Articles
- Page Path
- HOME > J Mov Disord > Volume 12(1); 2019 > Article
-
Review Article
Abnormal Eye Movements in Parkinsonism and Movement Disorders -
Ileok Jung1, Ji-Soo Kim2,3

-
Journal of Movement Disorders 2019;12(1):1-13.
DOI: https://doi.org/10.14802/jmd.18034
Published online: January 30, 2019
1Department of Neurology, Korea University College of Medicine, Korea University Ansan Hospital, Ansan, Korea
2Department of Neurology, Seoul National University College of Medicine, Seoul, Korea
3Dizziness Center, Clinical Neuroscience Center, and Department of Neurology, Seoul National University Bundang Hospital, Seongnam, Korea
- Corresponding author: Ji-Soo Kim, MD, PhD https://orcid.org/0000-0002-1508-2024 Department of Neurology, Seoul National University College of Medicine, Seoul National University Bundang Hospital, 82 Gumi-ro 173beon-gil, Bundang-gu, Seongnam 13620, Korea Tel: +82-31-787-7463 Fax: +82-31-787-4059 E-mail: jisookim@snu.ac.kr
• Received: July 18, 2018 • Revised: November 7, 2018 • Accepted: December 12, 2018
Copyright © 2019 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Abnormal eye movements are commonly observed in movement disorders. Ocular motility examination should include bedside evaluation and laboratory recording of ocular misalignment, involuntary eye movements, including nystagmus and saccadic intrusions/oscillations, triggered nystagmus, saccades, smooth pursuit (SP), and the vestibulo-ocular reflex. Patients with Parkinson’s disease (PD) mostly show hypometric saccades, especially for the self-paced saccades, and impaired SP. Early vertical saccadic palsy is characteristic of progressive supranuclear palsy-Richardson’s syndrome. Patients with cortico-basal syndrome typically show a delayed onset of saccades. Downbeat and gaze-evoked nystagmus and hypermetric saccades are characteristic ocular motor findings in ataxic disorders due to cerebellar dysfunction. In this review, we discuss various ocular motor findings in movement disorders, including PD and related disorders, ataxic syndromes, and hyperkinetic movement disorders. Systemic evaluation of the ocular motor functions may provide valuable information for early detection and monitoring of movement disorders, despite an overlap in the abnormal eye movements among different movement disorders.
- Parkinson’s disease
- In general, ocular motor deficits in PD are not as prominent as in progressive supranuclear palsy syndrome (PSPS) or Huntington’s disease (HD) and frequently require laboratory testing to bring out abnormalities [1]. In PD, steady fixation may be disrupted by saccadic intrusions, such as SWJs, that are characterized by involuntary saccadic movements from and back to the fixation point with an intersaccadic interval of approximately 200 ms and an amplitude of 0.5°–5° [8]. SWJs may be observed in normal elderly subjects [9], but the frequent and large SWJs in PD have been ascribed to compensatory increased activity in the frontal eye field [5,10].
- Saccades are typically hypometric, especially vertically [5]. Downward gaze paresis is not seen in PD, and if it is present, one should consider the diagnosis of progressive supranuclear palsy-Richardson’s syndrome (PSP-RS) (Table 1) [2,11]. Most patients with PD have difficulty making self-paced saccades between two continuously visible targets. When patients are verbally instructed to look between two widely spaced targets, PD patients typically make near-accurate saccades. However, when they are instructed to maintain this activity on their own, their saccades invariably become hypometric [12]. Hypometric voluntary saccades are supposed to be caused by increased inhibition of the superior colliculus (SC) and reduced preoculomotor drive due to dysfunction of frontal-basal ganglia-SC circuits [5]. However, reflexive saccades can be generated by direct projections from the parietal cortex onto the saccade-related neurons in the intermediate layer of the SC [13]. The latency and velocity of reflexive saccades are usually normal in PD. Since patients with PD have impaired inhibition of reflexive saccades to visual stimuli, testing of antisaccades may reveal abnormal executive function, even in the early stage of PD [14]. In one study, errors in antisaccades were related to freezing of gait, and increased latency was associated with impaired postural control [15]. Impaired inhibition of unwanted saccades may be associated with dopaminergic depletion in the prefrontal cortex, resulting in lack of suppression of the SC by the basal ganglia.
- SP is usually impaired in PD [5]. Combined eye-head tracking is abnormal to a similar degree as SP with the head stationary in most patients with PD [16]. Even though low-frequency rotational and caloric vestibular responses may be reduced in patients with PD [17], the VOR gain is close to 1.0 at higher frequencies of head rotation, corresponding to natural activities, especially visual fixation [18]. Patients also show abnormal eyelid movements, including reduced blinking, lid retraction and lid lag [19,20]. Unlike patients with PSP-RS, patients with PD habituate their blink responses when a flashlight is repetitively shone into the eyes [19].
- Levodopa treatment may improve saccadic accuracy, SP, and convergence insufficiency [21-23]. Electrical stimulation of the pallidum or subthalamic nuclei was reported to improve performance on memory-guided or antisaccade tasks [24,25].
- Progressive supranuclear palsy syndrome
- PSPS, also known as the PSP-RS, is a clinical syndrome comprising vertical gaze palsy and balance difficulties with backward falls and mild dementia [26]. The disturbance of eye movements is usually present early in the course of the disease but occasionally develops later [27]. Patients with PSP-RS may present with visual complaints, such as blurred vision or photophobia [26].
- Steady fixation is frequently disrupted by saccadic intrusions [28-30]. The SWJs are more common and larger in PSP-RS than in other parkinsonian disorders [27,28]. The prominent saccadic intrusions might be related to involvement of the SC and the adjacent central midbrain reticular formation [31].
- Horizontal saccades are initially hypometric but normal in speed. As the disease progresses, they also become slow. Vertical saccades are slower than horizontal ones of similar size (Figure 1, Supplementary Video 1 in the online-only Data Supplement) [18,32]. The limitation of vertical saccades is more or less asymmetric [27]. Early on, vertical saccades may take a curved or oblique trajectory to the target (“round the houses” sign) [33]. During large-field, vertical optokinetic stimulation, PSP-RS patients often show tonic deviation of the eyes in the direction of stripe motion, with small or absent resetting quick phases [34]. Vertical saccades are generated by “burst neurons” in the midbrain but horizontal saccades are generated by burst neurons in the pons. Thus, the selective involvement of vertical saccades in the early stage of PSP-RS indicates that the initial pathology involves the midbrain burst neurons, or their local circuitry (SC and the adjacent central mesencephalic reticular formation) [32]. The latency of horizontal saccades may be prolonged in some patients with PSP-RS, but others retain the ability to make short-latency or “express” saccades [35]. Patients with PSP-RS also make errors in antisaccade tasks [35]. Both the presence of express saccades and errors in the antisaccade tasks suggest frontal lobe dysfunction [36].
- Vertical SP or combined eye-head tracking may be relatively spared. Eventually, SP and saccades are both lost, constituting voluntary gaze palsy. The angular VOR is relatively preserved until the late stages of PSP-RS, however, the linear VOR is markedly impaired [37,38]. Bell’s phenomenon is usually absent. Convergence eye movements are commonly impaired [39]. Late in the disease, the ocular motor deficit progresses into a complete ophthalmoplegia.
- Patients with PSP-RS may show several eyelid abnormalities, including blepharospasm, lid-opening or eye-closing apraxia, lid retraction, and lid lag [40,41]. Quite consistently, patients cannot inhibit a blink when a penlight is shone into the eyes (visual glabelar or Myerson’s sign) [40].
- Other disorders
- Patients with cortico-basal syndrome (CBS) frequently present with the clinical features of PSPS and may be misdiagnosed as having PSPS due to vertical supranuclear gaze palsy and falls within the first 2 years [18,42]. However, patients with CBS had a delayed onset of vertical supranuclear gaze palsy, i.e., more than 3 years after symptom onset, and had predominant downgaze abnormalities. Furthermore, patients with CBS show a significantly increased latency of horizontal saccades while the patients with PSPS mostly manifest with decreased saccadic velocity [18]. In the early stage of CBS, SP may be slow, but the range of movements is generally full. As the illness progresses, patients with CBS gradually lose the ability to make saccades to verbal commands, with retained spontaneous saccades and OKN [43]. Blepharospasm and eyelid opening apraxia may be observed [44].
- Multiple systemic atrophy (MSA) is subdivided into cerebellar (MSA-C) and parkinsonian MSA (MSA-P) [45]. Patients with MSA-C may show saccadic intrusions, downbeat and gazeevoked nystagmus, perverted downbeat head-shaking nystagmus and positional downbeat nystagmus along with impaired smooth ocular and eye-head pursuit and saccadic dysmetria [46,47].
- Dementia with Lewy bodies may be associated with vertical gaze palsy [48,49], but systematic measurements of vertical saccades are not available to date.
PARKINSON’S DISEASE AND RELATED DISORDERS
- Substantial advances have been made in defining and understanding the molecular biology of the hereditary ataxias [50]. With the identification of genes responsible for the hereditary ataxias, attempts have been made to identify distinctive syndromes of abnormal eye movements and to link those phenotypes with genotypes [51-54]. Even though findings are quite distinctive for some gene mutations causing ataxia, there is an overlap in the abnormal eye movements among different genotypes, and substantial phenotypic variation even in those with the same mutation [55].
- There are three principal cerebellar syndromes; 1) the syndrome of the flocculus and paraflocculus, 2) the syndrome of the nodulus and ventral uvula, and 3) the syndrome of the dorsal vermis (lobules VI and VII) and underlying caudal fastigial nuclei. In addition, the cerebellar hemispheres contribute to the control of eye movements [56]. Understanding the characteristic eye movement abnormalities in each syndrome seems vital for interpretation of underlying cerebellar dysfunction in various ataxic disorders.
- Lesions involving the flocculus and paraflocculus give rise to downbeat, and gaze-evoked and rebound nystagmus [57]. Inactivation of the flocculus severely impairs smooth tracking with the eyes alone, but active eye-head tracking is spared. Saccades are generally spared, but postsaccadic drift may be observed due to a pulse-step mismatch. Unilateral lesions produce ipsilateral deficits in pursuit and gaze holding. In patients with cerebellar disease, defects in SP, combined eye-head tracking, and gazeholding frequently occur together [58]. The VOR may by enhanced during low frequency and velocity stimulation, but may be impaired during HIT with high velocity and acceleration [59]. Adaptation of the VOR is also impaired in floccular lesions [60].
- Lesions of the nodulus and ventral uvula generate positional nystagmus, especially downbeat and apogeotropic, periodic alternating nystagmus (PAN), perverted downbeat head-shaking nystagmus, increase in the duration of vestibular responses, and a failure of tilt-suppression of postrotatory nystagmus [61-67].
- Experimental lesions of the dorsal vermis and fastigial nuclei cause saccadic dysmetria, typically hypometria if the vermis alone is involved, and hypermetria if the deep nuclei are involved [68]. Lesions involving the dorsal vermis also produce deficits of SP, especially at onset [69]. Patients with dorsal vermis lesions show moderate, ipsilateral deficits of sustained pursuit. Similarly, inactivation of one fastigial nucleus impairs the onset of contralateral pursuit but enhances acceleration of ipsilateral pursuit [69].
- Spinocerebellar ataxia
- Spinocerebellar Ataxia (SCA) is a group of autosomal dominant neurodegenerative disorders that are genetically and clinically heterogenous [50]. Previous reports have documented various abnormalities of eye movements in patients with SCA, providing clues for the differential diagnosis of SCA based on ocular motor phenotypes (Table 2) [51-54,70]. Due to a significant overlap in the phenotypes, however, caution is necessary in trying to assign a patient to one group on the basis of eye movement findings alone. Nonetheless, the presence of very slow saccades is suggestive of SCA2 (Figure 2), in which pontine saccadic burst neurons are involved. However, saccades may also be slow in patients with SCA1, SCA7, and SCA28. Impaired VOR is a common feature in SCA3. The presence of prominent downbeat, gaze-evoked, and rebound nystagmus with normal saccade velocity is typical of SCA6 and episodic ataxia type 2 (EA2).
- In SCA1, GEN and pursuit abnormalities are frequently observed, and the prevalence of ophthalmoplegia is higher than that of other SCAs. The presence of ophthalmoplegia is correlated with higher ataxia score and poorer functional stage [70]. This indicates involvement of ocular motor neurons in later stages of SCA1. Since the characteristic pathological finding of SCA2 is the loss of burst neurons in the brainstem, saccadic slowing as well as increased saccadic latency is common in this disorder [71]. During follow-up, annual progression rates are significantly higher in patients with SCA2 [72]. SCA3, also named Machado-Joseph disease, is the most common form of autosomal dominant ataxia. Nystagmus is present in 88% of patients with SCA3 and the symptomatic patients with nystagmus tend to have a higher length of the CAG tract in the expanded allele [73]. Saccadic intrusions such as SWJs are frequent in SCA3. SCA6 is regarded a pure cerebellar form of SCA [74,75]. Downbeat and gaze-evoked nystagmus are frequent in SCA6 [76]. Perverted downbeat head shaking nystagmus may be a distinct feature of SCA6 [76]. The VOR gain during horizontal HIT shows a negative correlation with the ataxia score while the VOR gain in response to relatively low frequency stimuli remains normal or increased regardless of disease severity (Figure 3) [77]. SCA7 is characterized by retinal degeneration and ophthalmoplegia [78]. Oculomotor findings of SCA7 include nystagmus, saccadic intrusions, saccadic hypoand hypermetria, slow saccades, impaired SP and VOR cancellation, and late in the illness, ophthalmoparesis and ophthalmoplegia [79]. SCA31 is also a pure cerebellar form of ataxia, making it difficult to distinguish SCA31 from SCA6 based on clinical findings only. The onset age is slightly higher, and the frequency of positional downbeat nystagmus is lower, in SCA31 than in SCA6 [80].
- Friedreich ataxia
- Friedreich ataxia (FA) is the most common cause of autosomal recessive ataxias with an onset usually before age 20 years. FA is characterized by ataxia, hyporeflexia, extensor plantar reflexes, neuropathy, cardiomyopathy, and diabetes. FA is mostly due to an unstable GAA repeat expansion within intron 1 of frataxin [81]. Abnormal ocular motor findings of FA include fixation instability manifesting as SWJs and ocular flutter [82,83]. While saccadic velocity is essentially normal, saccadic latency is prolonged. The latency correlates with clinical measures of disease severity. Saccades may be both hypo- and hypermetric. SP and the VOR may be impaired [84-86]. Caloric tests are abnormal in the majority of FA patients [85,87]. Thus, severe vestibulopathy with essentially normal saccadic velocity are hallmarks of FA and differentiate it from a number of dominant SCA [82].
- Episodic ataxia
- Episodic ataxia (EA) is a genetic disorder characterized by recurrent episodes of truncal ataxia and incoordination lasting minutes to hours [88]. EA is a heterogeneous group of disorders mostly with an autosomal dominant inheritance [89,90]. Several subtypes have been defined according to clinical and genetic characteristics [91]. Among the subtypes, EA2 is most common. Patients with EA2 mostly suffer from recurrent ataxia and slurred speech for several hours [88]. Even between the attacks, downbeat, gazeevoked, rebound and positional nystagmus are typically observed (Figure 4) [92]. The onset is mostly before the age of 20 years, but may be delayed [93]. The common triggers of the attacks are exercise, and physical or emotional stress [94]. Acetazolamide and aminopyridine have been found to reduce the frequency of attacks and improve the quality of life in patients with EA2 [88].
- Ataxia telangiectasia
- Ataxia telangiectasia (AT) is an autosomal recessive disorder characterized clinically by progressive ataxia, dystonia, parkinsonism, choreoathetosis, myoclonus, tremor, and ocular and cutaneous telangiectasia [95-97]. Patients with AT also have immunodeficiency, endocrine abnormalities, radiosensitivity, and a predisposition to neoplasia [98]. Eye movement abnormalities in AT include 1) spontaneous, gaze-evoked and periodic alternating nystagmus, microsaccadic oscillations, and SWJs [99], 2) impaired generation of volitional and reflexive saccades (characterized by prolonged latency and hypometria) in association with head “thrusts” during attempted shifts of gaze, and 3) impairment of pursuit eye movements, VOR and convergence [100,101]. These abnormal eye movements most likely result from dysfunction of the cerebellar flocculus and paraflocculus, and from abnormal supranuclear control of the SC due to dysfunction of the cerebellar vermis or the basal ganglia.
- Ataxia-oculomotor apraxia
- Ataxia-oculomotor apraxia (AOA) is an autosomal recessive disorder characterized by early onset ataxia, choreoathetosis, and ocular motor apraxia mimicking AT but without extraneurological features of this disease [102]. AOA type 1 (AOA1) is caused by mutations in the aprataxin gene (APTX) on chromosome 9p13 [103]. Aprataxin is a nuclear protein that repairs single-strand DNA breaks [104,105]. Patients with AOA1 also may show hypercholesterolemia, hypoalbuminaemia, and deficiency of muscle coenzyme Q10 [106,107]. Most patients with AOA1 show saccade initiation difficulties. When the head is restrained, patients make a series of small saccades (horizontal or vertical) at normal latency to shift their gaze. Although small, the saccades are normal in speed for their size. When the head is free, patients make large head movements that overshoot the target, with their eyes in contraversion, and then rotating their head back to bring their eyes back to the central position in the orbit. Other findings include SWJs, gaze-evoked nystagmus, and impaired SP [108].
- AOA type 2 (AOA2) is caused by mutations of the senataxin gene on chromosome 9q34, which also causes a failure to repair single-strand DNA breaks [109-112]. The clinical features of AOA2 are similar to those of AOA1 with prominent hypometria of horizontal and vertical saccades, requiring a staircase of saccades to shift the gaze, but with normal saccadic latency and speed [113]. Other ocular motor findings include SWJs, gaze-evoked, rebound, and downbeat nystagmus, impaired SP, vergence dysfunction with exophoria, and normal or increased vestibulo-ocular responses. However, head movements are not prominent in AOA2 and are not used to shift gaze by overshooting the target [113]. Patients also show impaired antisaccades and memoryguided saccades [113].
- Paraneoplastic cerebellar degeneration
- Patients with paraneoplastic cerebellar degeneration (PCD) usually present with rapidly progressive dizziness and imbalance. In addition to gait and appendicular ataxia, examination almost always shows abnormal eye movements due to cerebellar dysfunction, which include downbeat, gaze-evoked, seesaw and positional nystagmus, ocular flutter, opsoclonus, saccadic dysmetria, and impaired SP (Figure 5) [63,114-117].
ATAXIC DISORDERS
- Patients with HD often show impaired initiation of saccades with prolonged latencies, especially when the saccade is made to command or in anticipation of a predictable target [118]. Saccadic latency may be used as a marker of disease progression given its direct correlation with the severity of HD [119,120]. Patients frequently use an obligatory blink or head turn to start eye movements [118,121]. Paradoxically, patients often cannot suppress saccades during fixation, with increased visual distractibility [122]. Saccades to visual stimuli are made at normal latency, while those made to command are delayed [120]. Patients with HD also make increased errors during the antisaccade task. In preclinical stages of HD, increase in saccadic latency and error rates may predict the age of symptom onset [123]. In some patients, saccades may be slow in both horizontal and vertical planes. SP may also be impaired, but gaze holding and the VOR are well-preserved [121]. Fixation may be abnormal due to saccadic intrusions (Figure 6) [121].
HUNTINGTON’S DISEASE
- Essential tremor
- Compared with normal controls, patients with essential tremor more frequently show perverted downbeat head-shaking nystagmus, positional downbeat nystagmus, reduced gain of pursuit initiation, and impaired suppression of postrotational nystagmus with head tilt, all suggestive of underlying cerebellar dysfunction [124,125]. Another study reported higher prevalence of slowed SWJs and delayed saccades, which were independent of disease duration, tremor severity, and medication [126].
- Wilson’s disease
- Detection of Kayser-Fleischer rings is diagnostic of Wilson’s disease. Ocular motor abnormalities in Wilson’s disease include distractibility of gaze, slow vertical saccades, and lid-opening apraxia (Figure 7) [127,128].
- Whipple’s disease
- Whipple’s disease may mimic parkinsonism or PSPS by involving the basal ganglia and rostral mesencephalon [129]. Initially, vertical saccades may be slow and curved while horizontal saccades may be relatively preserved. It is almost always accompanied by a supranuclear vertical gaze palsy. Eventually, all eye movements may be lost. Oculomasticatory myorhythmia is virtually pathognomonic of this disease, and consists of rhythmic movements of the masticatory and occasionally other skeletal muscles, synchronized with pendular vergence oscillations of the eyes [130]. However, many patients with Whipple’s disease have no ocular oscillations.
OTHER DISORDERS
- Patients with movement disorders show nearly all kinds of eye movement abnormalities. In general, increased SI, increased saccadic latency and saccadic hypometria, and impaired SP are quite common in movement disorders and have little differential diagnostic value among the disorders. However, saccadic slowing indicates damage to the burst neurons in the pons (horizontal) and mesodiencephalic junction (vertical) and their outflow tracts and thus characterizes the disorders involving the brainstem. This phenomenon is also observed in PSPS and MSA. Decreased VOR also suggests damage to the brainstem and peripheral vestibular organs, In contrast, downbeat, gaze-evoked, rebound and positional nystagmus, hypermetric saccades, and hyperactive VOR are ocular motor findings specific for cerebellar dysfunction. Thus, despite the wide overlap in ocular motor abnormalities among movement disorders, scrutinized evaluation of all subclasses of eye movements would provide valuable information on the underlying pathology, differential diagnosis and monitoring of disease progression.
CONCLUSIONS
Supplementary Materials
Supplementary Video Legends
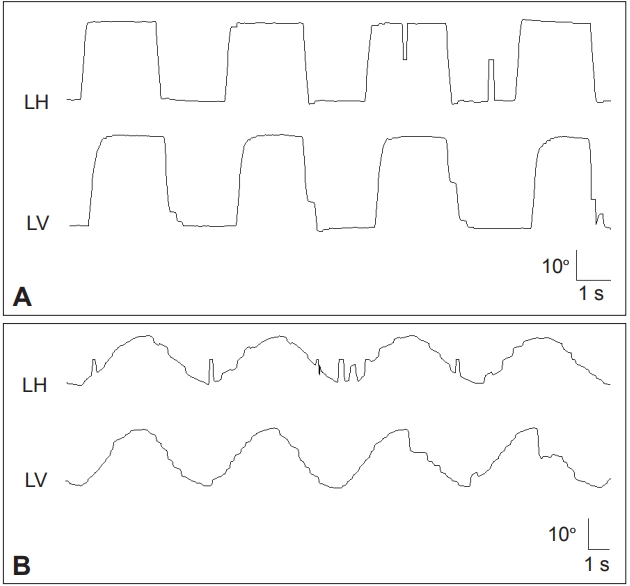
Figure 1.Frequent square wave jerks (A), slowed and hypometric saccades (B), and impaired SP are characteristic findings of progressive supranuclear palsy syndrome. Saccades and SP are more severely impaired in the vertical direction (B). LH: horizontal position of the left eye, LV: vertical position of the left eye, LT: torsional position of the left eye, SP: smooth pursuit.
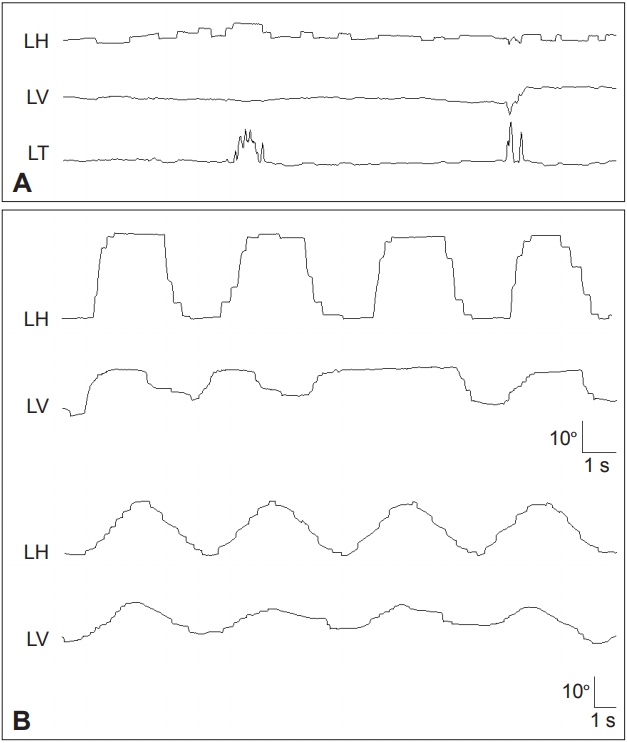

Figure 2.Patient with spinocerebellar ataxia type 2 shows slowed saccades especially in the vertical plane (A) and impaired smooth pursuit in both horizontal and vertical planes (B). LH: horizontal position of the left eye, LV: vertical position of the left eye.
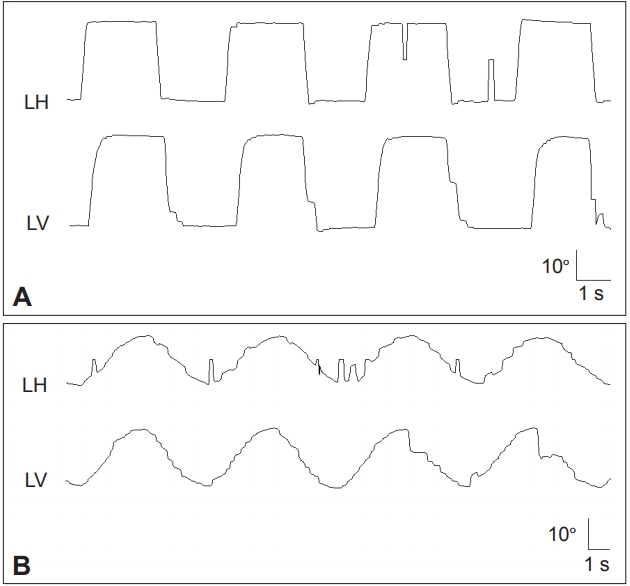

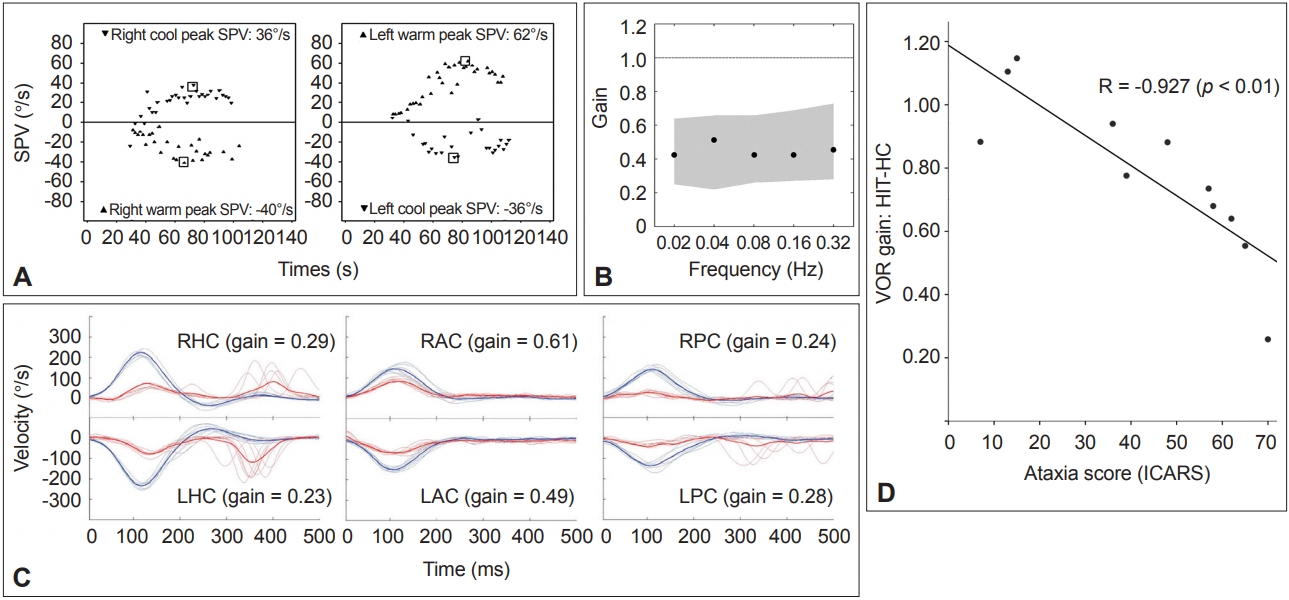
Figure 3.Results of the vestibular function tests in a representative patient with spinocerebellar ataxia type 6. A. Caloric tests. The sum of the peak SPV of the nystagmus in response to each caloric stimulus is increased at 174°/s (normal range=45–157°/s). B. Rotatory chair test. The gains of the VOR are normal during sinusoidal horizontal accelerations. Gray areas represent the normal ranges (mean ± 2SD). C. Head impulse tests. Head impulses in the plane of each SCC revealed decreased gain of the VOR for all six semicircular canals with overt catch-up saccades during stimulation of the horizontal and posterior SCCs. The blue lines indicate head velocity and the red lines represent eye velocity. D. The head impulse VOR gain for the HC is negatively correlated with the ataxia score as measured using the ICARS. SPV: slow-phase velocity, VOR: vestibulo-ocular reflex, SCC: semicircular canal, LAC: left anterior SCC, LHC: left horizontal SCC, LPC: left posterior SCC, RAC: right anterior SCC, RHC: right horizontal SCC, RPC: right posterior SCC, ICARS: International Cooperative Ataxia Rating Scale, HIT-HC: head impulse testing in the horizontal semicircular canal plane.
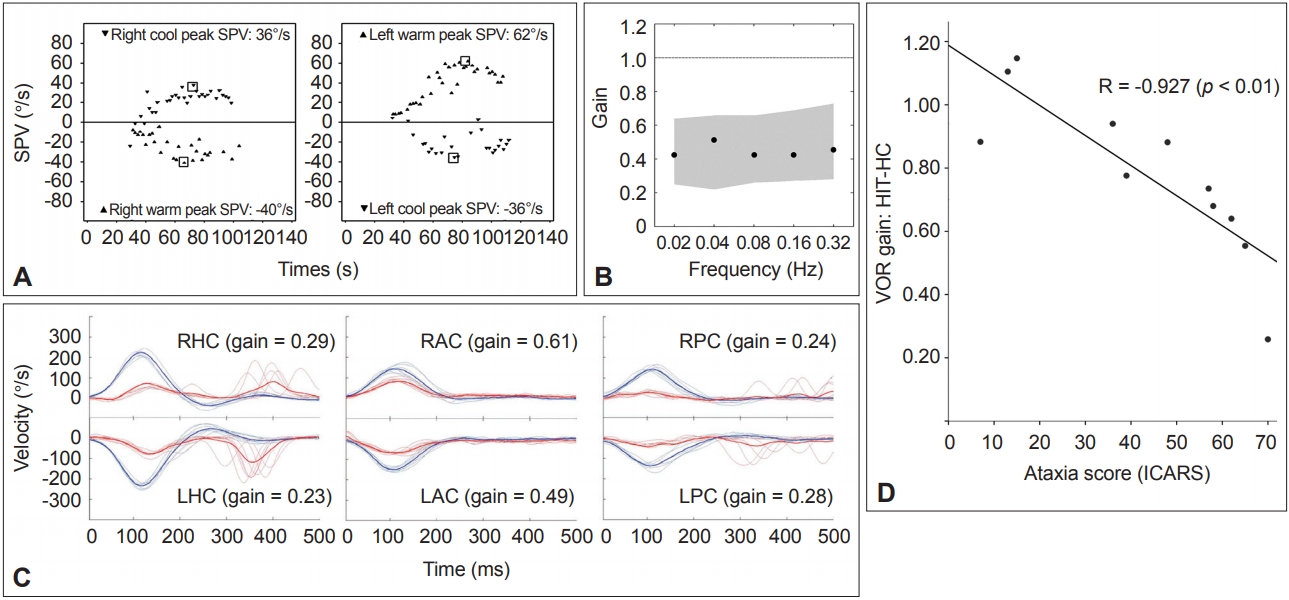

Figure 4.Findings in a patient with episodic ataxia type 2. A: The patient shows gaze-evoked (filled arrow) and downbeat nystagmus (open arrow) during leftward gaze that decrease gradually. On resuming the neutral position (arrow head), upbeat nystagmus develops without rebound nystagmus in the horizontal plane. Upward deflection indicates rightward and upward eye motion. H: horizontal eye position, V: vertical eye position. B: Pedigree of the patient. Squares males, circles females, open symbols unaffected, solid symbols affected. C. The chromatograms of a part of exon 31 and intron 31 of CACNA1A show a heterozygous point mutation at the splice donor site (c.4953+1G>A).
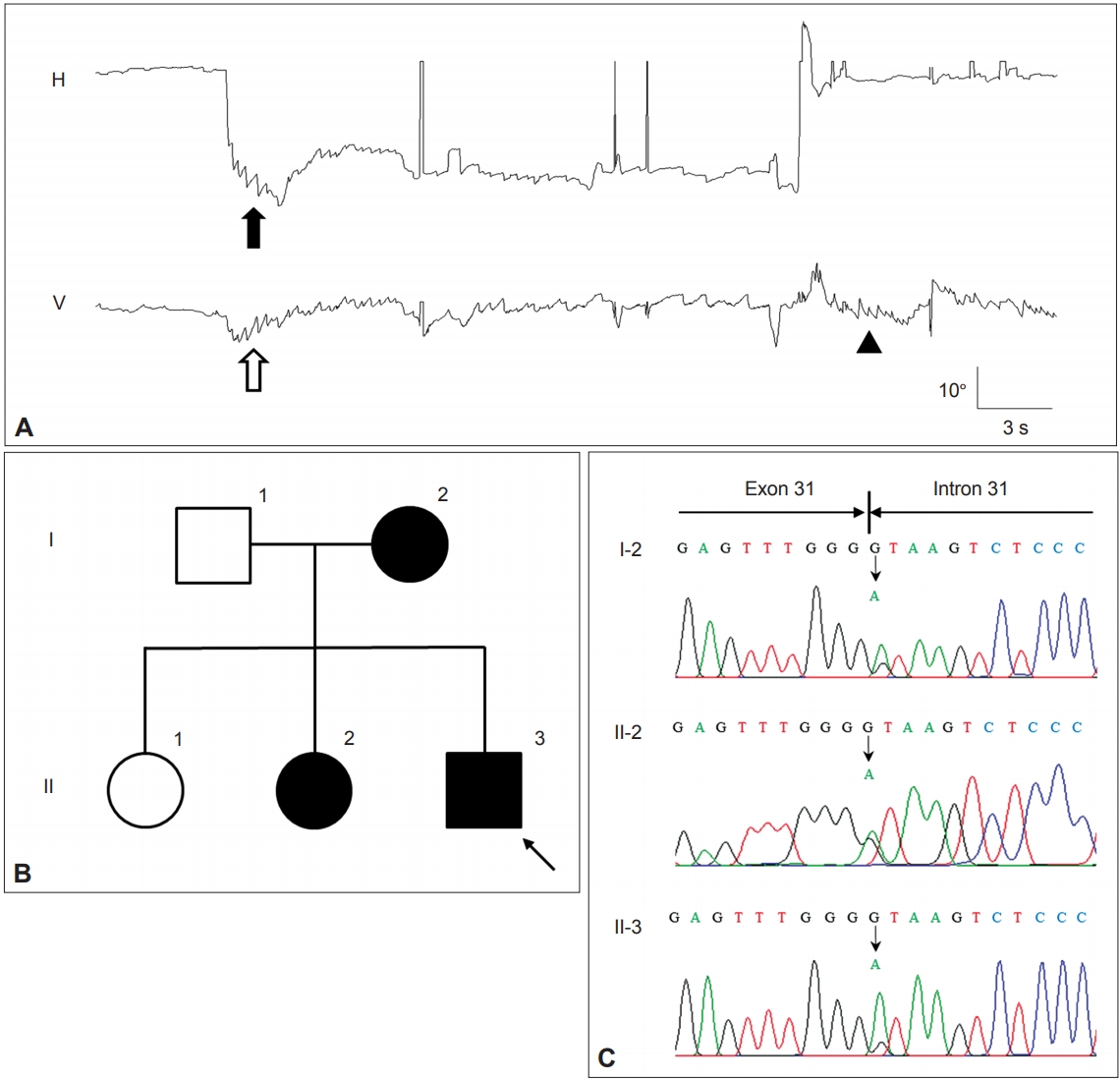

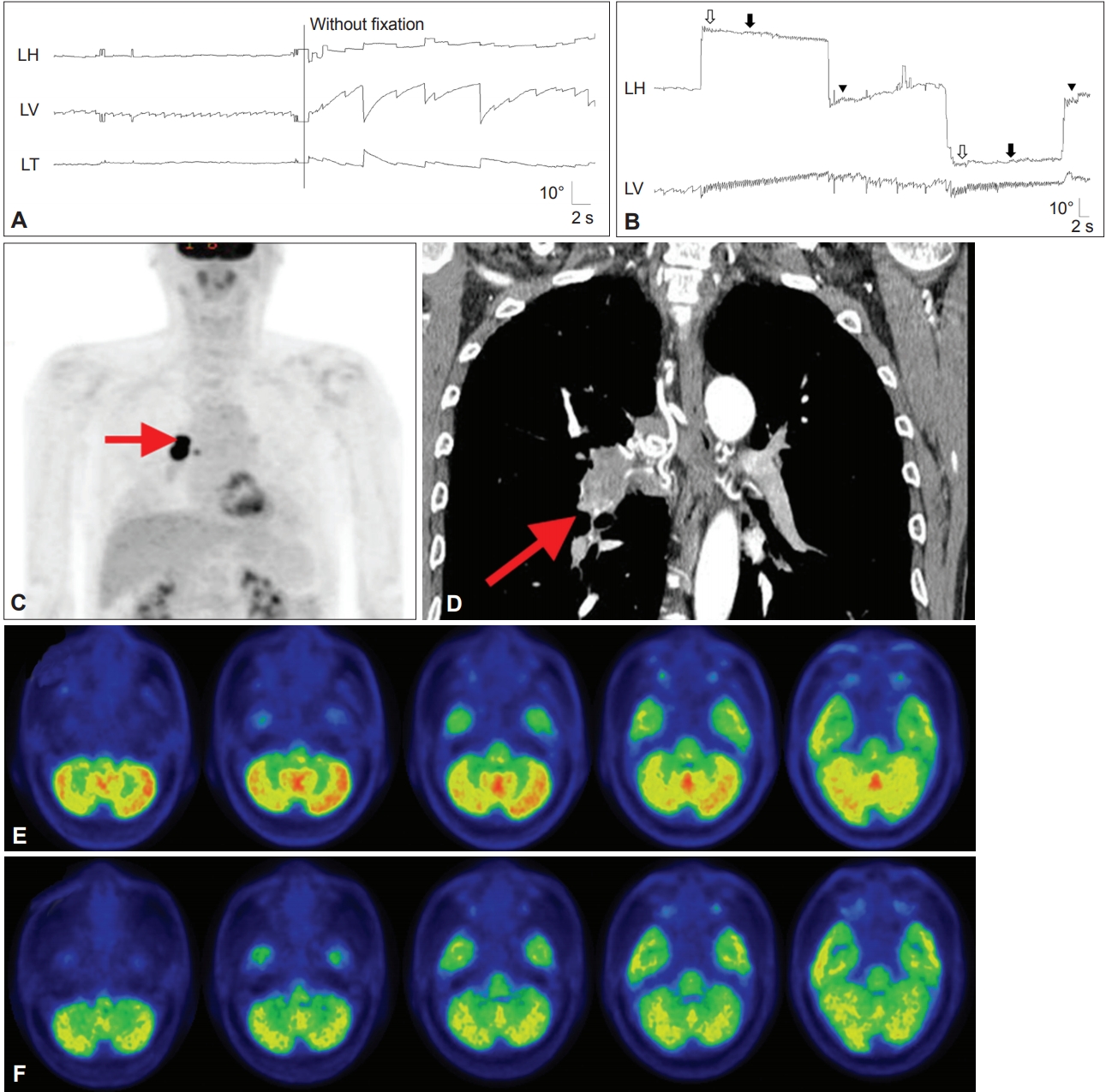
Figure 5.Findings in a patient with paraneoplastic cerebellar degeneration. A and B: Recording of eye movements using 3-dimensional video-oculography shows spontaneous downbeat nystagmus (DBN) with decreasing slow phases, which increases without fixation (A). DBN increases during lateral gazes with horizontal gaze-evoked (hollow arrows), centripetal (arrows), and rebound nystagmus (arrow heads) (B). C and D: Whole body 2-deoxy-2-[F18]fluoro-D-glucose-positron emission tomography (FDG-PET) (C) and chest CT (D) show a hypermetabolic mass in the right lower lobe of the lung (arrows). E: Brain FDG-PET reveals increased metabolism, especially in the nodulus. F: The hypermetabolism observed on the initial PET disappeared after right lung lobectomy and chemotherapy.


Figure 6.In a patient with Huntington’s disease, the horizontal saccades are markedly slowed (A). In the vertical plane, slowing is more prominent during downward saccades. In contrast, vertical SP is severely impaired while the horizontal SP is relatively preserved (B). LH: horizontal position of the left eye, LV: vertical position of the left eye, SP: smooth pursuit.
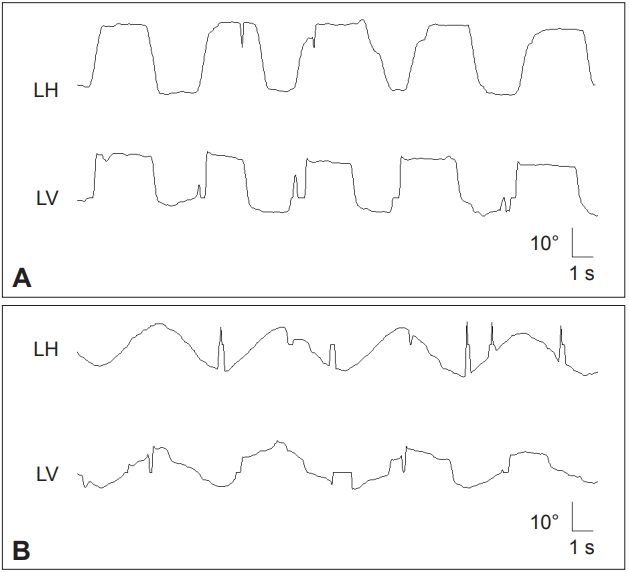

Figure 7.Findings in a patient with Wilson’s disease. A: Kayser-Fleischer ring. Color external photograph shows Kayser-Fleischer ring (arrows), a golden-brown discoloration of the cornea due to deposition of sulfur-copper complexes within the Descemet’s membrane. B: T2-weighted MRIs reveal symmetrical high signal intensity lesions in the putamina, thalami, midbrain and pontine tegmentum. C: Video-oculographic recording (SMI®, Teltow, Germany) of saccades shows selective slowing of downward saccades while the velocities of horizontal and upward saccades are normal. D: Smooth pursuit is impaired in the vertical direction. LH: horizontal position of the left eye, LV: vertical position of the left eye.
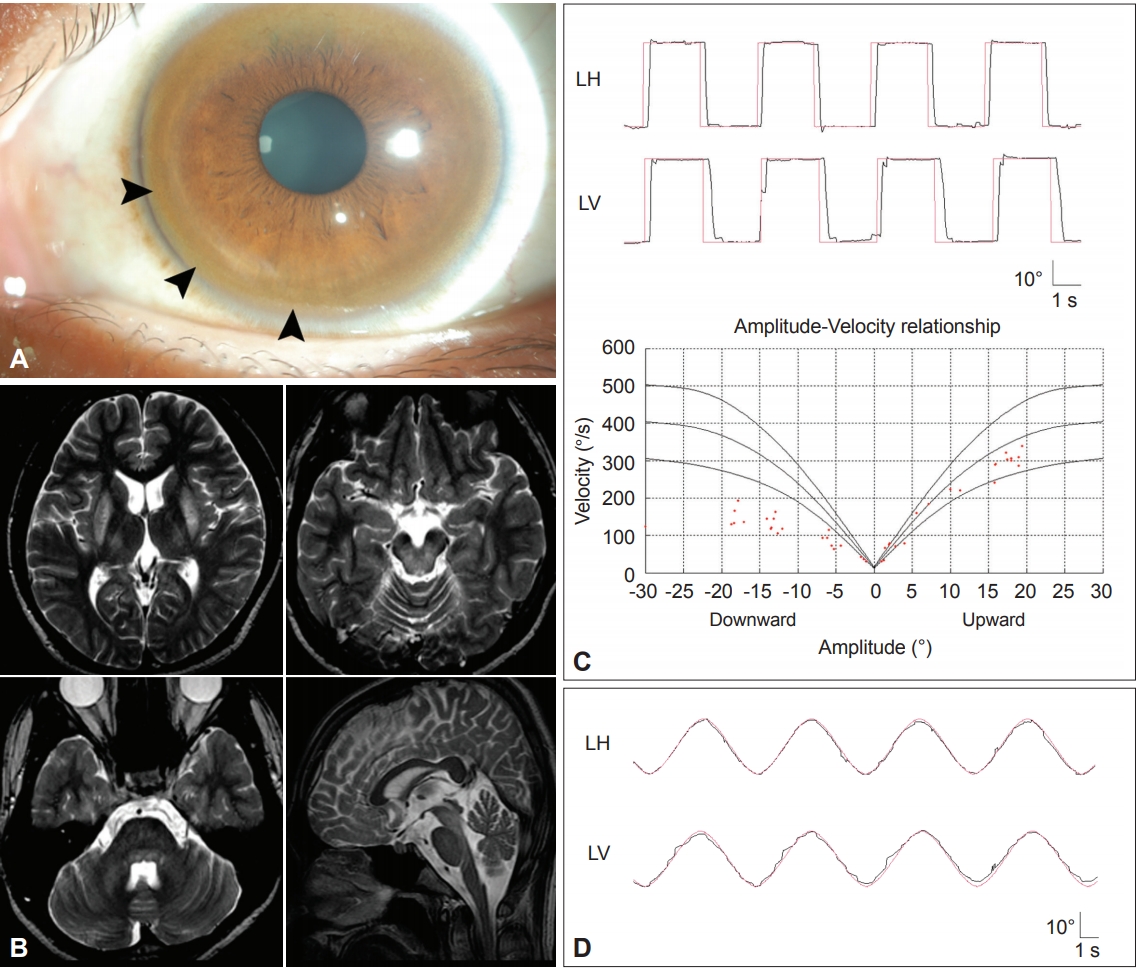

Table 1.Characteristics of ocular motor abnormalities in PD-related disorders
Table 2.Characteristic ocular motor abnormalities in ataxic disorders
- 1. Pinkhardt EH, Kassubek J. Ocular motor abnormalities in Parkinsonian syndromes. Parkinsonism Relat Disord 2011;17:223–230.ArticlePubMed
- 2. Antoniades CA, Kennard C. Ocular motor abnormalities in neurodegenerative disorders. Eye (Lond) 2015;29:200–207.ArticlePubMedPDF
- 3. Leigh JR, Zee DS. The neurology of eye movements. 5th ed. London: Oxford University Press; 2015.
- 4. Jung I, Kim JS. Approach to dizziness in the emergency department. Clin Exp Emerg Med 2015;2:75–88.ArticlePubMedPMCPDF
- 5. White OB, Saint-Cyr JA, Tomlinson RD, Sharpe JA. Ocular motor deficits in Parkinson’s disease: II. control of the saccadic and smooth pursuit systems. Brain 1983;106(Pt 3):571–587.ArticlePubMedPDF
- 6. Huh YE, Kim JS. Bedside evaluation of dizzy patients. J Clin Neurol 2013;9:203–213.ArticlePubMedPMC
- 7. Gaymard B, Pierrot-Deseilligny C. Neurology of saccades and smooth pursuit. Curr Opin Neurol 1999;12:13–19.ArticlePubMed
- 8. Herishanu YO, Sharpe JA. Normal square wave jerks. Invest Ophthalmol Vis Sci 1981;20:268–272.PubMed
- 9. Shallo-Hoffmann J, Sendler B, Muhlendyck H. Normal square wave jerks in differing age groups. Invest Ophthalmol Vis Sci 1990;31:1649–1652.PubMed
- 10. Sharpe JA, Herishanu YO, White OB. Cerebral square wave jerks. Neurology 1982;32:57–62.ArticlePubMed
- 11. Anderson T, Luxon L, Quinn N, Daniel S, David Marsden C, Bronstein A. Oculomotor function in multiple system atrophy: clinical and laboratory features in 30 patients. Mov Disord 2008;23:977–984.ArticlePubMed
- 12. Terao Y, Fukuda H, Ugawa Y, Hikosaka O. New perspectives on the pathophysiology of Parkinson’s disease as assessed by saccade performance: a clinical review. Clin Neurophysiol 2013;124:1491–1506.ArticlePubMed
- 13. May PJ. The mammalian superior colliculus: laminar structure and connections. Prog Brain Res 2006;151:321–378.ArticlePubMed
- 14. Antoniades CA, Demeyere N, Kennard C, Humphreys GW, Hu MT. Antisaccades and executive dysfunction in early drug-naive Parkinson’s disease: the discovery study. Mov Disord 2015;30:843–847.ArticlePubMed
- 15. Walton CC, O’Callaghan C, Hall JM, Gilat M, Mowszowski L, Naismith SL, et al. Antisaccade errors reveal cognitive control deficits in Parkinson’s disease with freezing of gait. J Neurol 2015;262:2745–2754.ArticlePubMedPDF
- 16. White OB, Saint-Cyr JA, Tomlinson RD, Sharpe JA. Ocular motor deficits in Parkinson’s disease. III. Coordination of eye and head movements. Brain 1988;111(Pt 1):115–129.ArticlePubMedPDF
- 17. White OB, Saint-Cyr JA, Sharpe JA. Ocular motor deficits in Parkinson’s disease. I. The horizontal vestibulo-ocular reflex and its regulation. Brain 1983;106(Pt 3):555–570.ArticlePubMedPDF
- 18. Rottach KG, Riley DE, DiScenna AO, Zivotofsky AZ, Leigh RJ. Dynamic properties of horizontal and vertical eye movements in parkinsonian syndromes. Ann Neurol 1996;39:368–377.ArticlePubMed
- 19. Litvan I. Atypical pakinsonian disorders-clinical and research aspects. New York: Humana Press; 2005.
- 20. Corin MS, Elizan TS, Bender MB. Oculomotor function in patients with Parkinson’s disease. J Neurol Sci 1972;15:251–265.ArticlePubMed
- 21. Sharpe JA, Fletcher WA, Lang AE, Zackon DH. Smooth pursuit during dose-related on-off fluctuations in Parkinson’s disease. Neurology 1987;37:1389–1392.ArticlePubMed
- 22. Gibson JM, Pimlott R, Kennard C. Ocular motor and manual tracking in Parkinson’s disease and the effect of treatment. J Neurol Neurosurg Psychiatry 1987;50:853–860.ArticlePubMedPMC
- 23. Racette BA, Gokden MS, Tychsen LS, Perlmutter JS. Convergence insufficiency in idiopathic Parkinson’s disease responsive to levodopa. Strabismus 1999;7:169–174.ArticlePubMed
- 24. Antoniades CA, Rebelo P, Kennard C, Aziz TZ, Green AL, FitzGerald JJ. Pallidal deep brain stimulation improves higher control of the oculomotor system in Parkinson’s disease. J Neurosci 2015;35:13043–13052.ArticlePubMedPMC
- 25. Antoniades CA, Carpenter RH, Temel Y. Deep brain stimulation of the subthalamic nucleus in Parkinson’s disease: similar improvements in saccadic and manual responses. Neuroreport 2012;23:179–183.ArticlePubMed
- 26. Williams DR, Lees AJ. Progressive supranuclear palsy: clinicopathological concepts and diagnostic challenges. Lancet Neurol 2009;8:270–279.ArticlePubMed
- 27. Chen AL, Riley DE, King SA, Joshi AC, Serra A, Liao K, et al. The disturbance of gaze in progressive supranuclear palsy: implications for pathogenesis. Front Neurol 2010;1:147.ArticlePubMedPMC
- 28. Otero-Millan J, Schneider R, Leigh RJ, Macknik SL, Martinez-Conde S. Saccades during attempted fixation in parkinsonian disorders and recessive ataxia: from microsaccades to square-wave jerks. PLoS One 2013;8:e58535. ArticlePubMedPMC
- 29. Rascol O, Sabatini U, Simonetta-Moreau M, Montastruc JL, Rascol A, Clanet M. Square wave jerks in parkinsonian syndromes. J Neurol Neurosurg Psychiatry 1991;54:599–602.ArticlePubMedPMC
- 30. Lee ES, Choi JY, Kim JS. Teaching video neuroimages: alternating horizontal single saccadic pulses in progressive supranuclear palsy. Neurology 2017;88:e32–e33.ArticlePubMed
- 31. Otero-Millan J, Serra A, Leigh RJ, Troncoso XG, Macknik SL, Martinez-Conde S. Distinctive features of saccadic intrusions and microsaccades in progressive supranuclear palsy. J Neurosci 2011;31:4379–4387.ArticlePubMedPMC
- 32. Bhidayasiri R, Riley DE, Somers JT, Lerner AJ, Buttner-Ennever JA, Leigh RJ. Pathophysiology of slow vertical saccades in progressive supranuclear palsy. Neurology 2001;57:2070–2077.ArticlePubMed
- 33. Quinn N. The “round the houses” sign in progressive supranuclear palsy. Ann Neurol 1996;40:951.Article
- 34. Garbutt S, Riley DE, Kumar AN, Han Y, Harwood MR, Leigh RJ. Abnormalities of optokinetic nystagmus in progressive supranuclear palsy. J Neurol Neurosurg Psychiatry 2004;75:1386–1394.ArticlePubMedPMC
- 35. Pierrot-Deseilligny C, Rivaud S, Pillon B, Fournier E, Agid Y. Lateral visually-guided saccades in progressive supranuclear palsy. Brain 1989;112(Pt 2):471–487.ArticlePubMedPDF
- 36. Goffinet AM, De Volder AG, Gillain C, Rectem D, Bol A, Michel C, et al. Positron tomography demonstrates frontal lobe hypometabolism in progressive supranuclear palsy. Ann Neurol 1989;25:131–139.ArticlePubMed
- 37. Das VE, Leigh RJ. Visual-vestibular interaction in progressive supranuclear palsy. Vision Res 2000;40:2077–2081.ArticlePubMed
- 38. Liao K, Wagner J, Joshi A, Estrovich I, Walker MF, Strupp M, et al. Why do patients with PSP fall? Evidence for abnormal otolith responses. Neurology 2008;70:802–809.ArticlePubMed
- 39. Kitthaweesin K, Riley DE, Leigh RJ. Vergence disorders in progressive supranuclear palsy. Ann N Y Acad Sci 2002;956:504–507.ArticlePubMed
- 40. Golbe LI, Davis PH, Lepore FE. Eyelid movement abnormalities in progressive supranuclear palsy. Mov Disord 1989;4:297–302.ArticlePubMed
- 41. Dehaene I. Apraxia of eyelid opening in progressive supranuclear palsy. Ann Neurol 1984;15:115–116.ArticlePubMed
- 42. Armstrong MJ, Litvan I, Lang AE, Bak TH, Bhatia KP, Borroni B, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 2013;80:496–503.ArticlePubMedPMC
- 43. Rinne JO, Lee MS, Thompson PD, Marsden CD. Corticobasal degeneration. A clinical study of 36 cases. Brain 1994;117(Pt 5):1183–1196.ArticlePubMedPDF
- 44. Riley DE, Lang AE, Lewis A, Resch L, Ashby P, Hornykiewicz O, et al. Cortical-basal ganglionic degeneration. Neurology 1990;40:1203–1212.ArticlePubMed
- 45. Fanciulli A, Wenning GK. Multiple-system atrophy. N Engl J Med 2015;372:249–263.ArticlePubMed
- 46. Bertholon P, Bronstein AM, Davies RA, Rudge P, Thilo KV. Positional down beating nystagmus in 50 patients: cerebellar disorders and possible anterior semicircular canalithiasis. J Neurol Neurosurg Psychiatry 2002;72:366–372.ArticlePubMedPMC
- 47. Lee JY, Lee WW, Kim JS, Kim HJ, Kim JK, Jeon BS. Perverted headshaking and positional downbeat nystagmus in patients with multiple system atrophy. Mov Disord 2009;24:1290–1295.ArticlePubMed
- 48. Brett FM, Henson C, Staunton H. Familial diffuse Lewy body disease, eye movement abnormalities, and distribution of pathology. Arch Neurol 2002;59:464–467.ArticlePubMed
- 49. de Bruin VM, Lees AJ, Daniel SE. Diffuse Lewy body disease presenting with supranuclear gaze palsy, parkinsonism, and dementia: a case report. Mov Disord 1992;7:355–358.ArticlePubMed
- 50. Durr A. Autosomal dominant cerebellar ataxias: polyglutamine expansions and beyond. Lancet Neurol 2010;9:885–894.ArticlePubMed
- 51. Buttner N, Geschwind D, Jen JC, Perlman S, Pulst SM, Baloh RW. Oculomotor phenotypes in autosomal dominant ataxias. Arch Neurol 1998;55:1353–1357.ArticlePubMed
- 52. Pula JH, Gomez CM, Kattah JC. Ophthalmologic features of the common spinocerebellar ataxias. Curr Opin Ophthalmol 2010;21:447–453.ArticlePubMed
- 53. Zee DS, Yee RD, Cogan DG, Robinson DA, Engel WK. Ocular motor abnormalities in hereditary cerebellar ataxia. Brain 1976;99:207–234.ArticlePubMedPDF
- 54. Moschner C, Perlman S, Baloh RW. Comparison of oculomotor findings in the progressive ataxia syndromes. Brain 1994;117(Pt 1):15–25.ArticlePubMedPDF
- 55. Durr A, Chneiweiss H, Khati C, Stevanin G, Cancel G, Feingold J, et al. Phenotypic variability in autosomal dominant cerebellar ataxia type I is unrelated to genetic heterogeneity. Brain 1993;116(Pt 6):1497–1508.ArticlePubMedPDF
- 56. Lewis RF, Zee DS. Ocular motor disorders associated with cerebellar lesions: pathophysiology and topical localization. Rev Neurol (Paris) 1993;149:665–677.PubMed
- 57. Zee DS, Yamazaki A, Butler PH, Gücer G. Effects of ablation of flocculus and paraflocculus of eye movements in primate. J Neurophysiol 1981;46:878–899.ArticlePubMed
- 58. Zee DS, Leigh RJ, Mathieu-Millaire F. Cerebellar control of ocular gaze stability. Ann Neurol 1980;7:37–40.ArticlePubMed
- 59. Park HK, Kim JS, Strupp M, Zee DS. Isolated floccular infarction: impaired vestibular responses to horizontal head impulse. J Neurol 2013;260:1576–1582.ArticlePubMedPDF
- 60. Lisberger SG, Miles FA, Zee DS. Signals used to compute errors in monkey vestibuloocular reflex: possible role of flocculus. J Neurophysiol 1984;52:1140–1153.ArticlePubMed
- 61. Huh YE, Kim JS. Patterns of spontaneous and head-shaking nystagmus in cerebellar infarction: imaging correlations. Brain 2011;134(Pt 12):3662–3671.ArticlePubMedPDF
- 62. Lee SU, Choi JY, Kim HJ, Park JJ, Zee DS, Kim JS. Impaired tilt suppression of post-rotatory nystagmus and cross-coupled head-shaking nystagmus in cerebellar lesions: image mapping study. Cerebellum 2017;16:95–102.ArticlePubMedPDF
- 63. Choi SY, Park SH, Kim HJ, Kim JS. Paraneoplastic downbeat nystagmus associated with cerebellar hypermetabolism especially in the nodulus. J Neurol Sci 2014;343:187–191.ArticlePubMed
- 64. Jeong HS, Oh JY, Kim JS, Kim J, Lee AY, Oh SY. Periodic alternating nystagmus in isolated nodular infarction. Neurology 2007;68:956–957.ArticlePubMed
- 65. Moon IS, Kim JS, Choi KD, Kim MJ, Oh SY, Lee H, et al. Isolated nodular infarction. Stroke 2009;40:487–491.ArticlePubMed
- 66. Nam J, Kim S, Huh Y, Kim JS. Ageotropic central positional nystagmus in nodular infarction. Neurology 2009;73:1163.ArticlePubMed
- 67. Oh YM, Choi KD, Oh SY, Kim JS. Periodic alternating nystagmus with circumscribed nodular lesion. Neurology 2006;67:399.ArticlePubMed
- 68. Takagi M, Tamargo R, Zee DS. Effects of lesions of the cerebellar oculomotor vermis on eye movements in primate: binocular control. Prog Brain Res 2003;142:19–33.ArticlePubMed
- 69. Takagi M, Zee DS, Tamargo RJ. Effects of lesions of the oculomotor cerebellar vermis on eye movements in primate: smooth pursuit. J Neurophysiol 2000;83:2047–2062.ArticlePubMed
- 70. Moscovich M, Okun MS, Favilla C, Figueroa KP, Pulst SM, Perlman S, et al. Clinical evaluation of eye movements in spinocerebellar ataxias: a prospective multicenter study. J Neuroophthalmol 2015;35:16–21.ArticlePubMedPMC
- 71. Geiner S, Horn AK, Wadia NH, Sakai H, Buttner-Ennever JA. The neuroanatomical basis of slow saccades in spinocerebellar ataxia type 2 (Wadia-subtype). Prog Brain Res 2008;171:575–581.ArticlePubMed
- 72. Rodriguez-Labrada R, Velazquez-Perez L, Auburger G, Ziemann U, Canales-Ochoa N, Medrano-Montero J, et al. Spinocerebellar ataxia type 2: measures of saccade changes improve power for clinical trials. Mov Disord 2016;31:570–578.ArticlePubMed
- 73. Raposo M, Vasconcelos J, Bettencourt C, Kay T, Coutinho P, Lima M. Nystagmus as an early ocular alteration in Machado-Joseph disease (MJD/SCA3). BMC Neurol 2014;14:17.ArticlePubMedPMCPDF
- 74. Stevanin G, Durr A, David G, Didierjean O, Cancel G, Rivaud S, et al. Clinical and molecular features of spinocerebellar ataxia type 6. Neurology 1997;49:1243–1246.ArticlePubMed
- 75. Stefanescu MR, Dohnalek M, Maderwald S, Thürling M, Minnerop M, Beck A, et al. Structural and functional MRI abnormalities of cerebellar cortex and nuclei in SCA3, SCA6 and Friedreich’s ataxia. Brain 2015;138(Pt 5):1182–1197.ArticlePubMedPMCPDF
- 76. Kim JS, Kim JS, Youn J, Seo DW, Jeong Y, Kang JH, et al. Ocular motor characteristics of different subtypes of spinocerebellar ataxia: distinguishing features. Mov Disord 2013;28:1271–1277.ArticlePubMed
- 77. Huh YE, Kim JS, Kim HJ, Park SH, Jeon BS, Kim JM, et al. Vestibular performance during high-acceleration stimuli correlates with clinical decline in SCA6. Cerebellum 2015;14:284–291.ArticlePubMedPDF
- 78. Enevoldson TP, Sanders MD, Harding AE. Autosomal dominant cerebellar ataxia with pigmentary macular dystrophy. A clinical and genetic study of eight families. Brain 1994;117(Pt 3):445–460.ArticlePubMedPDF
- 79. Horton LC, Frosch MP, Vangel MG, Weigel-DiFranco C, Berson EL, Schmahmann JD. Spinocerebellar ataxia type 7: clinical course, phenotype-genotype correlations, and neuropathology. Cerebellum 2013;12:176–193.ArticlePubMedPMCPDF
- 80. Yabe I, Matsushima M, Yoshida K, Ishikawa K, Shirai S, Takahashi I, et al. Rare frequency of downbeat positioning nystagmus in spinocerebellar ataxia type 31. J Neurol Sci 2015;350:90–92.ArticlePubMed
- 81. Campuzano V, Montermini L, Lutz Y, Cova L, Hindelang C, Jiralerspong S, et al. Frataxin is reduced in Friedreich ataxia patients and is associated with mitochondrial membranes. Hum Mol Genet 1997;6:1771–1780.ArticlePubMedPDF
- 82. Fahey MC, Cremer PD, Aw ST, Millist L, Todd MJ, White OB, et al. Vestibular, saccadic and fixation abnormalities in genetically confirmed Friedreich ataxia. Brain 2008;131(Pt 4):1035–1045.ArticlePubMedPDF
- 83. Spieker S, Schulz JB, Petersen D, Fetter M, Klockgether T, Dichgans J. Fixation instability and oculomotor abnormalities in Friedreich’s ataxia. J Neurol 1995;242:517–521.ArticlePubMedPDF
- 84. Kirkham TH, Guitton D, Katsarkas A, Kline LB, Andermann E. Oculomotor abnormalities in Friedreich’s ataxia. Can J Neurol Sci 1979;6:167–172.ArticlePubMed
- 85. Furman JM, Perlman S, Baloh RW. Eye movements in Friedreich’s ataxia. Arch Neurol 1983;40:343–346.ArticlePubMed
- 86. Monday LA, Lemieux B, St Vincent H, Barbeau A. Clinical and electronystagmographic findings in Friedreich’s ataxia. Can J Neurol Sci 1978;5:71–73.ArticlePubMed
- 87. Ell J, Prasher D, Rudge P. Neuro-otological abnormalities in Friedreich’s ataxia. J Neurol Neurosurg Psychiatry 1984;47:26–32.ArticlePubMedPMC
- 88. Jen JC, Graves TD, Hess EJ, Hanna MG, Griggs RC, Baloh RW; CINCH investigators. Primary episodic ataxias: diagnosis, pathogenesis and treatment. Brain 2007;130(Pt 10):2484–2493.ArticlePubMedPDF
- 89. Jen JC, Baloh RW. Genetics of episodic ataxia. Adv Neurol 2002;89:459–461.PubMed
- 90. Choi KD, Kim JS, Kim HJ, Jung I, Jeong SH, Lee SH, et al. Genetic variants associated with episodic ataxia in Korea. Sci Rep 2017;7:13855.ArticlePubMedPMCPDF
- 91. Jen JC, Wan J. Episodic ataxias. Handb Clin Neurol 2018;148:521–529.ArticlePubMed
- 92. Kim HJ, Kim JS, Choi JH, Shin JH, Choi KD, Zee DS. Rebound upbeat nystagmus after lateral gaze in episodic ataxia type 2. Cerebellum 2014;13:411–413.ArticlePubMedPDF
- 93. Choi KD, Jen JC, Choi SY, Shin JH, Kim HS, Kim HJ, et al. Late-onset episodic ataxia associated with SLC1A3 mutation. J Hum Genet 2017;62:443–446.ArticlePubMedPDF
- 94. Choi JH, Seo JD, Choi YR, Kim MJ, Shin JH, Kim JS, et al. Exercise-induced downbeat nystagmus in a Korean family with a nonsense mutation in CACNA1A. Neurol Sci 2015;36:1393–1396.ArticlePubMedPDF
- 95. Levy A, Lang AE. Ataxia-telangiectasia: a review of movement disorders, clinical features, and genotype correlations. Mov Disord 2018;33:1372.ArticlePubMed
- 96. Lohmann E, Kruger S, Hauser AK, Hanagasi H, Guven G, Erginel-Unaltuna N, et al. Clinical variability in ataxia-telangiectasia. J Neurol 2015;262:1724–1727.ArticlePubMedPDF
- 97. Boder E, Sedgwick RP. Ataxia-telangiectasia; a familial syndrome of progressive cerebellar ataxia, oculocutaneous telangiectasia and frequent pulmonary infection. Pediatrics 1958;21:526–554.PubMed
- 98. Teive HAG, Camargo CHF, Munhoz RP. More than ataxia - Movement disorders in ataxia-telangiectasia. Parkinsonism Relat Disord 2018;46:3–8.ArticlePubMed
- 99. Shaikh AG, Marti S, Tarnutzer AA, Palla A, Crawford TO, Straumann D, et al. Gaze fixation deficits and their implication in ataxia-telangiectasia. J Neurol Neurosurg Psychiatry 2009;80:858–864.ArticlePubMed
- 100. Lewis RF, Lederman HM, Crawford TO. Ocular motor abnormalities in ataxia telangiectasia. Ann Neurol 1999;46:287–295.ArticlePubMed
- 101. Baloh RW, Yee RD, Boder E. Eye movements in ataxia-telangiectasia. Neurology 1978;28:1099–1104.ArticlePubMed
- 102. Aicardi J, Barbosa C, Andermann E, Andermann F, Morcos R, Ghanem Q, et al. Ataxia-ocular motor apraxia: a syndrome mimicking ataxia-telangiectasia. Ann Neurol 1988;24:497–502.ArticlePubMed
- 103. Le Ber I, Moreira MC, Rivaud-Pechoux S, Chamayou C, Ochsner F, Kuntzer T, et al. Cerebellar ataxia with oculomotor apraxia type 1: clinical and genetic studies. Brain 2003;126:2761–2772.ArticlePubMedPDF
- 104. McKinnon PJ. DNA repair deficiency and neurological disease. Nat Rev Neurosci 2009;10:100–112.ArticlePubMedPMCPDF
- 105. Reynolds JJ, Stewart GS. A single strand that links multiple neuropathologies in human disease. Brain 2013;136(Pt 1):14–27.ArticlePubMedPDF
- 106. Quinzii CM, Kattah AG, Naini A, Akman HO, Mootha VK, DiMauro S, et al. Coenzyme Q deficiency and cerebellar ataxia associated with an aprataxin mutation. Neurology 2005;64:539–541.ArticlePubMed
- 107. Renaud M, Moreira MC, Ben Monga B, Rodriguez D, Debs R, Charles P, et al. Clinical, biomarker, and molecular delineations and genotype-phenotype correlations of ataxia with oculomotor apraxia type 1. JAMA Neurol 2018;75:495–502.ArticlePubMedPMC
- 108. Lee M, Kim NY, Huh JY, Kim YE, Kim YJ. Ataxia with oculomotor apraxia type 1 without oculomotor apraxia: a case report. J Clin Neurol 2016;12:126–128.ArticlePubMed
- 109. Anheim M, Monga B, Fleury M, Charles P, Barbot C, Salih M, et al. Ataxia with oculomotor apraxia type 2: clinical, biological and genotype/phenotype correlation study of a cohort of 90 patients. Brain 2009;132(Pt 10):2688–2698.ArticlePubMedPDF
- 110. Le Ber I, Bouslam N, Rivaud-Péchoux S, Guimarães J, Benomar A, Chamayou C, et al. Frequency and phenotypic spectrum of ataxia with oculomotor apraxia 2: a clinical and genetic study in 18 patients. Brain 2004;127:759–767.ArticlePubMedPDF
- 111. Moreira MC, Klur S, Watanabe M, Németh AH, Le Ber I, Moniz JC, et al. Senataxin, the ortholog of a yeast RNA helicase, is mutant in ataxiaocular apraxia 2. Nat Genet 2004;36:225–227.ArticlePubMedPDF
- 112. Tazir M, Ali-Pacha L, M’Zahem A, Delaunoy JP, Fritsch M, Nouioua S, et al. Ataxia with oculomotor apraxia type 2: a clinical and genetic study of 19 patients. J Neurol Sci 2009;278:77–81.ArticlePubMed
- 113. Panouillères M, Frismand S, Sillan O, Urquizar C, Vighetto A, Pélisson D, et al. Saccades and eye-head coordination in ataxia with oculomotor apraxia type 2. Cerebellum 2013;12:557–567.ArticlePubMedPDF
- 114. Wray SH, Martinez-Hernandez E, Dalmau J, Maheshwari A, Chen A, King S, et al. Paraneoplastic upbeat nystagmus. Neurology 2011;77:691–693.ArticlePubMedPMC
- 115. Gordon LK. Paraneoplastic syndromes in neuro-ophthalmology. J Neuroophthalmol 2015;35:306–314.ArticlePubMed
- 116. Ko MW, Dalmau J, Galetta SL. Neuro-ophthalmologic manifestations of paraneoplastic syndromes. J Neuroophthalmol 2008;28:58–68.ArticlePubMed
- 117. Rizvi MT, Cameron L, Kilbane C, Shaikh AG. Paraneoplastic seesaw nystagmus and opsoclonus provides evidence for hyperexcitable reciprocally innervating mesencephalic network. J Neurol Sci 2018;390:239–245.ArticlePubMed
- 118. Lasker AG, Zee DS. Ocular motor abnormalities in Huntington’s disease. Vision Res 1997;37:3639–3645.ArticlePubMed
- 119. Antoniades CA, Xu Z, Mason SL, Carpenter RH, Barker RA. Huntington’s disease: changes in saccades and hand-tapping over 3 years. J Neurol 2010;257:1890–1898.ArticlePubMedPDF
- 120. Patel SS, Jankovic J, Hood AJ, Jeter CB, Sereno AB. Reflexive and volitional saccades: biomarkers of Huntington disease severity and progression. J Neurol Sci 2012;313:35–41.ArticlePubMed
- 121. Leigh RJ, Newman SA, Folstein SE, Lasker AG, Jensen BA. Abnormal ocular motor control in Huntington’s disease. Neurology 1983;33:1268–1275.ArticlePubMed
- 122. Lasker AG, Zee DS, Hain TC, Folstein SE, Singer HS. Saccades in Huntington’s disease: initiation defects and distractibility. Neurology 1987;37:364–370.ArticlePubMed
- 123. Hicks SL, Robert MP, Golding CV, Tabrizi SJ, Kennard C. Oculomotor deficits indicate the progression of Huntington’s disease. Prog Brain Res 2008;171:555–558.ArticlePubMed
- 124. Helmchen C, Hagenow A, Miesner J, Sprenger A, Rambold H, Wenzelburger R, et al. Eye movement abnormalities in essential tremor may indicate cerebellar dysfunction. Brain 2003;126:1319–1332.ArticlePubMedPDF
- 125. Kim YE, Kim JS, Yang HJ, Yun JY, Kim HJ, Ehm G, et al. Perverted head-shaking and positional downbeat nystagmus in essential tremor. Cerebellum 2016;15:152–158.ArticlePubMedPDF
- 126. Gitchel GT, Wetzel PA, Baron MS. Slowed saccades and increased square wave jerks in essential tremor. Tremor Other Hyperkinet Mov (N Y) 2013;3:tre-03-178-4116-2. Article
- 127. Ingster-Moati I, Bui Quoc E, Pless M, Djomby R, Orssaud C, Guichard JP, et al. Ocular motility and Wilson’s disease: a study on 34 patients. J Neurol Neurosurg Psychiatry 2007;78:1199–1201.ArticlePubMedPMC
- 128. Jung HK, Choi SY, Kim JM, Kim JS. Selective slowing of downward saccades in Wilson’s disease. Parkinsonism Relat Disord 2013;19:134–135.ArticlePubMed
- 129. Averbuch-Heller L, Paulson GW, Daroff RB, Leigh RJ. Whipple’s disease mimicking progressive supranuclear palsy: the diagnostic value of eye movement recording. J Neurol Neurosurg Psychiatry 1999;66:532–535.ArticlePubMedPMC
- 130. Schwartz MA, Selhorst JB, Ochs AL, Beck RW, Campbell WW, Harris JK, et al. Oculomasticatory myorhythmia: a unique movement disorder occurring in Whipple’s disease. Ann Neurol 1986;20:677–683.ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- The semicircular canal function is preserved with little impact on falls in patients with mild Parkinson's disease
Jun-Pyo Hong, Hanim Kwon, Euyhyun Park, Sun-Uk Lee, Chan-Nyoung Lee, Byung-Jo Kim, Ji-Soo Kim, Kun-Woo Park
Parkinsonism & Related Disorders.2024; 118: 105933. CrossRef - Abnormal Ocular Movement in the Early Stage of Multiple-System Atrophy With Predominant Parkinsonism Distinct From Parkinson’s Disease
Hong Zhou, Luhua Wei, Yanyan Jiang, Xia Wang, Yunchuang Sun, Fan Li, Jing Chen, Wei Sun, Lin Zhang, Guiping Zhao, Zhaoxia Wang
Journal of Clinical Neurology.2024; 20(1): 37. CrossRef - Smartphone video nystagmography using convolutional neural networks: ConVNG
Maximilian U. Friedrich, Erich Schneider, Miriam Buerklein, Johannes Taeger, Johannes Hartig, Jens Volkmann, Robert Peach, Daniel Zeller
Journal of Neurology.2023; 270(5): 2518. CrossRef - Midbrain and pons MRI shape analysis and its clinical and CSF correlates in degenerative parkinsonisms: a pilot study
C. Painous, S. Pascual-Diaz, E. Muñoz-Moreno, V. Sánchez, JC. Pariente, A. Prats-Galino, M. Soto, M. Fernández, A. Pérez-Soriano, A. Camara, E. Muñoz, F. Valldeoriola, N. Caballol, C. Pont-Sunyer, N. Martin, M. Basora, M. Tio, J. Rios, MJ. Martí, N. Barga
European Radiology.2023; 33(7): 4540. CrossRef - Saw-tooth vertical saccades in progressive supranuclear palsy
Min-Ku Kim, Dongje Lee, Xu Yang, Hyo-Jung Kim, Jeong‑Yoon Choi, Ji-Soo Kim
Journal of Neurology.2023; 270(7): 3644. CrossRef - Non-Verbal Social Skills Assessment in Rett Syndrome: a Systematic Review
Xin-yan Zhang, Karen Spruyt
Review Journal of Autism and Developmental Disorders.2023;[Epub] CrossRef - Spinocerebellar ataxia type 14 (SCA14) in an Argentinian family: a case report
Niharika Duggirala, Kathie J. Ngo, Sabrina M. Pagnoni, Alberto L. Rosa, Brent L. Fogel
Journal of Medical Case Reports.2023;[Epub] CrossRef - Abnormal eye movements in Parkinson's disease: From experimental study to clinical application
Han Li, Xue Zhang, Yong Yang, Anmu Xie
Parkinsonism & Related Disorders.2023; 115: 105791. CrossRef - Oculomotor Abnormalities in a Sheep (Ovis aries) Model of Huntington’s Disease: Towards a Biomarker for Assessing Therapeutic Efficacy
Sebastian D. McBride, Jan Ober, Jacek Dylak, William Schneider, A. Jennifer Morton
Journal of Huntington's Disease.2023; 12(3): 189. CrossRef - Head-impulse tests aid in differentiation of multiple system atrophy from Parkinson’s disease
Jae-Gyum Kim, Sung-Hwan Kim, Sun-Uk Lee, Chan-Nyoung Lee, Byung-Jo Kim, Ji-Soo Kim, Kun-Woo Park
Journal of Neurology.2022; 269(6): 2972. CrossRef - Overt Oculomotor Behavior Reveals Covert Temporal Predictions
Alessandro Tavano, Sonja A. Kotz
Frontiers in Human Neuroscience.2022;[Epub] CrossRef - Eye tracking identifies biomarkers in α-synucleinopathies versus progressive supranuclear palsy
Mahboubeh Habibi, Wolfgang H. Oertel, Brian J. White, Donald C. Brien, Brian C. Coe, Heidi C. Riek, Julia Perkins, Rachel Yep, Laurent Itti, Lars Timmermann, Christoph Best, Elisabeth Sittig, Annette Janzen, Douglas P. Munoz
Journal of Neurology.2022; 269(9): 4920. CrossRef - A Case of AOA2 With Compound Heterozygous SETX Mutations
Hee Jin Chang, Ryul Kim, Minchae Kim, Jangsup Moon, Man Jin Kim, Han-Joon Kim
Journal of Movement Disorders.2022; 15(2): 178. CrossRef - Reading Warm-Up, Reading Skill, and Reading Prosody When Reading the My Grandfather Passage: An Exploratory Study Born Out of the Motor Planning Theory of Prosody and Reading Prosody Research
Frank Boutsen, Eunsun Park, Justin D. Dvorak
Journal of Speech, Language, and Hearing Research.2022; 65(6): 2047. CrossRef - Eye movement abnormalities in neurodegenerative langerhans cell histiocytosis
Lila Autier, Bertrand Gaymard, Eléonore Bayen, Antoine Del Cul, Fleur Cohen-Aubart, Nadine Martin-Duverneuil, Julien Haroche, Karima Mokhtari, Sébastien Héritier, Jean Donadieu, Khê Hoang-Xuan, Ahmed Idbaih
Neurological Sciences.2022; 43(11): 6539. CrossRef - Multiple step saccades in simply reactive saccades could serve as a complementary biomarker for the early diagnosis of Parkinson’s disease
Wenbo Ma, Min Li, Junru Wu, Zhihao Zhang, Fangfang Jia, Mingsha Zhang, Hagai Bergman, Xuemei Li, Zhipei Ling, Xin Xu
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - The effects of age and sex on the incidence of multiple step saccades and corrective saccades
Wenbo Ma, Mingsha Zhang
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - Oculomotor impairments in de novo Parkinson’s disease
Meng-Xi Zhou, Qin Wang, Yin Lin, Qian Xu, Li Wu, Ya-Jing Chen, Yu-Han Jiang, Qing He, Lei Zhao, You-Rong Dong, Jian-Ren Liu, Wei Chen
Frontiers in Aging Neuroscience.2022;[Epub] CrossRef - Eye movement especially vertical oculomotor impairment as an aid to assess Parkinson’s disease
JianYuan Zhang, Bin Zhang, QingGuo Ren, Qing Zhong, Ye Li, GuoTao Liu, XiaoTian Ma, CuiPing Zhao
Neurological Sciences.2021; 42(6): 2337. CrossRef - Abnormalities of smooth pursuit in Parkinson’s disease: A systematic review
Karen Frei
Clinical Parkinsonism & Related Disorders.2021; 4: 100085. CrossRef - Central nystagmus in progressive supranuclear palsy: A neglected clinical feature?
Maja Klarendic, Manja Hribar, Nina Bozanic Urbancic, Nina Zupancic, Milica G. Kramberger, Maja Trost, Saba Battelino, Diego Kaski, Maja Kojovic
Parkinsonism & Related Disorders.2021; 84: 15. CrossRef - Analysis of vertical eye movements in Parkinson’s disease and its potential for diagnosis
Sajjad Farashi
Applied Intelligence.2021; 51(11): 8260. CrossRef - Abnormal eye movements in parkinsonism: a historical view
Tereza Ko, Augusto Mädke Brenner, Nicholas Pili Monteiro, Mariana Severo Debastiani, Alberto Chitolina Nesello, Arlete Hilbig
Arquivos de Neuro-Psiquiatria.2021; 79(5): 457. CrossRef - Morphological classification and changes in dementia (Review)
Alexandra Tudor, Antonia Vasile, Simona Trifu, Mihai Cristea
Experimental and Therapeutic Medicine.2021;[Epub] CrossRef - BCAP31-related syndrome: The first de novo report
Berardo Rinaldi, Evelien Van Hoof, Anniek Corveleyn, Annick Van Cauter, Thomy de Ravel
European Journal of Medical Genetics.2020; 63(2): 103732. CrossRef - Hypomania and saccadic changes in Parkinson’s disease: influence of D2 and D3 dopaminergic signalling
Esther A. Pelzer, Barbara Dillenburger, Sophie Grundmann, Vladimir Iliaev, Sophie Aschenberg, Corina Melzer, Martin Hess, Gereon R. Fink, Carsten Eggers, Marc Tittgemeyer, Lars Timmermann
npj Parkinson's Disease.2020;[Epub] CrossRef - Management of Visual Dysfunction in Patients with Parkinson’s Disease
Joseph Savitt, Rachid Aouchiche, Bastiaan R. Bloem, Patrik Brundin
Journal of Parkinson's Disease.2020; 10(s1): S49. CrossRef - Ocular motor manifestations of movement disorders
Grace F. Crotty, Bart K. Chwalisz
Current Opinion in Ophthalmology.2019; 30(6): 443. CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite