, Cozens B, Harper A, Hetherington C, et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 1996;87:493–506.ArticlePubMed
- 25. Lee ST, Chu K, Jung KH, Im WS, Park JE, Lim HC, et al. Slowed progression in models of Huntington disease by adipose stem cell transplantation. Ann Neurol 2009;66:671–681.ArticlePubMed
- 26. Adamala K, Szostak JW. Nonenzymatic template-directed RNA synthesis inside model protocells. Science 2013;342:1098–1100.ArticlePubMedPMC
- 27. Simpson RJ, Jensen SS, Lim JW. Proteomic profiling of exosomes: current perspectives. Proteomics 2008;8:4083–4099.ArticlePubMed
- 28. Butovsky O, Jedrychowski MP, Cialic R, Krasemann S, Murugaiyan G, Fanek Z, et al. Targeting miR-155 restores abnormal microglia and attenuates disease in SOD1 mice. Ann Neurol 2015;77:75–99.ArticlePubMedPMC
- 29. Wu Y, Crawford M, Mao Y, Lee RJ, Davis IC, Elton TS, et al. Therapeutic delivery of microRNA-29b by cationic lipoplexes for lung cancer. Mol Ther Nucleic Acids 2013;2:e84.ArticlePubMedPMC
- 30. Coleman BM, Hill AF. Extracellular vesicles--their role in the packaging and spread of misfolded proteins associated with neurodegenerative diseases. Semin Cell Dev Biol 2015;40:89–96.ArticlePubMed
- 31. Bellingham SA, Guo BB, Coleman BM, Hill AF. Exosomes: vehicles for the transfer of toxic proteins associated with neurodegenerative diseases? Front Physiol 2012;3:124.ArticlePubMedPMC
- 32. Fauré J, Lachenal G, Court M, Hirrlinger J, Chatellard-Causse C, Blot B, et al. Exosomes are released by cultured cortical neurones. Mol Cell Neurosci 2006;31:642–648.ArticlePubMed
- 33. Korkut C, Ataman B, Ramachandran P, Ashley J, Barria R, Gherbesi N, et al. Trans-synaptic transmission of vesicular Wnt signals through Evi/Wntless. Cell 2009;139:393–404.ArticlePubMedPMC
- 34. Jovicic A, Zaldivar Jolissaint JF, Moser R, Silva Santos Mde F, Luthi-Carter R. MicroRNA-22 (miR-22) overexpression is neuroprotective via general anti-apoptotic effects and may also target specific Huntington’s disease-related mechanisms. PLoS One 2013;8:e54222.ArticlePubMedPMC
- 35. Sinha M, Ghose J, Bhattarcharyya NP. Micro RNA -214,-150,-146a and-125b target Huntingtin gene. RNA Biol 2011;8:1005–1021.ArticlePubMed
- 36. Ghose J, Sinha M, Das E, Jana NR, Bhattacharyya NP. Regulation of miR-146a by RelA/NFkB and p53 in STHdh(Q111)/Hdh(Q111) cells, a cell model of Huntington’s disease. PLoS One 2011;6:e23837. ArticlePubMedPMC
- 37. Prades R, Guerrero S, Araya E, Molina C, Salas E, Zurita E, et al. Delivery of gold nanoparticles to the brain by conjugation with a peptide that recognizes the transferrin receptor. Biomaterials 2012;33:7194–7205.ArticlePubMed
- 38. Wu X, Reddy DS. Integrins as receptor targets for neurological disorders. Pharmacol Ther 2012;134:68–81.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- The role of exosomes in adult neurogenesis: implications for neurodegenerative diseases
Zhuoyang Yu, Yan Teng, Jing Yang, Lu Yang
Neural Regeneration Research.2024; 19(2): 282. CrossRef - A vivid outline demonstrating the benefits of exosome-mediated drug delivery in CNS-associated disease environments
Rachayeeta Ray, Sougata Ghosh Chowdhury, Parimal Karmakar
Archives of Biochemistry and Biophysics.2024; 753: 109906. CrossRef - Current progression in application of extracellular vesicles in central nervous system diseases
Xiang-Min Zhang, Jie Huang, Xiao-Ying Ni, Hui-Ru Zhu, Zhong-Xin Huang, Shuang Ding, Xin-Yi Yang, Yan-Di Tan, Jian-Fu Chen, Jin-Hua Cai
European Journal of Medical Research.2024;[Epub] CrossRef - Therapeutic advances in neural regeneration for Huntington's disease
Francesco D'Egidio, Vanessa Castelli, Giorgia Lombardozzi, Fabrizio Ammannito, Annamaria Cimini, Michele d'Angelo
Neural Regeneration Research.2024; 19(9): 1991. CrossRef - Extracellular vesicles in nanomedicine and regenerative medicine: A review over the last decade
Saeid Moghassemi, Arezoo Dadashzadeh, Maria João Sousa, Hanne Vlieghe, Jie Yang, Cecibel María León-Félix, Christiani A. Amorim
Bioactive Materials.2024; 36: 126. CrossRef - Most recent advances and applications of extracellular vesicles in tackling neurological challenges
Mandeep Kaur, Salvatore Fusco, Bram Van den Broek, Jaya Aseervatham, Abdolmohamad Rostami, Lorraine Iacovitti, Claudio Grassi, Barbara Lukomska, Amit K. Srivastava
Medicinal Research Reviews.2024;[Epub] CrossRef - Dual Role of Exosome in Neurodegenerative Diseases: A Review Study
Mohsen Sheykhhasan, Fatemeh Heidari, Mohsen Eslami Farsani, Maryam Azimzadeh, Naser Kalhor, Shima Ababzadeh, Reihaneh Seyedebrahimi
Current Stem Cell Research & Therapy.2024; 19(6): 852. CrossRef -
Posttranscriptional regulation of
FAN1
by miR-124-3p at rs3512 underlies onset-delaying genetic modification in Huntington’s disease
Kyung-Hee Kim, Eun Pyo Hong, Yukyeong Lee, Zachariah L. McLean, Emanuela Elezi, Ramee Lee, Seung Kwak, Branduff McAllister, Thomas H. Massey, Sergey Lobanov, Peter Holmans, Michael Orth, Marc Ciosi, Darren G. Monckton, Jeffrey D. Long, Diane Lucente, Vane
Proceedings of the National Academy of Sciences.2024;[Epub] CrossRef - Exosome-Based Macromolecular neurotherapeutic drug delivery approaches in overcoming the Blood-Brain barrier for treating brain disorders
Krishna Yadav, R. Vijayalakshmi, Kantrol Kumar Sahu, Pavani Sure, Kavita Chahal, Renu Yadav, Sucheta, Akhilesh Dubey, Megha Jha, Madhulika Pradhan
European Journal of Pharmaceutics and Biopharmaceutics.2024; : 114298. CrossRef - Exosomal microRNAs as diagnostic biomarkers and therapeutic applications in neurodegenerative diseases
Xuehereti Sataer, Zhu Qifeng, Zhang Yingying, He Chunhua, Feng Bingzhenga, Xu Zhiran, Li Wanli, Yang Yuwei, Chen Shuangfeng, Wu Lingling, Huang Hongri, Chen Jibing, Ren Xiaoping, Gao Hongjun
Neurological Research.2023; 45(3): 191. CrossRef - The role of microRNAs in neurodegenerative diseases: a review
Shijie Li, Zhixin Lei, Taolei Sun
Cell Biology and Toxicology.2023; 39(1): 53. CrossRef - Exosomes based strategies for brain drug delivery
Fawad Ur Rehman, Yang Liu, Meng Zheng, Bingyang Shi
Biomaterials.2023; 293: 121949. CrossRef - Contribution of Extracellular Vesicles and Molecular Chaperones in Age-Related Neurodegenerative Disorders of the CNS
Leila Noori, Kamila Filip, Zohreh Nazmara, Simin Mahakizadeh, Gholamreza Hassanzadeh, Celeste Caruso Bavisotto, Fabio Bucchieri, Antonella Marino Gammazza, Francesco Cappello, Maciej Wnuk, Federica Scalia
International Journal of Molecular Sciences.2023; 24(2): 927. CrossRef - Nucleic acid drug vectors for diagnosis and treatment of brain diseases
Zhi-Guo Lu, Jie Shen, Jun Yang, Jing-Wen Wang, Rui-Chen Zhao, Tian-Lu Zhang, Jing Guo, Xin Zhang
Signal Transduction and Targeted Therapy.2023;[Epub] CrossRef - Exosome-Based Carrier for RNA Delivery: Progress and Challenges
Yicheng Lu, Wei Huang, Meng Li, Aiping Zheng
Pharmaceutics.2023; 15(2): 598. CrossRef - MicroRNA‑124: an emerging therapeutic target in central nervous system disorders
Wen-Hao Zhang, Lian Jiang, Mei Li, Jing Liu
Experimental Brain Research.2023; 241(5): 1215. CrossRef - The role of exosomes in pathogenesis and the therapeutic efficacy of mesenchymal stem cell-derived exosomes against Parkinson’s disease
Maryam Abrishamdar, Maryam Sadat Jalali, Neda Yazdanfar
Neurological Sciences.2023; 44(7): 2277. CrossRef - Small Extracellular Vesicles’ miRNAs: Biomarkers and Therapeutics for Neurodegenerative Diseases
Wei Qing Lim, Kie Hoon Michelle Luk, Kah Yee Lee, Nasuha Nurul, Sin Jade Loh, Zhen Xiong Yeow, Qi Xuan Wong, Qi Hao Daniel Looi, Pan Pan Chong, Chee Wun How, Sharina Hamzah, Jhi Biau Foo
Pharmaceutics.2023; 15(4): 1216. CrossRef - Bioengineered Mesenchymal-Stromal-Cell-Derived Extracellular Vesicles as an Improved Drug Delivery System: Methods and Applications
Cristiana Ulpiano, Cláudia L. da Silva, Gabriel A. Monteiro
Biomedicines.2023; 11(4): 1231. CrossRef - Mmu-miR-25-3p promotes macrophage autophagy by targeting DUSP10 to reduce mycobacteria survival
Wenqi Yuan, Xuehua Zhan, Wei Liu, Rong Ma, Yueyong Zhou, Guangxian Xu, Zhaohui Ge
Frontiers in Cellular and Infection Microbiology.2023;[Epub] CrossRef - Exosomes in brain diseases: Pathogenesis and therapeutic targets
Qingying Si, Linlin Wu, Deshui Pang, Pei Jiang
MedComm.2023;[Epub] CrossRef - Repressor Element-1 Binding Transcription Factor (REST) as a Possible Epigenetic Regulator of Neurodegeneration and MicroRNA-Based Therapeutic Strategies
Ajmal Nassar, Sairaj Satarker, Prasada Chowdari Gurram, Dinesh Upadhya, SM Fayaz, Madhavan Nampoothiri
Molecular Neurobiology.2023; 60(10): 5557. CrossRef - Standardization Approaches for Extracellular Vesicle Loading with Oligonucleotides and Biologics
Josepha Roerig, Michaela Schulz‐Siegmund
Small.2023;[Epub] CrossRef - Small extracellular vesicles: a novel drug delivery system for neurodegenerative disorders
Renjie Pan, Dongdong Chen, Lanlan Hou, Rong Hu, Zhigang Jiao
Frontiers in Aging Neuroscience.2023;[Epub] CrossRef - Mesenchymal Stem Cell-Derived Extracellular Vesicles: An Emerging Diagnostic and Therapeutic Biomolecules for Neurodegenerative Disabilities
Mahmoud Kandeel, Mohamed A. Morsy, Khalid M. Alkhodair, Sameer Alhojaily
Biomolecules.2023; 13(8): 1250. CrossRef - Research progress on the role of extracellular vesicles in neurodegenerative diseases
Zhengzhe Li, Xiaoling Wang, Xiaoxing Wang, Xiaomei Yi, Yin Kwan Wong, Jiyang Wu, Fangfang Xie, Die Hu, Qi Wang, Jigang Wang, Tianyu Zhong
Translational Neurodegeneration.2023;[Epub] CrossRef - Exosomes as cell-derivative carriers in the diagnosis and treatment of central nervous system diseases
Gayatri Gopal Shetgaonkar, Shirleen Miriam Marques, Cleona E. M. DCruz, R. J. A. Vibhavari, Lalit Kumar, Rupesh Kalidas Shirodkar
Drug Delivery and Translational Research.2022; 12(5): 1047. CrossRef - Emerging prospects of extracellular vesicles for brain disease theranostics
Ruoning Wang, Xiaohong Wang, Yuting Zhang, Huacong Zhao, Jiwei Cui, Junsong Li, Liuqing Di
Journal of Controlled Release.2022; 341: 844. CrossRef - Stem Cell Transplantation Therapy and Neurological Disorders: Current Status and Future Perspectives
Md. Rahman, Md. Islam, Mohammad Islam, Md. Harun-Or-Rashid, Mahfuzul Islam, Sabirin Abdullah, Mohammad Uddin, Sumit Das, Md. Rahaman, Muniruddin Ahmed, Fahad Alhumaydhi, Talha Emran, Amany Mohamed, Mohammad Faruque, Mayeen Khandaker, Gomaa Mostafa-Hedeab
Biology.2022; 11(1): 147. CrossRef - Non-viral vectors for RNA delivery
Yi Yan, Xiao-Yu Liu, An Lu, Xiang-Yu Wang, Lin-Xia Jiang, Jian-Cheng Wang
Journal of Controlled Release.2022; 342: 241. CrossRef - MicroRNA-22: a Novel and Potent Biological Therapeutics in Neurological Disorders
Seyed Hamidreza Rastegar-moghaddam, Alireza Ebrahimzadeh-Bideskan, Sara Shahba, Amir Mohammad Malvandi, Abbas Mohammadipour
Molecular Neurobiology.2022; 59(5): 2694. CrossRef - Functional Extracellular Vesicles for Regenerative Medicine
Han Young Kim, Seunglee Kwon, Wooram Um, Sol Shin, Chan Ho Kim, Jae Hyung Park, Byung‐Soo Kim
Small.2022;[Epub] CrossRef - Emerging role of exosomes in the pathology of chronic obstructive pulmonary diseases; destructive and therapeutic properties
Hadi Rajabi, Nur Konyalilar, Sinem Erkan, Deniz Mortazavi, Seval Kubra Korkunc, Ozgecan Kayalar, Hasan Bayram, Reza Rahbarghazi
Stem Cell Research & Therapy.2022;[Epub] CrossRef - MicroRNA Alteration, Application as Biomarkers, and Therapeutic Approaches in Neurodegenerative Diseases
T. P. Nhung Nguyen, Mandeep Kumar, Ernesto Fedele, Giambattista Bonanno, Tiziana Bonifacino
International Journal of Molecular Sciences.2022; 23(9): 4718. CrossRef - Protective Signature of IFNγ-Stimulated Microglia Relies on miR-124-3p Regulation From the Secretome Released by Mutant APP Swedish Neuronal Cells
Gonçalo Garcia, Adelaide Fernandes, Frank Stein, Dora Brites
Frontiers in Pharmacology.2022;[Epub] CrossRef - Therapeutic Potential of Exosomes Derived from Diabetic Adipose Stem Cells in Cutaneous Wound Healing of db/db Mice
Hsiang-Hao Hsu, Aline Yen Ling Wang, Charles Yuen Yung Loh, Ashwin Alke Pai, Huang-Kai Kao
Pharmaceutics.2022; 14(6): 1206. CrossRef - Recent progresses in exosome-based systems for targeted drug delivery to the brain
Santosh Bashyal, Chhitij Thapa, Sangkil Lee
Journal of Controlled Release.2022; 348: 723. CrossRef - MicroRNA-124 attenuates PTSD-like behaviors and reduces the level of inflammatory cytokines by downregulating the expression of TRAF6 in the hippocampus of rats following single-prolonged stress
Yao Chen, Qi An, Shu-Ting Yang, Yu-Lu Chen, Lei Tong, Li-Li Ji
Experimental Neurology.2022; 356: 114154. CrossRef - Exosomes─Nature’s Lipid Nanoparticles, a Rising Star in Drug Delivery and Diagnostics
Rumiana Tenchov, Janet M. Sasso, Xinmei Wang, Wen-Shing Liaw, Chun-An Chen, Qiongqiong Angela Zhou
ACS Nano.2022; 16(11): 17802. CrossRef - The emerging role of long non-coding RNAs, microRNAs, and an accelerated epigenetic age in Huntington’s disease
Soudeh Ghafouri-Fard, Tayyebeh Khoshbakht, Bashdar Mahmud Hussen, Mohammad Taheri, Kaveh Ebrahimzadeh, Rezvan Noroozi
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - Intracellular and intercellular transport of RNA organelles in CXG repeat disorders: The strength of weak ties
Deepti Kailash Nabariya, Annika Heinz, Sabrina Derksen, Sybille Krauß
Frontiers in Molecular Biosciences.2022;[Epub] CrossRef - Extracellular vesicles in neurodegenerative diseases: Insights and new perspectives
Qian Yuan, Xiao-dong Li, Si-miao Zhang, Hong-wei Wang, Yun-liang Wang
Genes & Diseases.2021; 8(2): 124. CrossRef - Therapeutic antisense oligonucleotides for movement disorders
Epaminondas Doxakis
Medicinal Research Reviews.2021; 41(5): 2656. CrossRef - An update on the role of miR-124 in the pathogenesis of human disorders
Soudeh Ghafouri-Fard, Hamed Shoorei, Zahra Bahroudi, Atefe Abak, Jamal Majidpoor, Mohammad Taheri
Biomedicine & Pharmacotherapy.2021; 135: 111198. CrossRef - Ancient Evolutionary Origin and Properties of Universally Produced Natural Exosomes Contribute to Their Therapeutic Superiority Compared to Artificial Nanoparticles
Phillip W. Askenase
International Journal of Molecular Sciences.2021; 22(3): 1429. CrossRef - Intriguing Biomedical Applications of Synthetic and Natural Cell-Derived Vesicles: A Comparative Overview
Brateen Datta, Debashish Paul, Uttam Pal, Tatini Rakshit
ACS Applied Bio Materials.2021; 4(4): 2863. CrossRef - Nucleic acid delivery with extracellular vesicles
Michaela Schulz-Siegmund, Achim Aigner
Advanced Drug Delivery Reviews.2021; 173: 89. CrossRef - Emerging Roles of Exosomes in Huntington’s Disease
Hanadi Ananbeh, Petr Vodicka, Helena Kupcova Skalnikova
International Journal of Molecular Sciences.2021; 22(8): 4085. CrossRef - Stem Cell-Derived Exosomes: a New Strategy of Neurodegenerative Disease Treatment
Nashmin Fayazi, Mohsen Sheykhhasan, Sara Soleimani Asl, Rezvan Najafi
Molecular Neurobiology.2021; 58(7): 3494. CrossRef - Altered microRNA expression in animal models of Huntington’s disease and potential therapeutic strategies
Bridget Martinez, PhilipV Peplow
Neural Regeneration Research.2021; 16(11): 2159. CrossRef - Roles of glia-derived extracellular vesicles in central nervous system diseases: an update
Hao Sun, Xiaojuan Su, Shiping Li, Dezhi Mu, Yi Qu
Reviews in the Neurosciences.2021; 32(8): 833. CrossRef - Oligonucleotides as therapeutic tools for brain disorders: Focus on major depressive disorder and Parkinson's disease
Analia Bortolozzi, Sharon Manashirov, Alon Chen, Francesc Artigas
Pharmacology & Therapeutics.2021; 227: 107873. CrossRef - Perspective insights and application of exosomes as a novel tool against neurodegenerative disorders: An expository appraisal
Amey Kadbhane, Monica Patel, Saurabh Srivastava, Pankaj Kumar Singh, Jitender Madan, Shashi Bala Singh, Dharmendra Kumar Khatri
Journal of Drug Delivery Science and Technology.2021; 63: 102526. CrossRef - Harnessing the therapeutic potential of extracellular vesicles for cancer treatment
Ying Zhang, Yu Xiao, Gaoge Sun, Xue Jin, Lerui Guo, Tian Li, Hang Yin
Seminars in Cancer Biology.2021; 74: 92. CrossRef - Neural Stem Cell-Derived Exosomes Regulate Neural Stem Cell Differentiation Through miR-9-Hes1 Axis
Ping Yuan, Lu Ding, Huili Chen, Yi Wang, Chunhong Li, Shu Zhao, Xiaoyu Yang, Yizhao Ma, Jie Zhu, Xinrui Qi, Yanyan Zhang, Xiaohuan Xia, Jialin C. Zheng
Frontiers in Cell and Developmental Biology.2021;[Epub] CrossRef - Diagnostic and Therapeutic Potential of Exosomal MicroRNAs for Neurodegenerative Diseases
Miao He, Hai-nan Zhang, Zhen-chu Tang, Shu-guang Gao, Laura Baroncelli
Neural Plasticity.2021; 2021: 1. CrossRef - Profiling of Extracellular Small RNAs Highlights a Strong Bias towards Non-Vesicular Secretion
Helena Sork, Mariana Conceicao, Giulia Corso, Joel Nordin, Yi Xin Fiona Lee, Kaarel Krjutskov, Jakub Orzechowski Westholm, Pieter Vader, Marie Pauwels, Roosmarijn E. Vandenbroucke, Matthew JA Wood, Samir EL Andaloussi, Imre Mäger
Cells.2021; 10(6): 1543. CrossRef - Neuroinflammation: The next target of exosomal microRNAs derived from mesenchymal stem cells in the context of neurological disorders
Mehrdad Hajinejad, Sajad Sahab‐Negah
Journal of Cellular Physiology.2021; 236(12): 8070. CrossRef - Exosomal microRNAs from mesenchymal stem/stromal cells: Biology and applications in neuroprotection
Aida Nasirishargh, Priyadarsini Kumar, Lalithasri Ramasubramanian, Kaitlin Clark, Dake Hao, Sabrina V Lazar, Aijun Wang
World Journal of Stem Cells.2021; 13(7): 776. CrossRef - Central Nervous System Cell-Derived Exosomes in Neurodegenerative Diseases
Yang Tian, Chen Fu, Yifan Wu, Yao Lu, Xuemei Liu, Yunling Zhang, Yanfang Xian
Oxidative Medicine and Cellular Longevity.2021; 2021: 1. CrossRef - The regulatory roles of microRNAs toward pathogenesis and treatments in Huntington's disease
Chih-Wei Tung, Pin-Yu Huang, Siew Chin Chan, Pei-Hsun Cheng, Shang-Hsun Yang
Journal of Biomedical Science.2021;[Epub] CrossRef - Dosing extracellular vesicles
Dhanu Gupta, Antje Maria Zickler, Samir El Andaloussi
Advanced Drug Delivery Reviews.2021; 178: 113961. CrossRef - Therapeutic Potential of Nucleic Acids when Combined with Extracellular Vesicles
Brian Jurgielewicz, Steven Stice, Yao Yao
Aging and disease.2021; 12(6): 1476. CrossRef - Effects of exosomal miRNAs in the diagnosis and treatment of Alzheimer’s disease
Guo-hong Cui, Jing Zhu, Ya-chao Wang, Jing Wu, Jian-ren Liu, Hai-dong Guo
Mechanisms of Ageing and Development.2021; 200: 111593. CrossRef - MicroRNA-124: A Key Player in Microglia-Mediated Inflammation in Neurological Diseases
Jiuhan Zhao, Zhenwei He, Jialu Wang
Frontiers in Cellular Neuroscience.2021;[Epub] CrossRef - DNAJB6b-enriched small extracellular vesicles decrease polyglutamine aggregation in in vitro and in vivo models of Huntington disease
Bhagyashree S. Joshi, Sameh A. Youssef, Reinier Bron, Alain de Bruin, Harm H. Kampinga, Inge S. Zuhorn
iScience.2021; 24(11): 103282. CrossRef - Diagnostic and Therapeutic Potential of Exosomes in Neurodegenerative Diseases
Panyue Gao, Xinrong Li, Xinzhe Du, Sha Liu, Yong Xu
Frontiers in Aging Neuroscience.2021;[Epub] CrossRef - MiR-124 and the Underlying Therapeutic Promise of Neurodegenerative Disorders
Dong Han, Xiaoyu Dong, Dongming Zheng, Jianfei Nao
Frontiers in Pharmacology.2020;[Epub] CrossRef - Circulating Exosomal miRNA as Diagnostic Biomarkers of Neurodegenerative Diseases
Lin Wang, Lijuan Zhang
Frontiers in Molecular Neuroscience.2020;[Epub] CrossRef - Current Status of microRNA-Based Therapeutic Approaches in Neurodegenerative Disorders
Sujay Paul, Luis Alberto Bravo Vázquez, Samantha Pérez Uribe, Paula Roxana Reyes-Pérez, Ashutosh Sharma
Cells.2020; 9(7): 1698. CrossRef - Progress in the molecular pathogenesis and nucleic acid therapeutics for Parkinson's disease in the precision medicine era
Dunhui Li, Frank L. Mastaglia, Sue Fletcher, Steve D. Wilton
Medicinal Research Reviews.2020; 40(6): 2650. CrossRef - MicroRNAs Dysregulation and Mitochondrial Dysfunction in Neurodegenerative Diseases
Mariano Catanesi, Michele d’Angelo, Maria Grazia Tupone, Elisabetta Benedetti, Antonio Giordano, Vanessa Castelli, Annamaria Cimini
International Journal of Molecular Sciences.2020; 21(17): 5986. CrossRef - Exosome engineering: Current progress in cargo loading and targeted delivery
Shengyang Fu, Yi Wang, Xiaohuan Xia, Jialin C. Zheng
NanoImpact.2020; 20: 100261. CrossRef - Strategies and materials of "SMART" non-viral vectors: Overcoming the barriers for brain gene therapy
Yan Li, Linying Liu, Weihong Ji, Huang Peng, Ruichen Zhao, Xin Zhang
Nano Today.2020; 35: 101006. CrossRef - Therapeutic miRNA-Enriched Extracellular Vesicles: Current Approaches and Future Prospects
Javaria Munir, Jeong Kyo Yoon, Seongho Ryu
Cells.2020; 9(10): 2271. CrossRef - The Role of Exosomal microRNAs and Oxidative Stress in Neurodegenerative Diseases
Xiaoyu Wang, Yunxiang Zhou, Qiannan Gao, Dongnan Ping, Yali Wang, Wei Wu, Xu Lin, Yuanjian Fang, Jianmin Zhang, Anwen Shao, Yanfang Xian
Oxidative Medicine and Cellular Longevity.2020; 2020: 1. CrossRef - Nanocarriers as a powerful vehicle to overcome blood-brain barrier in treating neurodegenerative diseases: Focus on recent advances
Xiaoqian Niu, Jiejian Chen, Jianqing Gao
Asian Journal of Pharmaceutical Sciences.2019; 14(5): 480. CrossRef - Blood exosomes as a tool for monitoring treatment efficacy and progression of neurodegenerative diseases
Hanin Abdel-Haq
Neural Regeneration Research.2019; 14(1): 72. CrossRef - MiR-124 Enriched Exosomes Promoted the M2 Polarization of Microglia and Enhanced Hippocampus Neurogenesis After Traumatic Brain Injury by Inhibiting TLR4 Pathway
Yongxiang Yang, Yuqin Ye, Chuiguang Kong, Xinhong Su, Xin Zhang, Wei Bai, Xiaosheng He
Neurochemical Research.2019; 44(4): 811. CrossRef - Resveratrol (3, 5, 4′-Trihydroxy-trans-Stilbene) Attenuates a Mouse Model of Multiple Sclerosis by Altering the miR-124/Sphingosine Kinase 1 Axis in Encephalitogenic T Cells in the Brain
K. Alexa Orr Gandy, Jiajia Zhang, Prakash Nagarkatti, Mitzi Nagarkatti
Journal of Neuroimmune Pharmacology.2019; 14(3): 462. CrossRef - Advances in therapeutic applications of extracellular vesicles
Oscar P. B. Wiklander, Meadhbh Á. Brennan, Jan Lötvall, Xandra O. Breakefield, Samir EL Andaloussi
Science Translational Medicine.2019;[Epub] CrossRef - Exosome-mediated therapeutic delivery: A new horizon for human neurodegenerative disorders’ treatment (with a focus on siRNA delivery improvement)
Nayer Seyfizadeh, Narges Seyfizadeh, Sajad Borzouisileh, Farideh Elahimanesh, Vahid Hosseini, Mohammad Nouri
Process Biochemistry.2019; 85: 164. CrossRef - Exosomes released from neural progenitor cells and induced neural progenitor cells regulate neurogenesis through miR-21a
Yizhao Ma, Chunhong Li, Yunlong Huang, Yi Wang, Xiaohuan Xia, Jialin C. Zheng
Cell Communication and Signaling.2019;[Epub] CrossRef - Exosomal miRNAs in central nervous system diseases: biomarkers, pathological mediators, protective factors and therapeutic agents
Xiaohuan Xia, Yi Wang, Yunlong Huang, Han Zhang, Hongfang Lu, Jialin C. Zheng
Progress in Neurobiology.2019; 183: 101694. CrossRef - Role of Exosomes in Central Nervous System Diseases
Wanying Liu, Xiaodan Bai, Ao Zhang, Juanjuan Huang, Shixin Xu, Junping Zhang
Frontiers in Molecular Neuroscience.2019;[Epub] CrossRef - miR-124 and Parkinson’s disease: A biomarker with therapeutic potential
Efthalia Angelopoulou, Yam Nath Paudel, Christina Piperi
Pharmacological Research.2019; 150: 104515. CrossRef - The Potential Role of MicroRNA-124 in Cerebral Ischemia Injury
Xiaolu Liu, Zhitao Feng, Lipeng Du, Yaguang Huang, Jinwen Ge, Yihui Deng, Zhigang Mei
International Journal of Molecular Sciences.2019; 21(1): 120. CrossRef - Epigenetic regulation of astrocyte function in neuroinflammation and neurodegeneration
Matthew Neal, Jason R. Richardson
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease.2018; 1864(2): 432. CrossRef - Potential of Extracellular Vesicles in Neurodegenerative Diseases: Diagnostic and Therapeutic Indications
Mehrnaz Izadpanah, Arshia Seddigh, Somayeh Ebrahimi Barough, Seyed Abolhassan Shahzadeh Fazeli, Jafar Ai
Journal of Molecular Neuroscience.2018; 66(2): 172. CrossRef - Regenerative Approaches in Huntington’s Disease: From Mechanistic Insights to Therapeutic Protocols
Jenny Sassone, Elsa Papadimitriou, Dimitra Thomaidou
Frontiers in Neuroscience.2018;[Epub] CrossRef - Exosome-based small RNA delivery: progress and prospects
Mei Lu, Haonan Xing, Zhe Xun, Tianzhi Yang, Pingtian Ding, Cuifang Cai, Dongkai Wang, Xiaoyun Zhao
Asian Journal of Pharmaceutical Sciences.2017;[Epub] CrossRef - Extracellular Vesicles in Brain Tumors and Neurodegenerative Diseases
Federica Ciregia, Andrea Urbani, Giuseppe Palmisano
Frontiers in Molecular Neuroscience.2017;[Epub] CrossRef - The miR-124 family of microRNAs is critical for regeneration of the brain and visual system in the planarian Schmidtea mediterranea
Sasidharan Vidyanand, Srujan Marepally, Sarah A. Elliott, Srishti Baid, Vairavan Lakshmanan, Nishtha Nayyar, Dhiru Bansal, Alejandro Sánchez Alvarado, Praveen Kumar Vemula, Dasaradhi Palakodeti
Development.2017;[Epub] CrossRef - Mutant Huntingtin Inhibits αB-Crystallin Expression and Impairs Exosome Secretion from Astrocytes
Yan Hong, Ting Zhao, Xiao-Jiang Li, Shihua Li
The Journal of Neuroscience.2017; 37(39): 9550. CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite