Articles
- Page Path
- HOME > J Mov Disord > Volume 6(1); 2013 > Article
-
Case Report
Ventricular Bigeminy after Subcutaneous Administration of Apomorphine in a Patient with Refractory Parkinson’s Disease: A Case Report - Anastasia N. Kaminiotia, Georgios T. Nikitasb, Apostolos K. Terlisa, Athanasios G. Manolisb, Thomas Thomaidesa, Aggeliki N. Panousopouloua
-
Journal of Movement Disorders 2013;6(1):9-12.
DOI: https://doi.org/10.14802/jmd.13002
Published online: April 30, 2013
aDepartments of Neurology, Red Cross Hospital, Athens, Greece
bCardiology, Red Cross Hospital, Athens, Greece
- Corresponding author: Aggeliki N. Panousopoulou Department of Neurology, Red Cross Hospital, Athens, Greece Tel +30-210-641-4705, Fax +30-210-641-4587, E-mail andakamin@yahoo.gr
- The authors have no financial conflicts of interest.
• Received: January 15, 2013 • Accepted: March 15, 2013
Copyright © 2013 The Korean Movement Disorder Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 30,652 Views
- 62 Download
- 7 Crossref
ABSTRACT
- Apomorphine is a well established treatment for the management of sudden, unexpected and refractory levodopa-induced “off” states in fluctuating Parkinson’s disease either as bolus injections or as continuous infusions. Incidents of atrial fibrillation associated with the administration of the drug have been reported in the past but no incidents of ventricular arrhythmias. We report a case of ventricular bigeminy recorded in a female patient after the administration of apomorphine.
- An 87 years old woman with a history of PD for the last 18 years, treated with L-Dopa and dopamine receptor antagonists, presented with “off” episodes during the last two months despite combined medical therapy. At presentation she was treated with carbidopa 87.5 mg/day, levodopa 875 mg/day, pramipexol 1.4 mg/day and mitrazapine 7.5 mg/day. She was administered to the Neurology Department for scheduled initiation of apomorphine treatment.
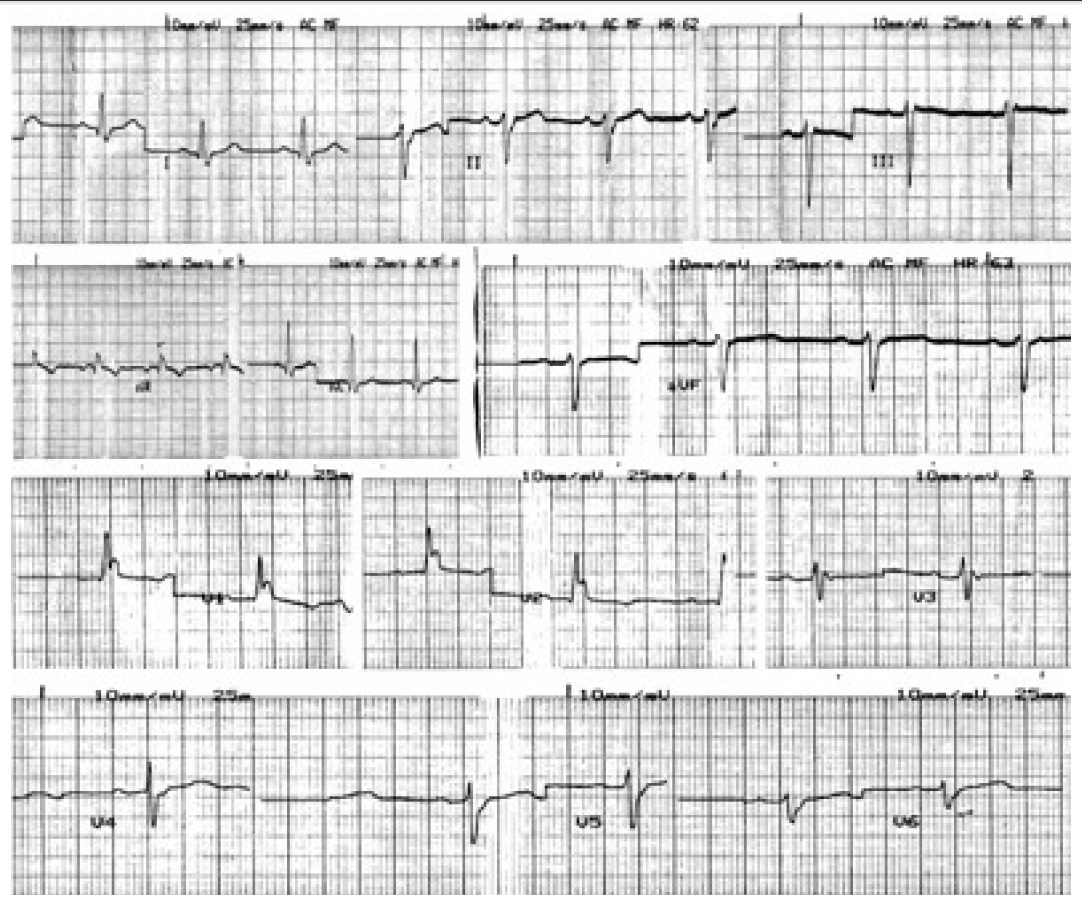
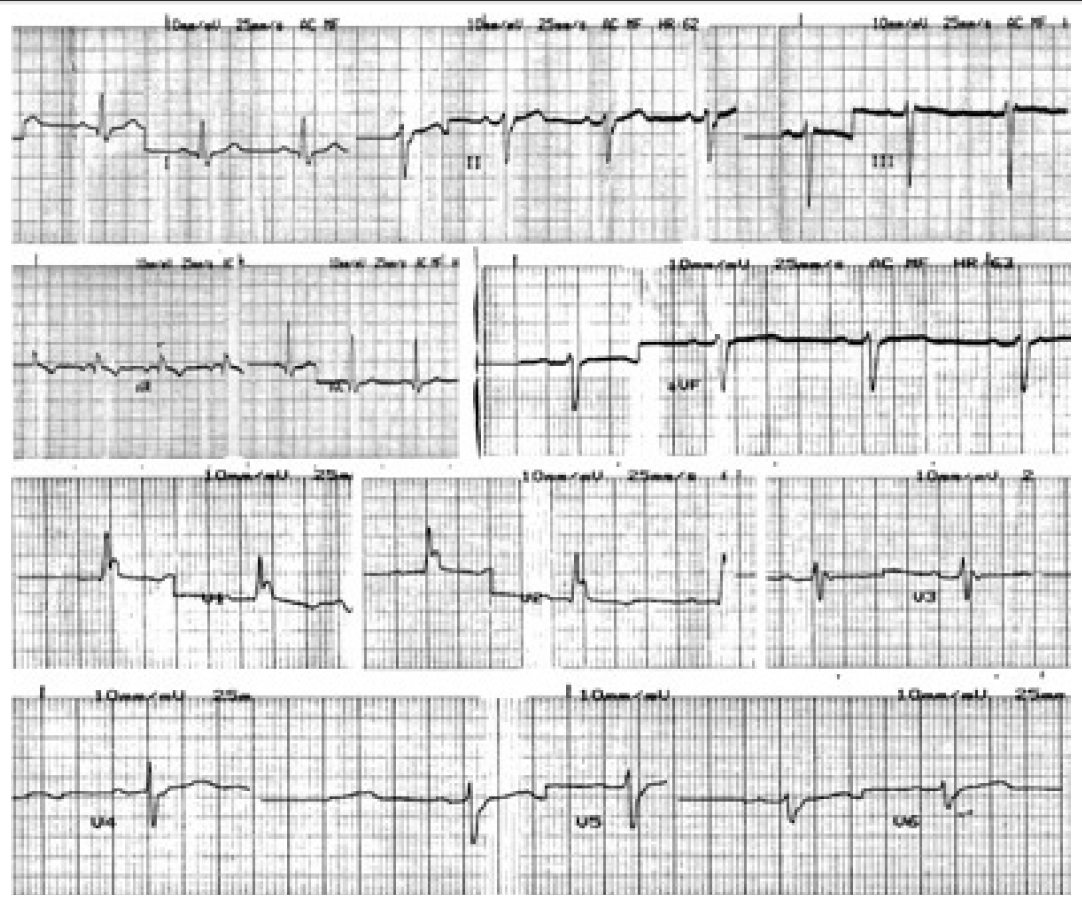
- There was no other medical history recorded, despite the advanced age of the patient, and the standard electrocardiogram (ECG) (Figure 1) at her admission was at normal range. Full blood tests, including liver and renal function and electrolytes’ levels, were conducted and no abnormal values were detected.
- The previous medical therapy was discontinued 48 hours before the initiation of apomorphine, according to the usual practice.
- The patient was haemodynamically stable before the initiation of apomorphine and the heart rate and blood pressure was repeatedly monitored.
- Ten minutes after the first subcutaneous administration of 10 mg of apomorphine, a heart rhythm disturbance was observed and recorded in a 12-lead ECG. The disturbance was ventricular bigeminy (Figure 2), with a left bundle branch block morphology of the ventricular origin QRS complexes and a frontal QRS axis at −10°. No haemodynamic instability was recorded and no symptom (dizziness, palpitations, pre-syncope or syncope) was reported by the patient. The patients’ heart rhythm and blood pressure was monitored repeatedly during the following hours.
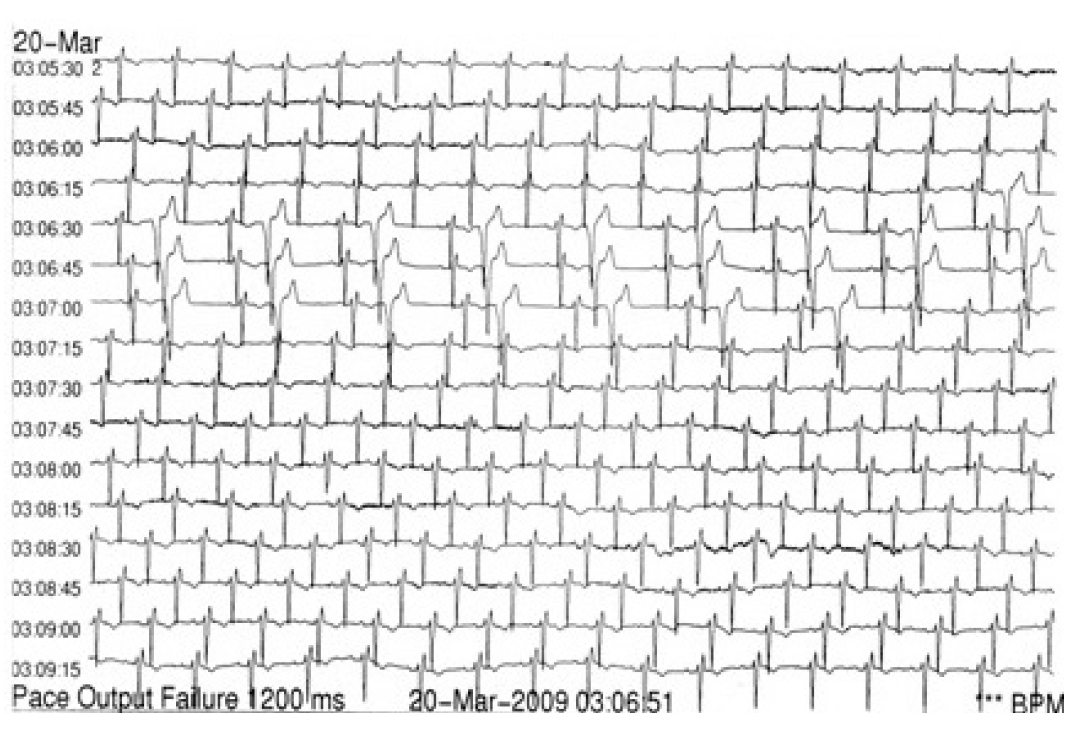
- The next day the 12-lead ECG showed ventricular bigeminy again, so we decided to monitor the patients’ heart rhythm with a 24-hour ambulatory recorder (Holter). The recording showed brief periods of ventricular bigeminy (Figure 3) and no other arrhythmias. We conducted a two dimensional echo-cardiogram without any pathological findings that could pre-dispose to the emergence of a ventricular arrhythmia (e.g., ventricular hypertrophy or any indication of ischemic heart disease), and three days later we repeated the 24-hour ambulatory recording, which revealed no heart rhythm disturbances at all.
- The patient returned to the previous treatment for PD as it is mentioned above, and no such arrhythmia was recorded in the follow up visits of the patient.
Case
- This was a rare case of heart rhythm disturbance associated with apomorphine treatment for PD.7 We conducted a review of related literature in PubMed and Medline and no such case was ever reported. The available drug information does not mention such an arrhythmia as a possible adverse event related to the drug administration. The QT interval prolongation is the only electrocardiographic disturbance previously referred,8 but in our patient no change in QT interval was observed.
- The aetiology of ventricular bigeminy in this setting is difficult to determine. The advanced age of the patient might be a predisposing factor to cardiac arrhythmias,9 although there was no previous history or symptoms of cardiac disease reported nor any structural abnormality detected.
- Apomorphine is a potent dopamine receptor agonist, and it is known that other agonists like dopamine may increase the Purkinje fibers automaticity and have a biphasic effect on action-potential duration causing atrial and ventricular tachyarrhythmias.10 Yet, there is no evidence that such a mechanism is responsible for the reported incident of bigeminy. Besides, the affinity of apomorphine to the β1 adrenergic receptors through which dopamine exerts its cardiac effects is very low.11
- Although the arrhythmia was self terminated and didn’t cause any symptoms or haemodynamic instability, it would be reasonable to monitor inpatients closely for the emergence of cardiac dysrythmias at the initiation of the treatment with apomorphine and during dose titration. As far as outpatients are concerned, regular follow up for the emergence of cardiac arrhythmias should be recommended.
Discussion
Figure 2.The electrocardiogram just after the first injection of apomorphine shows ventricular bigeminy with left bundle brunch block.
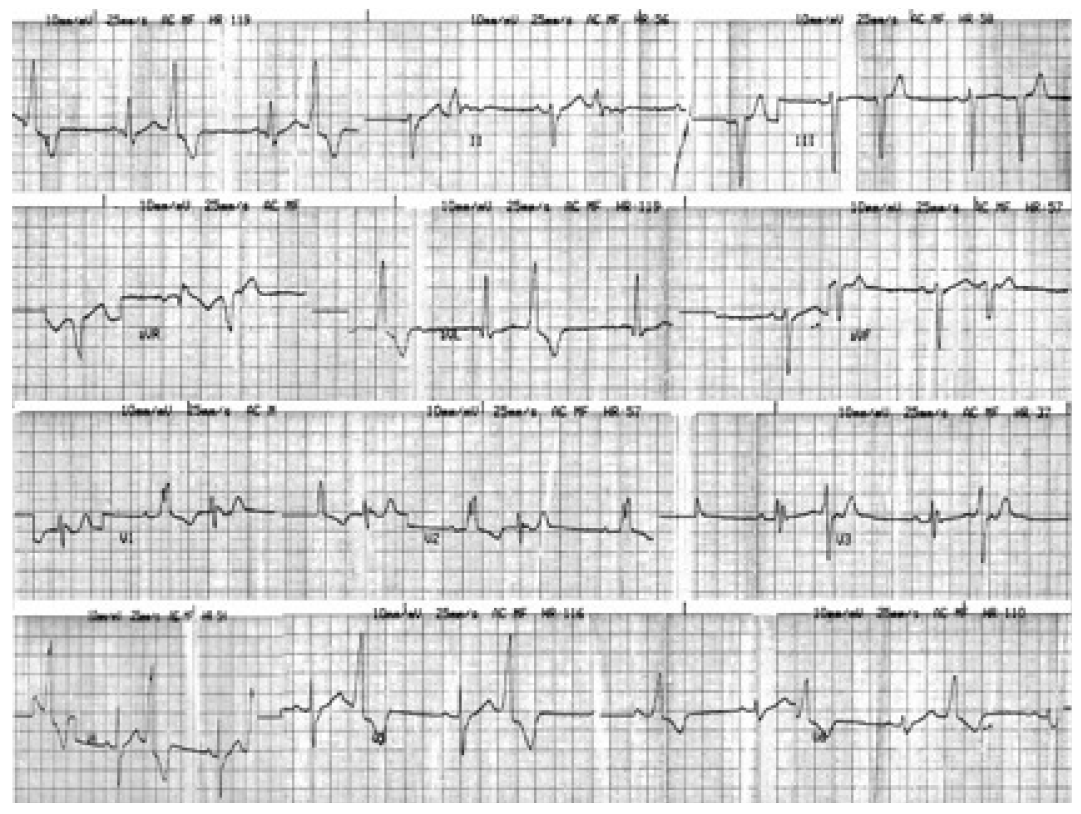

Figure 3.The 24-hour ambulatory recorder one day after the apomorphine injection shows ventricular bigeminy.


- 1. Siderowf A. Parkinson’s disease: clinical features, epidemiology and genetics. Neurol Clin 2001;19:565–578.vi.ArticlePubMed
- 2. Dressler D. [Apomorphine in the treatment of Parkinson’s Disease]. Nervenarzt 2005;76:681–689.ArticlePubMed
- 3. Antonini A, Odin P. Pros and cons of apomorphine and L-dopa continuous infusion in advanced Parkinson’s disease. Parkinsonism Relat Disord 2009;15(Suppl 4):S97–S100.ArticlePubMed
- 4. Antonini A, Tolosa E. Apomorphine and levodopa infusion therapies for advanced Parkinson’s disease: selection criteria and patient management. Expert Rev Neurother 2009;9:859–867.ArticlePubMed
- 5. Pahwa R, Koller WC, Trosch RM, Sherry JH; APO303 Study Investigators. Subcutaneous apomorphine in patients with advanced Parkinson’s disease: a dose-escalation study with randomized, double-blind, placebo-controlled crossover evaluation of a single dose. J Neurol Sci 2007;258:137–143.ArticlePubMed
- 6. Ruggieri S, Stocchi F, Carta A, Agnoli A. Side-effects of subcutaneous apomorphine in Parkinson’s disease. Lancet 1989;1:566.Article
- 7. Ardolino G, D’Adda E, Nobile-Orazio E. Recurrent atrial fibrillation after subcutaneous apomorphine. Parkinsonism Relat Disord 2008;14:173–174.author reply 175.. ArticlePubMed
- 8. Humphrey SJ, Turman CN, Curry JT, Wheeler GJ. Cardiovascular and electrocardiographic effects of the dopamine receptor agonists ropinirole, apomorphine, and PNU-142774E in conscious beagle dogs. J Cardiovasc Pharmacol 2006;47:337–347.ArticlePubMed
- 9. Hiss RG, Lamb LE. Electrocardiographic findings in 122,043 individuals. Circulation 1962;25:947–961.ArticlePubMed
- 10. Tisdale JE, Patel RV, Webb CR, Borzak S, Zarowitz BJ. Proarrhythmic effects of intravenous vasopressors. Ann Pharmacother 1995;29:269–281.ArticlePubMed
- 11. Millan MJ, Maiofiss L, Cussac D, Audinot V, Boutin JA, Newman-Tancredi A. Differential actions of antiparkinson agents at multiple classes of monoaminergic receptor. I. A multivariate analysis of the binding profiles of 14 drugs at 21 native and cloned human receptor subtypes. J Pharmacol Exp Ther 2002;303:791–804.ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Parkinson's disease advanced therapies - A systematic review: More unanswered questions than guidance
Luca Marsili, Matteo Bologna, Janis M. Miyasaki, Carlo Colosimo
Parkinsonism & Related Disorders.2021; 83: 132. CrossRef - Emerging Pharmacotherapies for Motor Symptoms in Parkinson’s Disease
Mohammad Al Majali, Michael Sunnaa, Pratap Chand
Journal of Geriatric Psychiatry and Neurology.2021; 34(4): 263. CrossRef - Safety and Tolerability of Pharmacotherapies for Parkinson’s Disease in Geriatric Patients
Martin Klietz, Stephan Greten, Florian Wegner, Günter U. Höglinger
Drugs & Aging.2019; 36(6): 511. CrossRef - Pharmacological Insights into the Use of Apomorphine in Parkinson’s Disease: Clinical Relevance
Manon Auffret, Sophie Drapier, Marc Vérin
Clinical Drug Investigation.2018; 38(4): 287. CrossRef - Cardiac abnormalities in Parkinson’s disease and Parkinsonism
Fulvio A. Scorza, Ana C. Fiorini, Carla A. Scorza, Josef Finsterer
Journal of Clinical Neuroscience.2018; 53: 1. CrossRef - Apomorphine en perfusion sous-cutanée continue dans la maladie de Parkinson : le point de vue pharmacologique
M. Auffret, S. Drapier, M. Vérin, P. Sauleau
Pratique Neurologique - FMC.2016; 7(1): 25. CrossRef - Practical management of adverse events related to apomorphine therapy
Roongroj Bhidayasiri, Pedro J. Garcia Ruiz, Tove Henriksen
Parkinsonism & Related Disorders.2016;[Epub] CrossRef
Comments on this article
 KMDS
KMDS
 E-submission
E-submission
 PubReader
PubReader ePub Link
ePub Link Cite
Cite